Nuclear Chemistry Part 1 Radiation Basics Part 2

Nuclear Chemistry Part 1: Radiation Basics Part 2: Types of Nuclear Reactions

Part 1: Radiation Basics
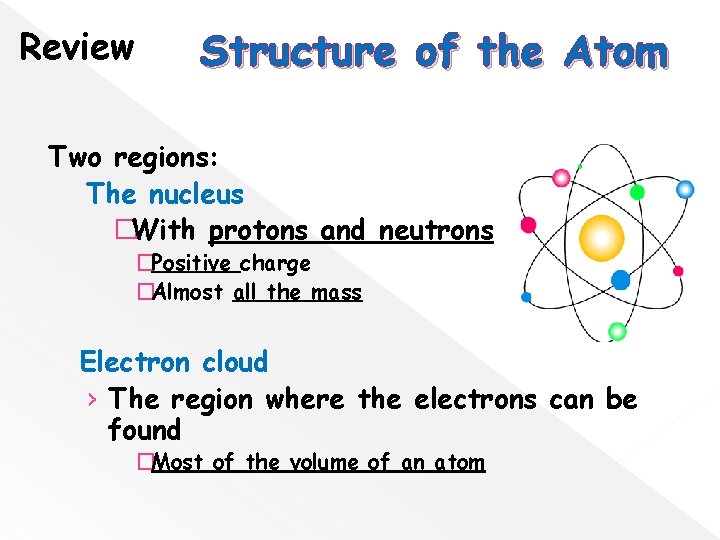
Review Structure of the Atom Two regions: The nucleus �With protons and neutrons �Positive charge �Almost all the mass Electron cloud › The region where the electrons can be found �Most of the volume of an atom

Chemical Reactions VS Nuclear Reactions Chemical 1. Occur when bonds are broken or formed 2. Atoms keep same identity but might gain, lose, or share electrons 3. Involve only valence electrons 4. Associated with small energy changes Nuclear 1. Occur when nuclei combine, split, and emit radiation 2. Atoms of one element are often converted into atoms of another element 3. Involves protons + neutrons 4. Associated with large energy changes

Radioisotopes � Radioisotopes: isotopes of atoms with unstable nuclei that undergo radioactive decay

Radioactivity � Radioactivity: process by which materials emit particles or rays › Also called radioactive decay �Involves the conversion of an element into another element = transmutation �Nuclei of atoms are joined or separated �Energy is released

Radiation Video � Is radiation dangerous? � https: //www. youtube. com/watch? v=z. I 2 v. Rw. F Kn. HQ

Nuclear Stability � Strong Nuclear Force: strong attractive forces between nucleons (protons and neutrons) that occur over very short distances � In stable nuclei, the attractive forces (between protons and neutrons) are greater than the repulsive forces.

Stable VS Unstable Nuclei � Stable: generally have equal numbers of protons and neutrons � Unstable: # of neutrons is significantly less or greater than the # of protons › Heavier elements tend to have less protons than neutrons

Types of Radiation emitted during radioactive decay. Property Alpha Radiation Beta Radiation Gamma Radiation Symbol α β γ Composition Alpha particles Beta particles High-energy electromagnetic radiation Description of Radiation Helium Electrons 0 e -1 Photons Charge +2 -1 0 Mass 6. 64 x 10 -27 kg 9. 11 x 10 -31 kg 0 Relative penetrating power Blocked by paper (low) Blocked by metal foil (medium) Not completely blocked by lead or concrete (high)

Alpha Particles � Have the same composition as a helium nucleus – with a charge of +2 � Are emitted by nuclei with 84 or more protons (Polonium and above) � Have low penetrating power – can be stopped by a sheet of paper or skin
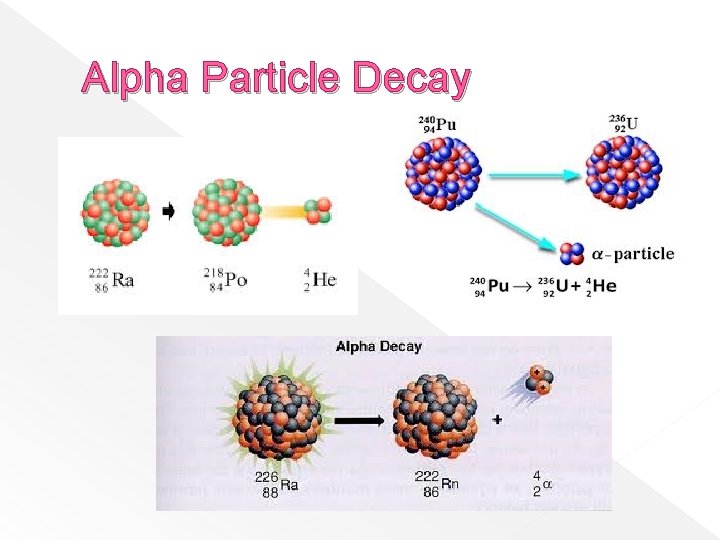
Alpha Particle Decay

Beta Particles � Are fast-moving electrons that are emitted when too many neutrons are present. � Negative particles � Have charge and less mass than alpha higher penetrating power than alpha particles – can be stopped by aluminum foil or wood
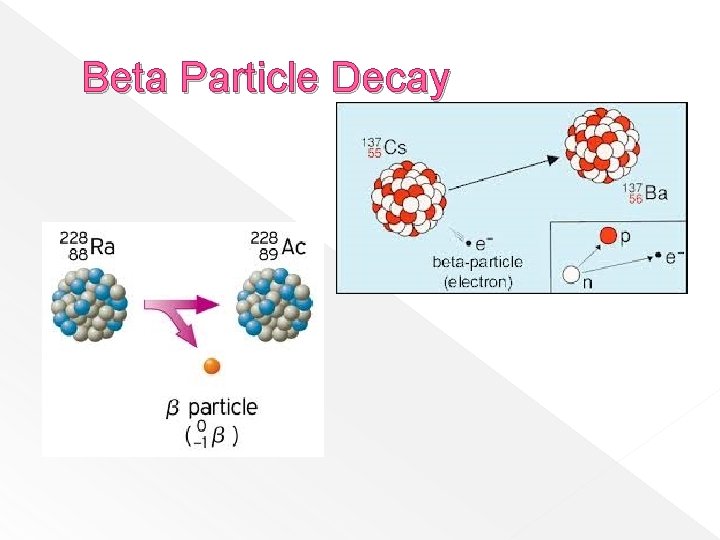
Beta Particle Decay

Gamma Rays � Are photons, which are high-energy electromagnetic radiation. � Have no mass or charge � Emission does not change the A# or M# of a nucleus. � Have high penetrating power - can pass through 1 m of concrete, human body
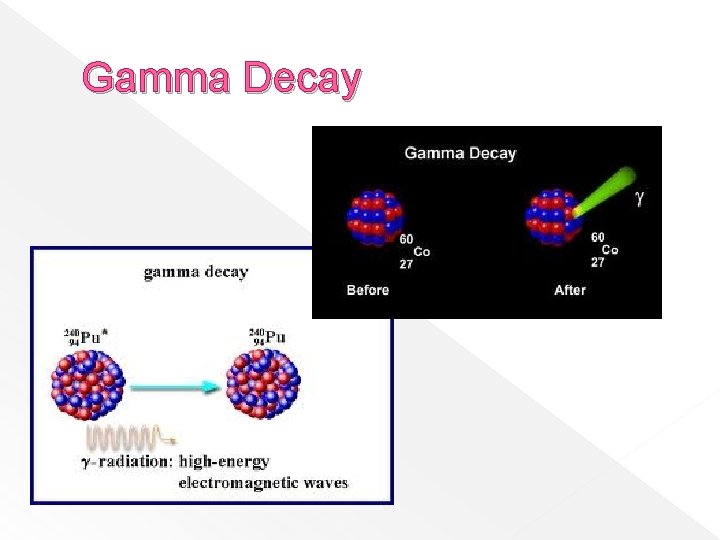
Gamma Decay

Nuclear Radiation Videos � Three types of radiation � https: //www. youtube. com/watch? v=5 o. Uago. F _vi. Q

Part 2: Types of Nuclear Reactions

Nuclear Reactions � Two types of nuclear reactions that release large amounts of energy › Fission › Fusion

Nuclear Fission � Heavy nuclei tend to be unstable. To gain stability, they can fragment into several smaller nuclei. � Nuclear Fission: splitting of nucleus into 2 smaller nuclei

Nuclear Fission Nuclear power plants use nuclear fission to generate power in nuclear reactors. � As shown in the previous picture, each fission releases neutrons, which can generate additional fission reactions. � Chain Reaction: selfsustaining process in which one reaction initiate the next reaction. �

Nuclear Power � Nuclear reactors used controlled fission to produce energy � Energy generated is in the form of heat › The heat is used to generate steam which drives a turbine that in turn generates electricity � Use uranium and plutonium as isotope source � Many advantages and disadvantages
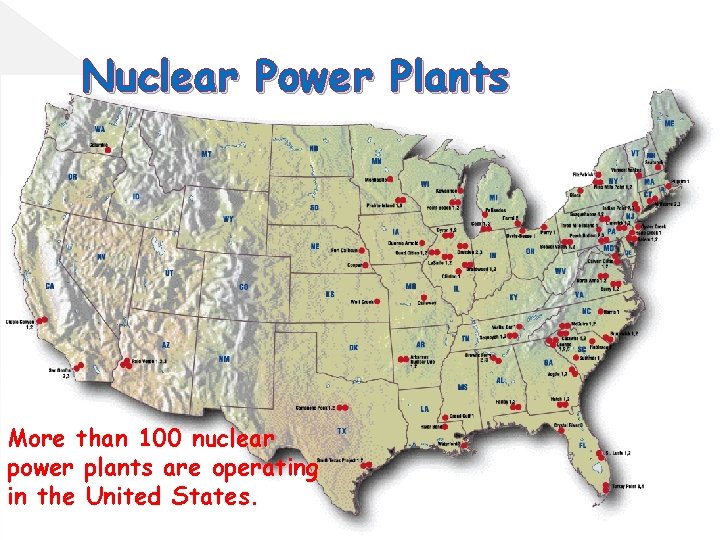
Nuclear Power Plants More than 100 nuclear power plants are operating in the United States.
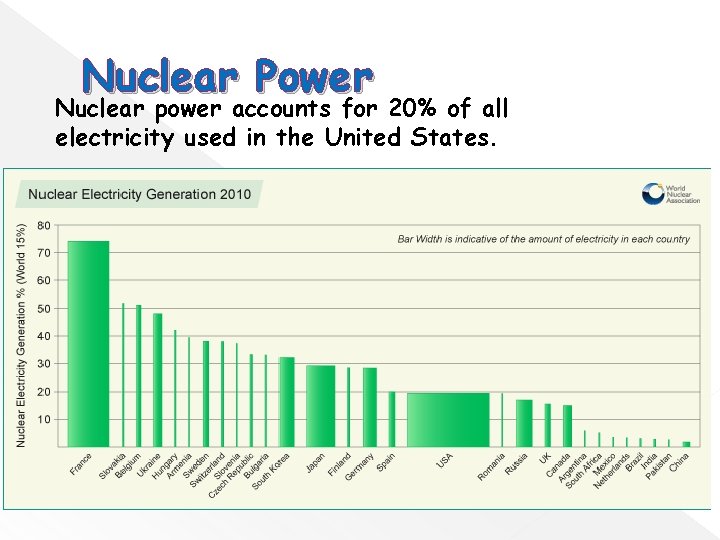
Nuclear Power Nuclear power accounts for 20% of all electricity used in the United States.

Nuclear Power Videos � Nuclear Power � http: //www. pbs. org/video/2220837749/ � New Approach to Nuclear Power on the Way � http: //news. discovery. com/tech/videos/newapproach-to-nuclear-power-on-the-way-video -141001. htm

Nuclear Fission � The number of fissions and energy released can increase rapidly. � The explosion from an atomic bomb is an example of an uncontrolled chain reaction.

Animations of Nuclear Fission
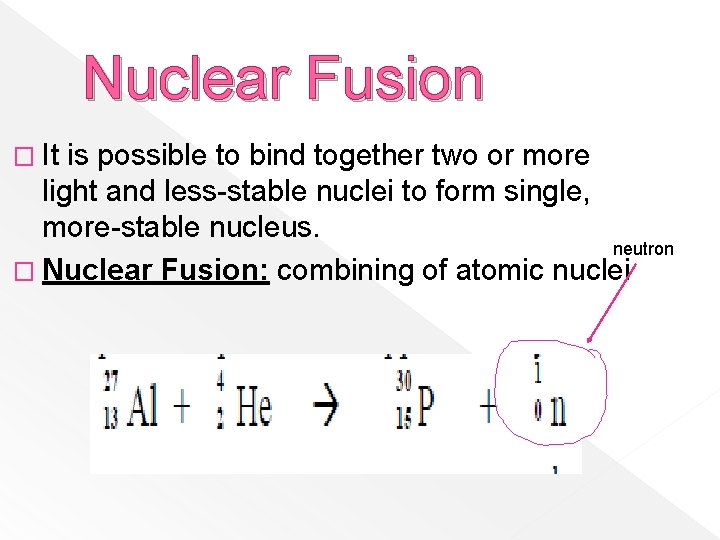
Nuclear Fusion � It is possible to bind together two or more light and less-stable nuclei to form single, more-stable nucleus. neutron � Nuclear Fusion: combining of atomic nuclei

Nuclear Fusion Uses � Fusion reactions release very large amounts of energy. � Fusion reactions occur in the Sun and the stars; hydrogen atoms fuse to form helium atoms. � Fusion requires extremely high energy, which is only attainable at extremely high temperatures

Fusion Videos � Fusion energy advances � http: //www. pbslearningmedia. org/resource/n hsci. tech. fusion/fusion-energy-advances/

Animations of Nuclear Fusion

Fission • • • 235 U is limited danger of meltdown toxic waste No greenhouse gases released produces energy vs. Fusion • • • fuel is abundant no danger of meltdown no toxic waste not yet sustainable produces energy

Fission VS Fusion Videos � Nuclear Energy from fission and fusion � https: //www. youtube. com/watch? v=Lekac. Mu M 12 Y
- Slides: 33