Nuclear Chemistry Chapter 25 Radiation In 1896 Antoine




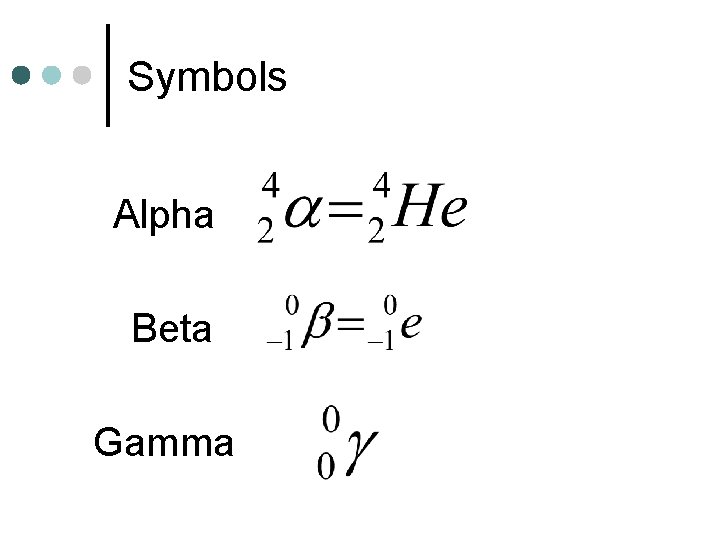














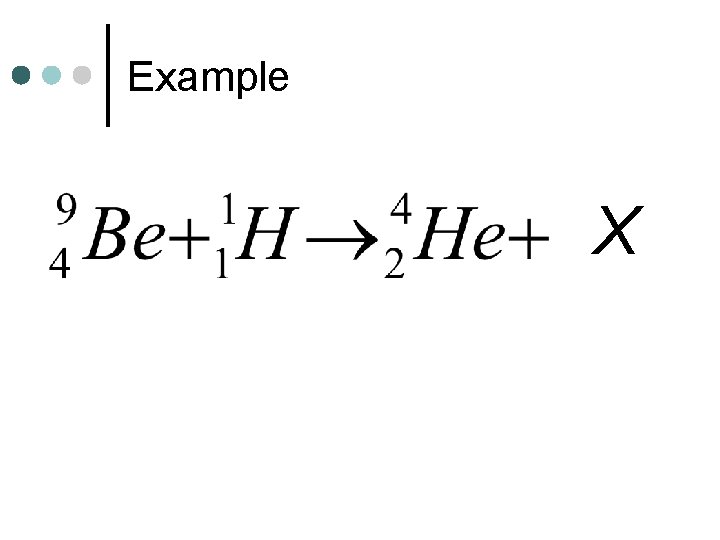
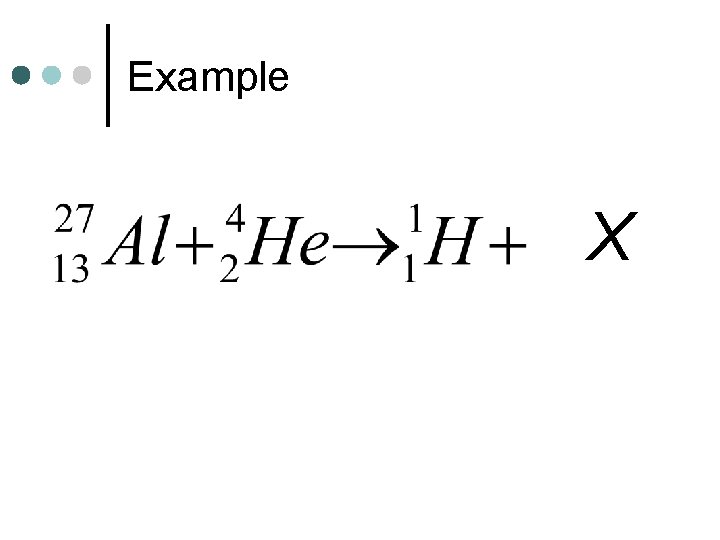



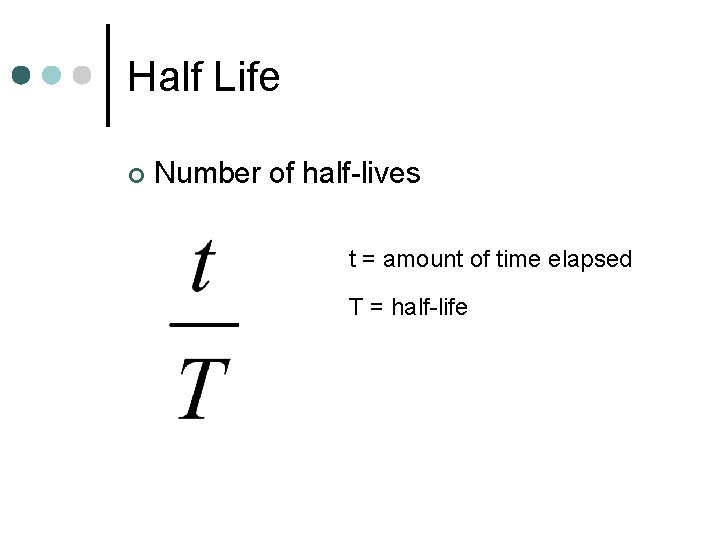
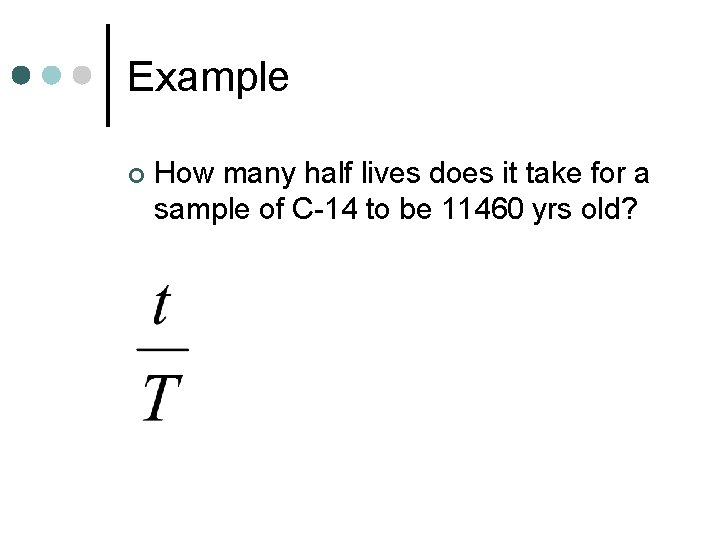
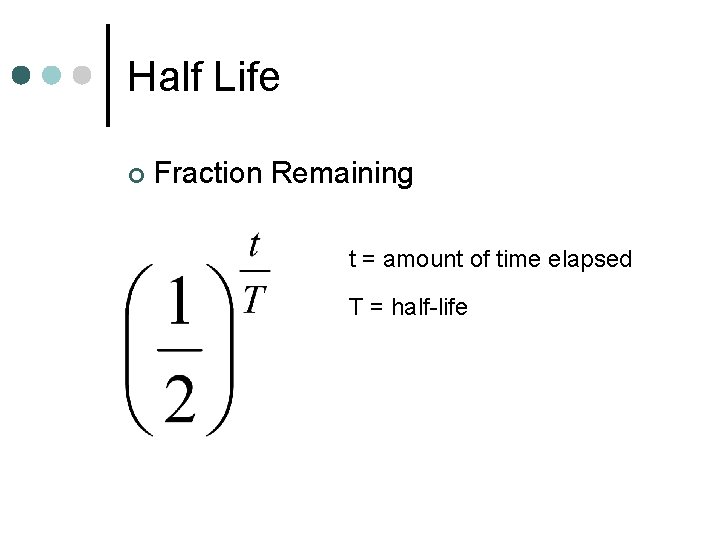


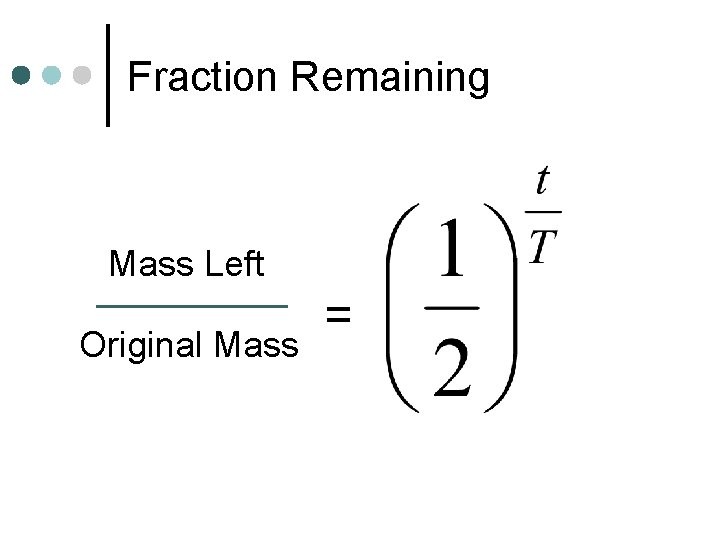
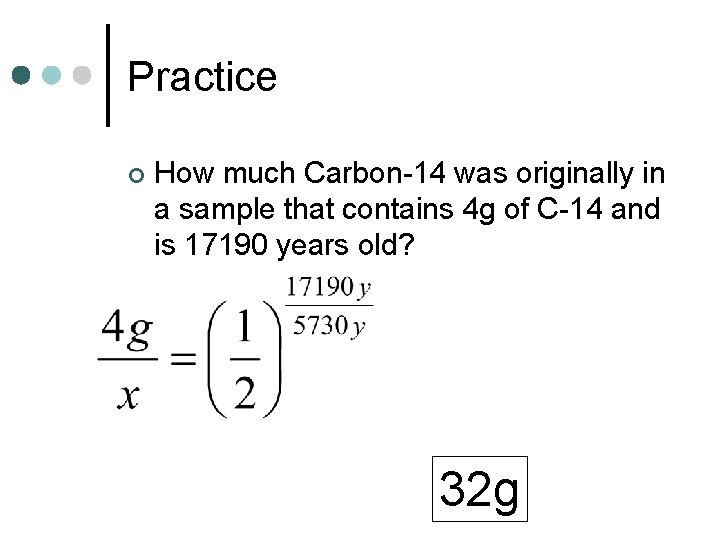

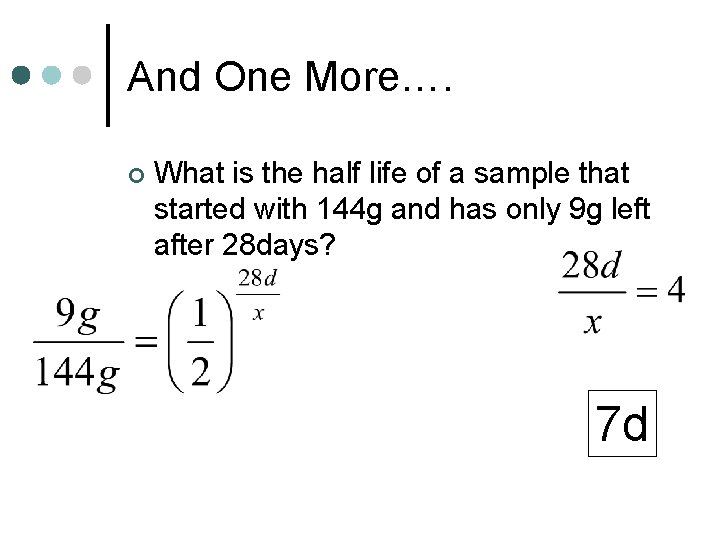
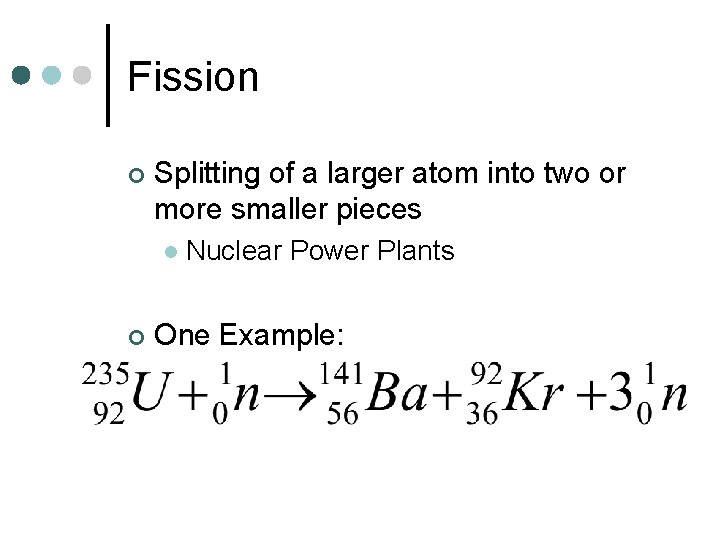




















- Slides: 69

Nuclear Chemistry Chapter 25

Radiation ¢ In 1896, Antoine Henri Becquerel discovered radiation by accident. His associates at the time were Marie and Pierre Curie. l Marie Curie gets credit for naming radioactivity. l

Radioisotopes ¢ Nuclei of unstable isotopes are called radioisotopes. ¢ An unstable nucleus releases energy by emitting radiation during the process of radioactive decay

Radiation ¢ Three Types Alpha l Beta l Gamma l Helium Nucleus Electron Light wave
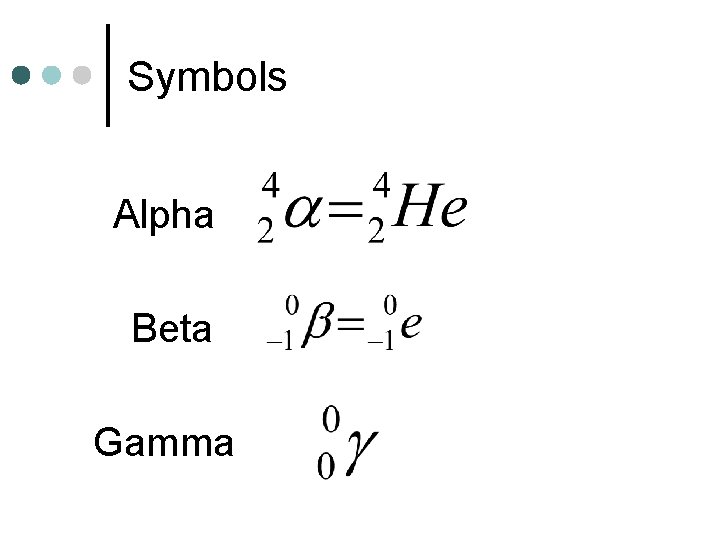
Symbols Alpha Beta Gamma

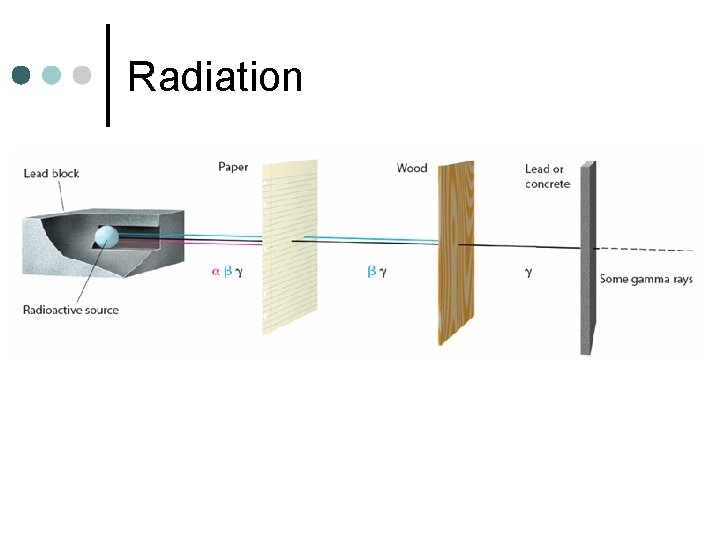
Radiation

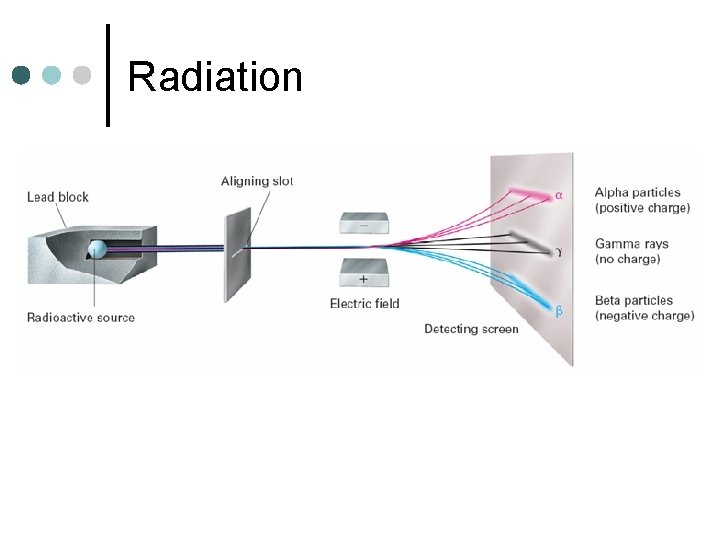
Radiation

Nuclear Symbols ¢ Table O

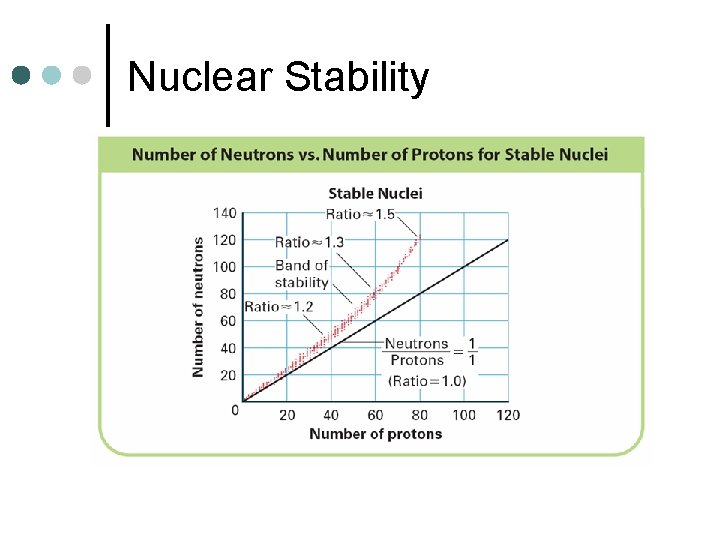
Nuclear Stability ¢ For smaller atoms a ratio of 1: 1 neutrons to protons helps to maintain stability l ¢ C-12, N-14, O-16 For larger atoms, more neutrons than protons are required to maintain stability l Pb-207, Au-198, Ta-181

Nuclear Stability

Radioactive Decay ¢ Radioisotopes will undergo decay reactions to become more stable Alpha Decay ¢ Beta Decay ¢ Positron Emission ¢

Nuclear Reactions ¢ Unstable isotopes of one element are transformed into stable isotopes of a different element. ¢ They are not affected by outside factors, like temp and pressure. They can not be sped up or slowed down.

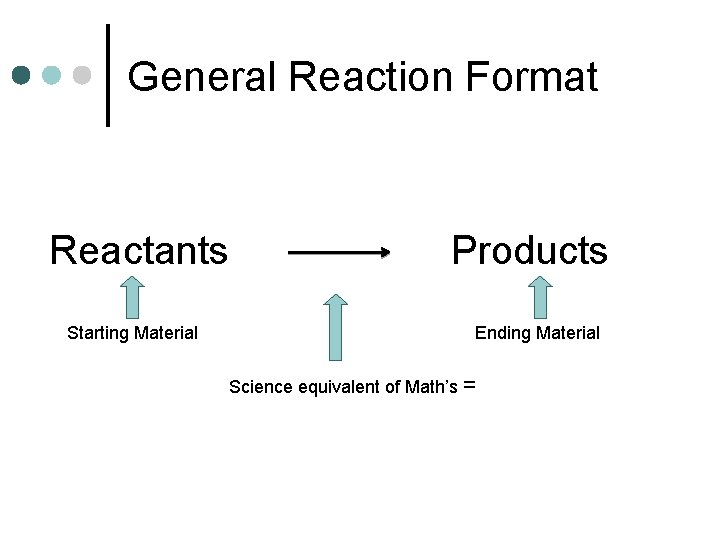
General Reaction Format Reactants Starting Material Products Ending Material Science equivalent of Math’s =

Reaction Format ¢ In Math class you might say: l ¢ A–B=C In Science, we don’t use subtraction A B+C l A breaks into B and C l

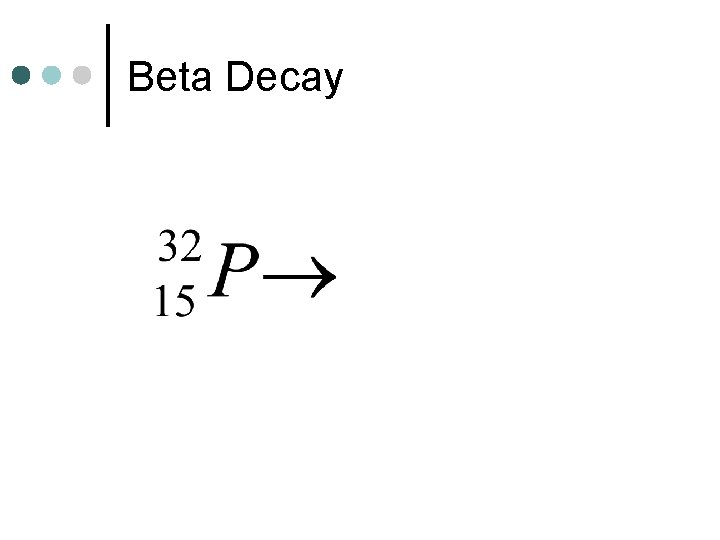
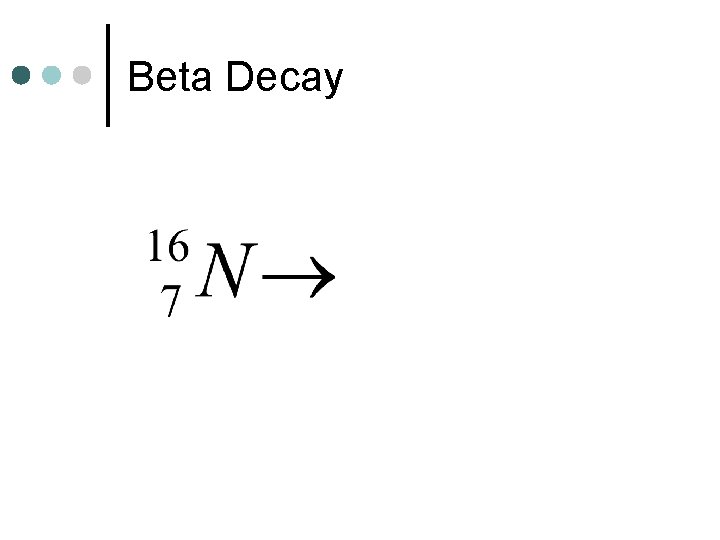
Decay Reactions ¢ Decay reactions involve one unstable nuclei decaying (breaking down) into 2 (or more) smaller nuclei. l Alpha Decay - one of the products is an alpha particle l Beta Decay - one of the products is a beta particle

Nuclear Reactions ¢ ¢ ¢ Reactions must always Balance Mass Numbers have to balance Atomic Numbers have to balance 238 = 4 + 234 92 = 2 + 90

Alpha Decay

Alpha Decay

Alpha Decay

Beta Decay

Beta Decay

Beta Decay

Positron Emission

Transmutations ¢ Any reaction where one element is transformed into a different element ¢ Two main types Natural l Artificial l

Transmutations ¢ Natural Usually has one reactant l Alpha and Beta Decay l ¢ Artificial Usually has more than one reactant l Particle Accelerators l
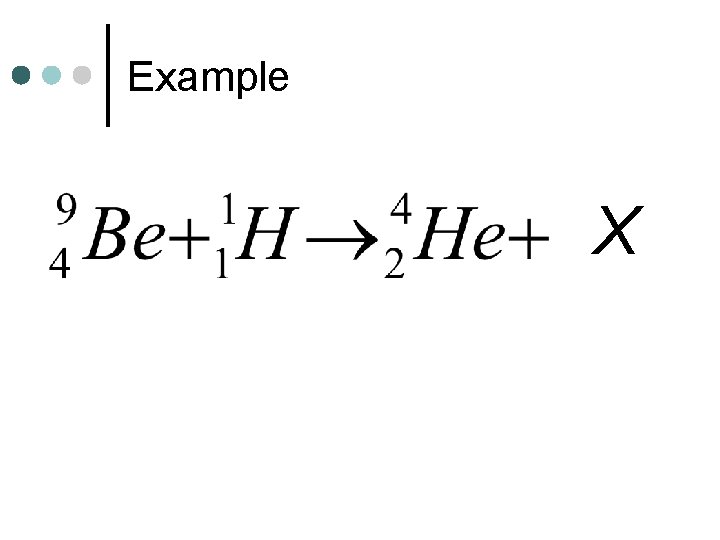
Example X
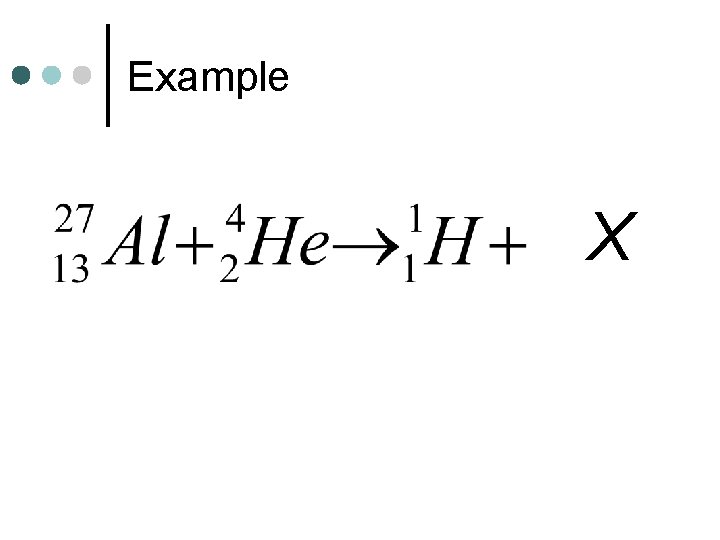
Example X

Example X

Half Life ¢ Amount of time for half of a sample to decay into a new element ¢ Parent Atoms l ¢ Undecayed atoms Daughter Atoms l Decayed atoms

Half Life Number of Halflives Fraction left 0 1 1 1/2 2 1/4 3 1/8 4 1/16 5 1/32
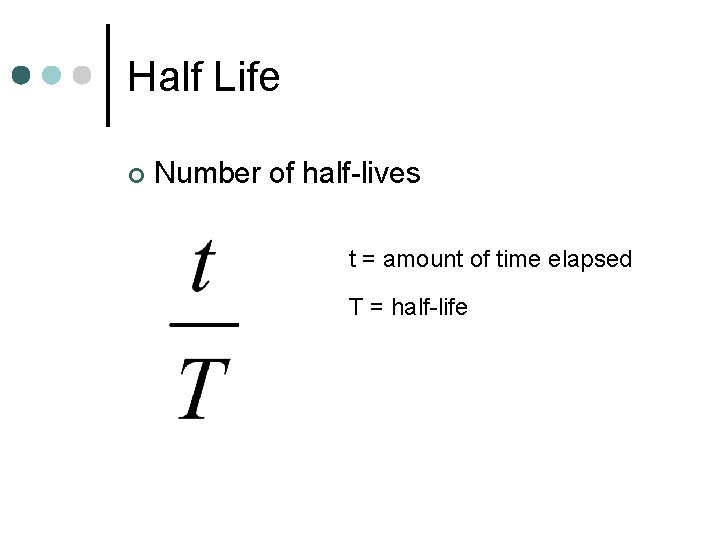
Half Life ¢ Number of half-lives t = amount of time elapsed T = half-life
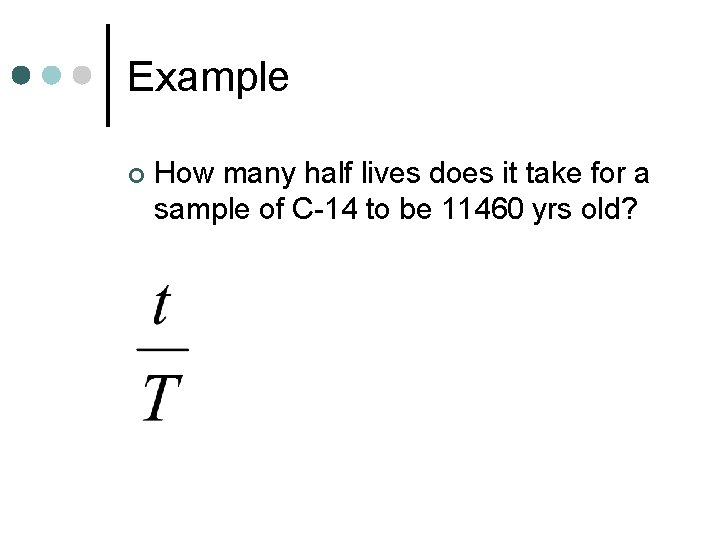
Example ¢ How many half lives does it take for a sample of C-14 to be 11460 yrs old?
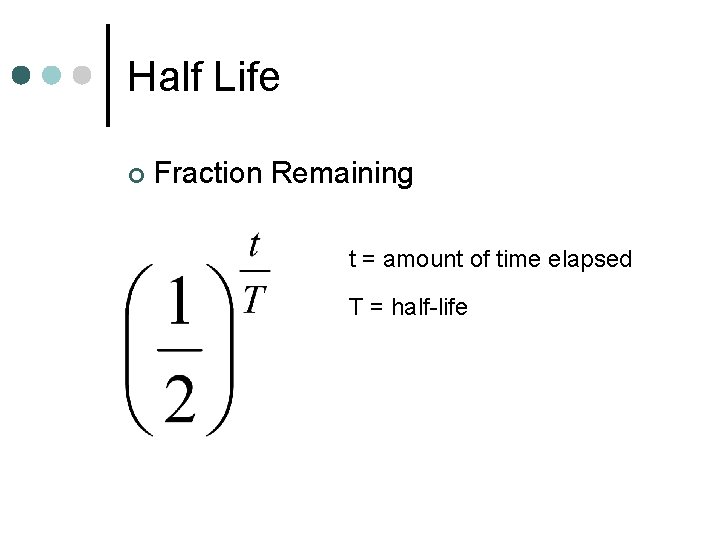
Half Life ¢ Fraction Remaining t = amount of time elapsed T = half-life

Example ¢ What fraction of P-32 is left after 42. 9 days?

Example ¢ How long will a sample of Rn-222 take to decay down to 1/4 of the original sample? 7. 64 d
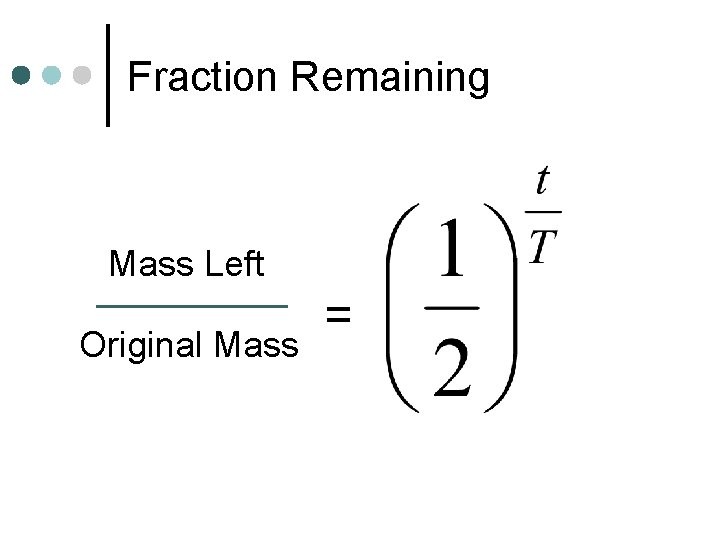
Fraction Remaining Mass Left Original Mass =
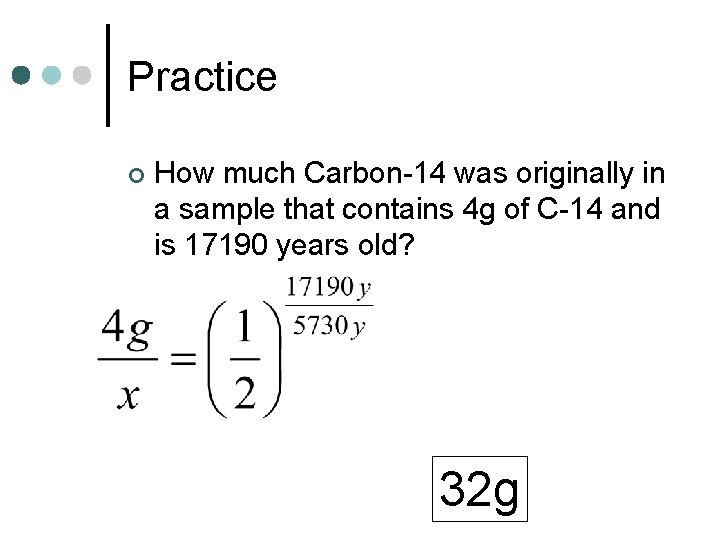
Practice ¢ How much Carbon-14 was originally in a sample that contains 4 g of C-14 and is 17190 years old? 32 g

More Practice ¢ How much 226 Ra will be left in a sample that is 4800 years old, if it initially contained 408 g? 51 g
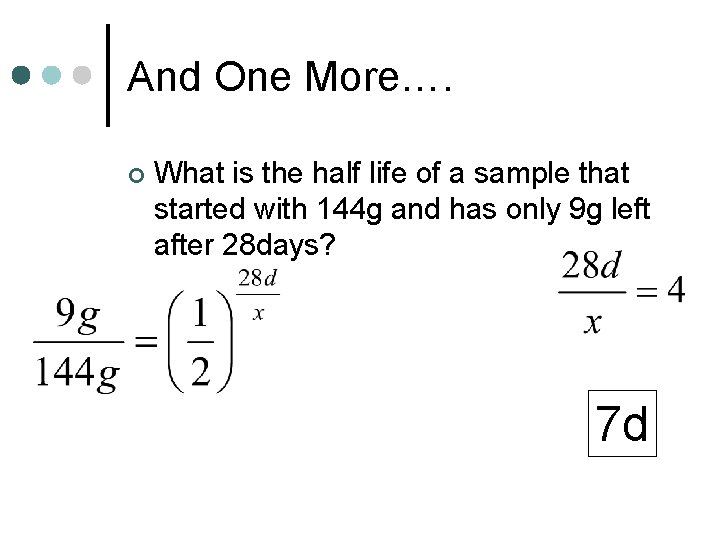
And One More…. ¢ What is the half life of a sample that started with 144 g and has only 9 g left after 28 days? 7 d
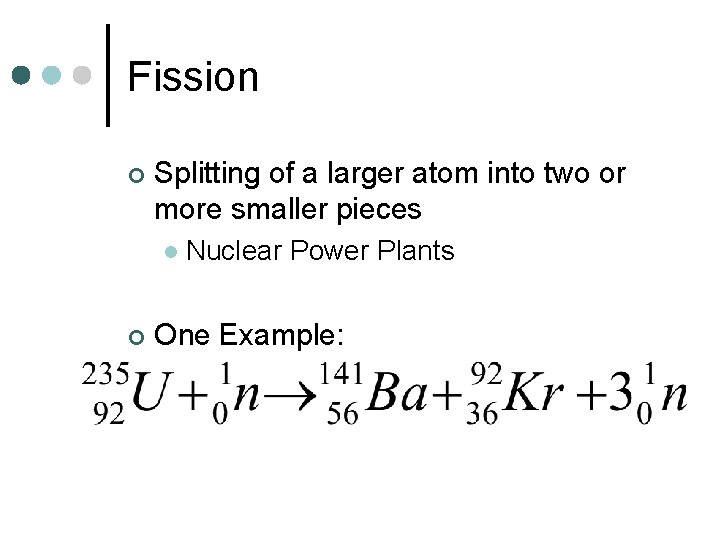
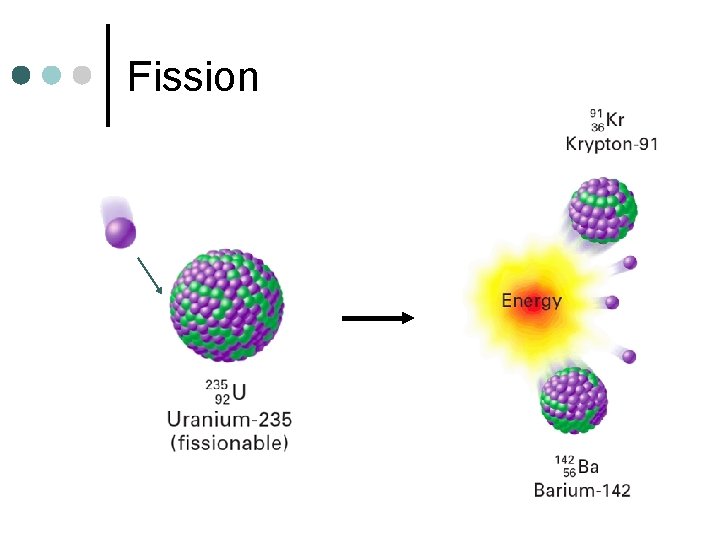
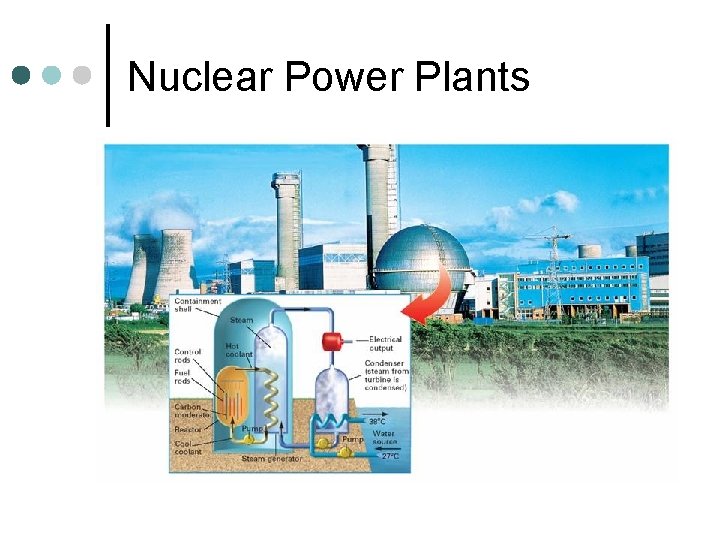
Fission ¢ Splitting of a larger atom into two or more smaller pieces l ¢ Nuclear Power Plants One Example:

Fission

Energy Production ¢ Energy is produced by a small amount of mass being converted to energy ¢ E=mc 2

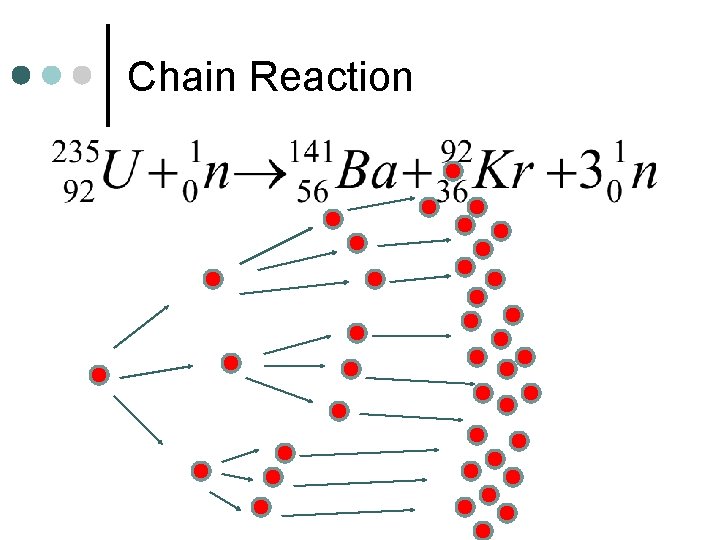
Chain Reaction ¢ Reaction that produces material that can initiate more than one reaction

Chain Reaction

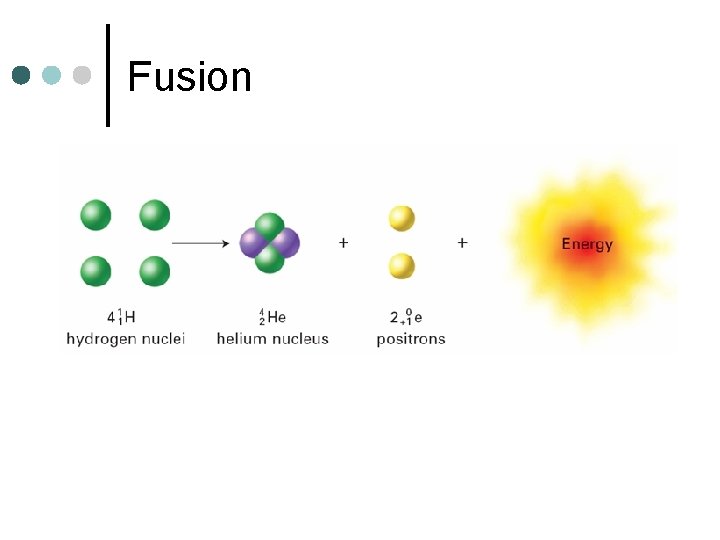
Fusion ¢ Joining of two or more smaller pieces to make a larger piece l ¢ Sun, Stars One Example:

Fusion ¢ More Examples:

Fusion

Energy Production ¢ Energy is produced by a small amount of mass being converted to energy l ¢ More energy is produced by fusion than any other source E=mc 2

Fission vs. Fusion ¢ Advantages of Fission Produces a lot of energy l Can be a controlled reaction l Material is somewhat abundant l

Fission vs. Fusion ¢ Disadvantages of Fission Uses hazardous material l Produces hazardous material l • Long Half Life Reaction can run out of control. l Limited amount of fissionable material l

Fission vs. Fusion ¢ Advantages of Fusion Lighter weight material l Easily available material l Produces waste that is lighter and has shorter half-life l Produces more energy than fission l

Fission vs. Fusion ¢ Disadvantages of Fusion l Must be done at very high temperatures • Only been able to attain 3, 000 K l Have not been able to sustain stable reaction for energy production

Uses of Radioisotopes Smoke Detectors ¢ Food Irradiation ¢ Radioactive Dating ¢ Medical Tracers ¢ Nuclear Power Plants ¢ Nuclear Weapons ¢ Origin of Elements ¢

Smoke Detectors ¢ Americium produces radiation that is monitored by an electrical circuit ¢ Smoke interferes with the current, triggering the alarm

Food Irradiation ¢ Food is exposed to radiation, killing bacteria and mold ¢ Food is cleaner and lasts longer

Radioactive Dating ¢ Ratio of Parent atoms to Daughter atoms provides an age ¢ Examples C-14 used to date organic material l U-238 used to date geological formations l

Medical Tracers Radioisotopes replace stable isotopes ¢ Radiation produced can be detected by machines ¢ Example ¢ I-131 is used for thyroid disorders l Barium milk shakes l Co-60 for Cancer l

Radioisotopes ¢ You must know these radioisotopes and uses l I-131 • Diagnosing and treating thyroid disorders l Co-60 • Treating cancer

Radioisotopes ¢ You must know these radioisotopes and uses l C-14 • Dating living organisms • Compare to C-12 l U-238 • Dating geologic formations • Compare to Pb-206

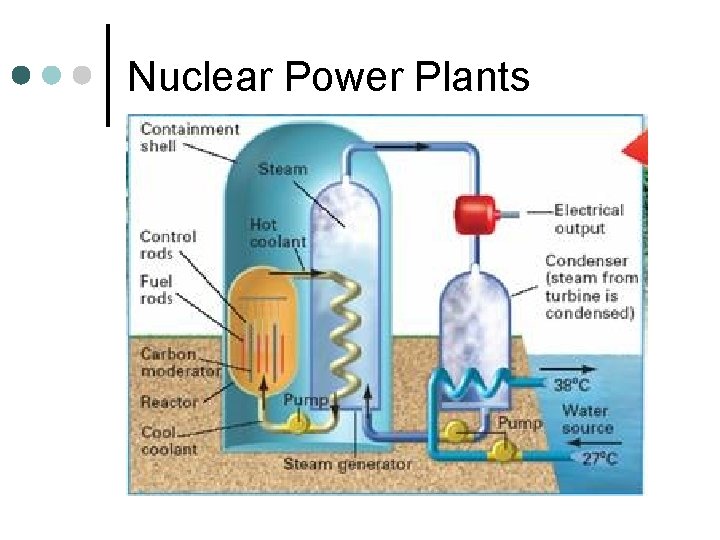
Nuclear Power Plants

Nuclear Power Plants

Nuclear Power Plants

Nuclear Weapons

Video ¢ Origin of Elements ¢ You. Tube

Stability ¢ ¢ Elements 1 -26 are made in the core of stars Elements 27 -92, excluding 43 and 61, are made during a Supernova explosion No element larger than 83 has a stable isotope No element larger than 92 is made in nature