Nuclear Chemistry Chapter 21 Objectives Define radioactivity Explain

Nuclear Chemistry Chapter 21

Objectives • Define radioactivity. • Explain trends in the relative stability of nuclei. • Complete half-life calculations. • Write, balance, and interpret nuclear reactions. • Identify types of radioactive decay. • Explain how fusion and fission occur. • Discuss applications of nuclear chemistry in everyday life. • Outline the potential hazards of nuclear energy.

Chemical Reactions vs Nuclear Reactions
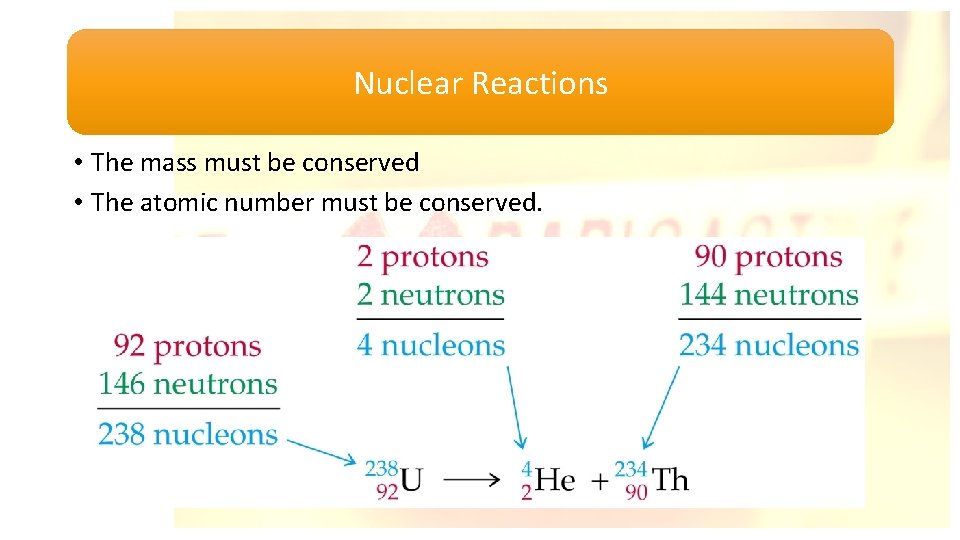
Nuclear Reactions • The mass must be conserved • The atomic number must be conserved.
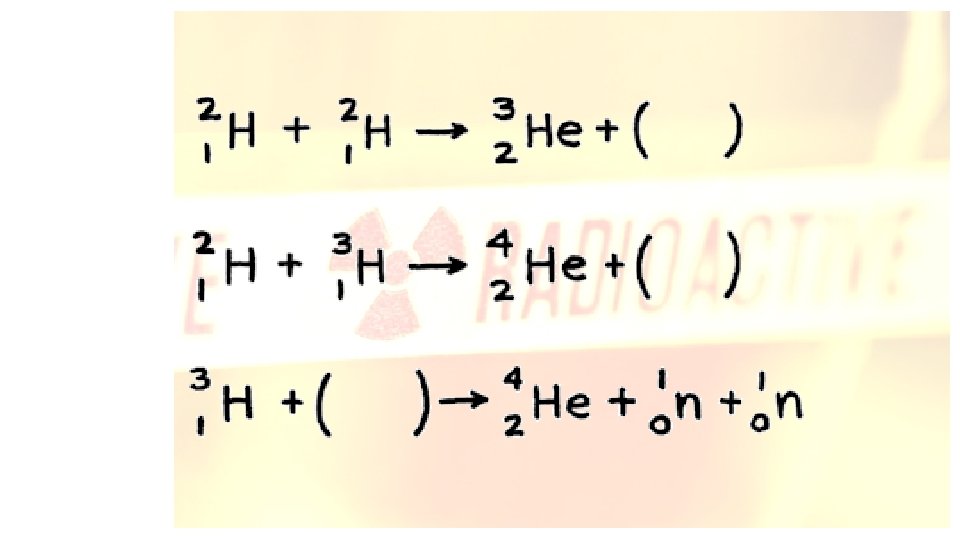

Nuclear Stability • Certain numbers of neutrons and protons are extra stable − n or p = 2, 8, 20, 50, 82 and 126 − Like extra stable numbers of electrons in noble gases (e- = 2, 10, 18, 36, 54 and 86) • Nuclei with even numbers of both protons and neutrons are more stable than those with odd numbers of neutrons and protons • All isotopes of the elements with atomic numbers higher than 83 are radioactive • All isotopes of Tc and Pm are radioactive
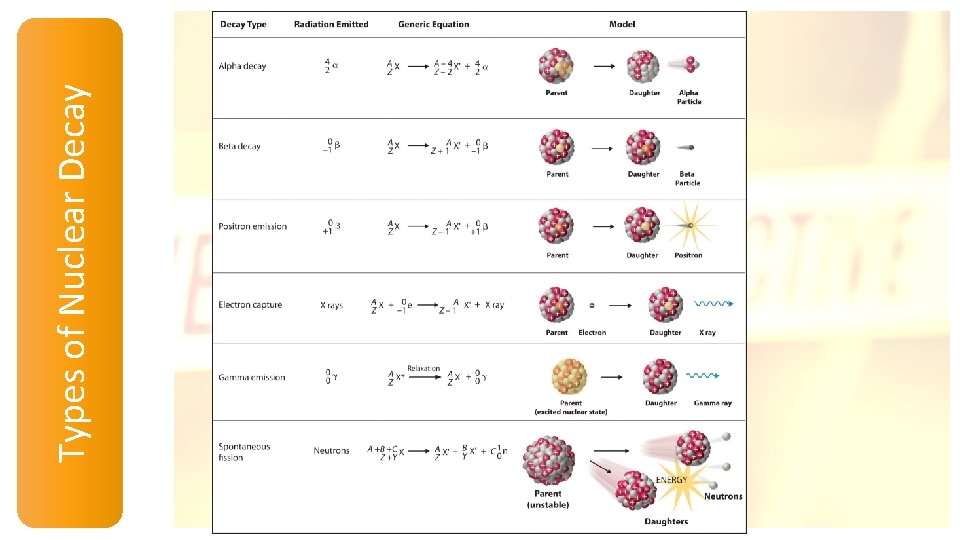
Types of Nuclear Decay
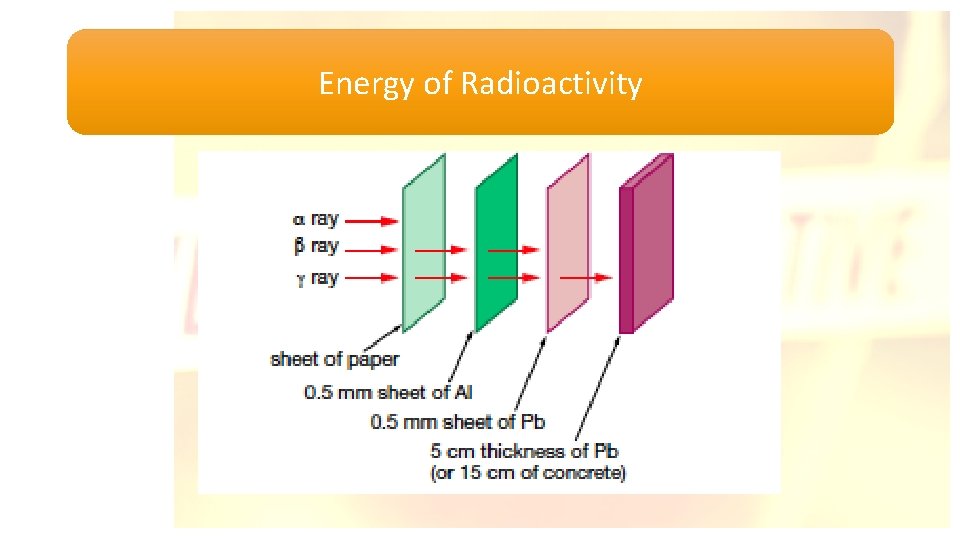
Energy of Radioactivity

Nuclear Stability and Radioactive Decay Beta decay 14 C 6 14 N 7 40 K 19 +-10 b 40 Ca 20 Decrease # of neutrons by 1 + -10 b 1 n 0 Increase # of protons by 1 1 p 1 +-10 b Positron decay 11 C 6 38 K 19 11 B + 0 b 5 +1 38 Ar + 0 b 18 +1 1 p 1 Increase # of neutrons by 1 Decrease # of protons by 1 1 n 0 ++10 b

Nuclear Stability and Radioactive Decay Electron capture decay 37 Ar + 0 e 18 -1 55 0 26 Fe +-1 e 37 Cl 17 55 Mn 25 1 p 1 Increase number of neutrons by 1 Decrease number of protons by 1 + -10 e 1 n 0 Alpha decay 212 Po 84 4 He 2 + 208 Pb 82 Spontaneous fission 252 Cf 98 1 n 2125 In + 2 49 0 Decrease number of neutrons by 2 Decrease number of protons by 2


Complete the following nuclear reaction equations.

Complete the following nuclear reaction equations. • Alpha decay for Thorium-232 • Plutonium-244 undergoes beta decay • Radon-222 decays into Astatine-210 • Francium-223 emits a gamma particle

What Are Your Questions?

What is Half-Life? • The amount of time it takes for half a given amount of material to radioactively decay into something else.

Initial Amount After 1 Half-Life After 2 Half-Lives After 3 Half-Lives

Based solely on the following half-lives, which radioactive atom listed is the most radioactive? A. C-14 (5730 yrs) B. Ra-226 (1620 yrs) C. U-238 (4. 5 million yrs) D. Po-214 (1. 6 ms) E. P-32 (14 days)

Radioactive Dating Radiocarbon Dating 14 N 7 + 01 n 14 C 6 14 N 7 + 11 H + -10 b + n t½ = 5730 years Uranium-238 Dating 238 U 92 206 Pb 82 + 8 24 a + 6 -10 b t½ = 4. 51 x 109 years t½ = 0. 693 l
A 4. 5 gram sample of radon decomposes over 3 half lives. How much of the sample remains?
The half-life of C-14 is 5730 years. How long will it take for 50% of 25 g of C-14 to decompose? How long will it take for 75% to decompose?
An artifact contains 12. 5% of the original amount of C-14. How old is this sample? (C-14 half-life is 5730 years. )
The half-life of the beta particle emitter tritium, 3 H, is 12 years. How much of a 1. 00 g sample of 3 H remains after 48 years?

What Are Your Questions?
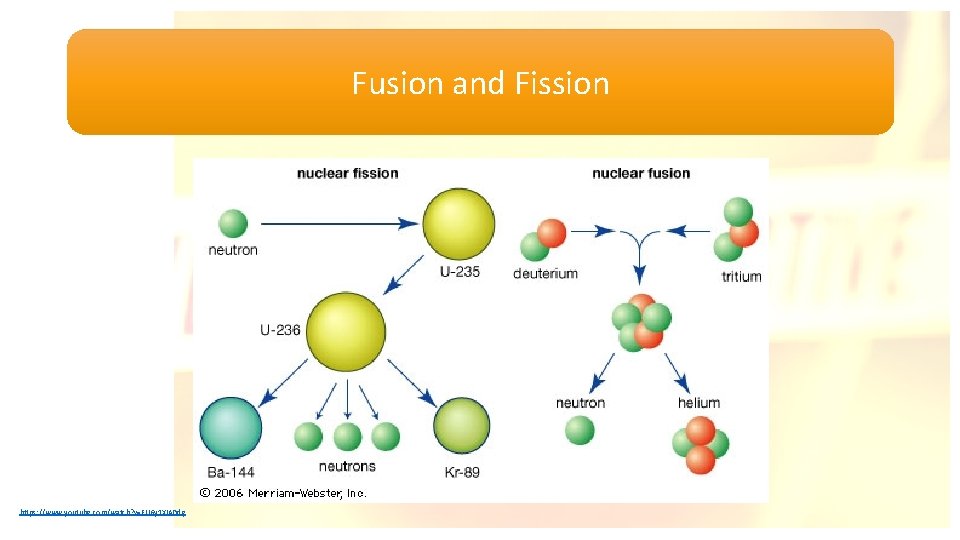
Fusion and Fission https: //www. youtube. com/watch? v=FU 6 y 1 XIADdg
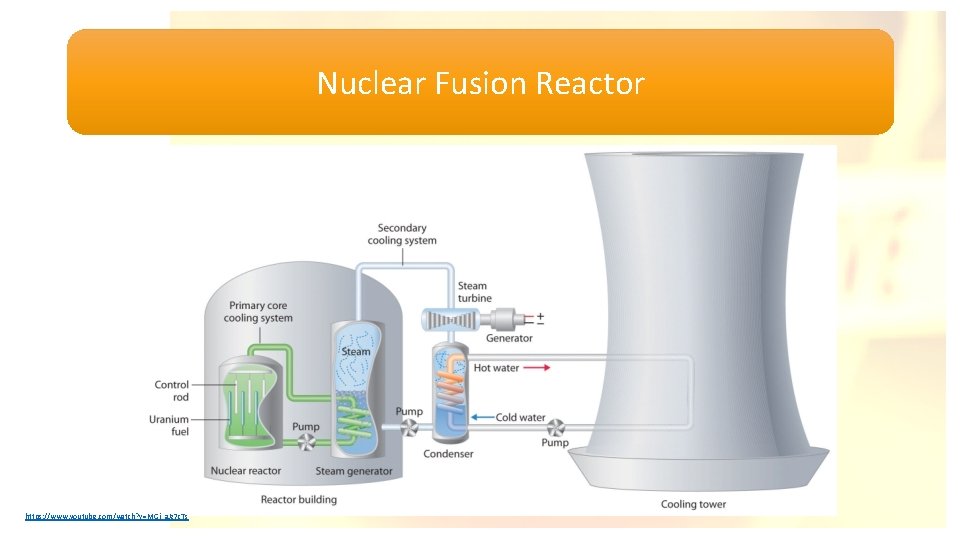
Nuclear Fusion Reactor https: //www. youtube. com/watch? v=MGj_a. Jz 7 c. Ts
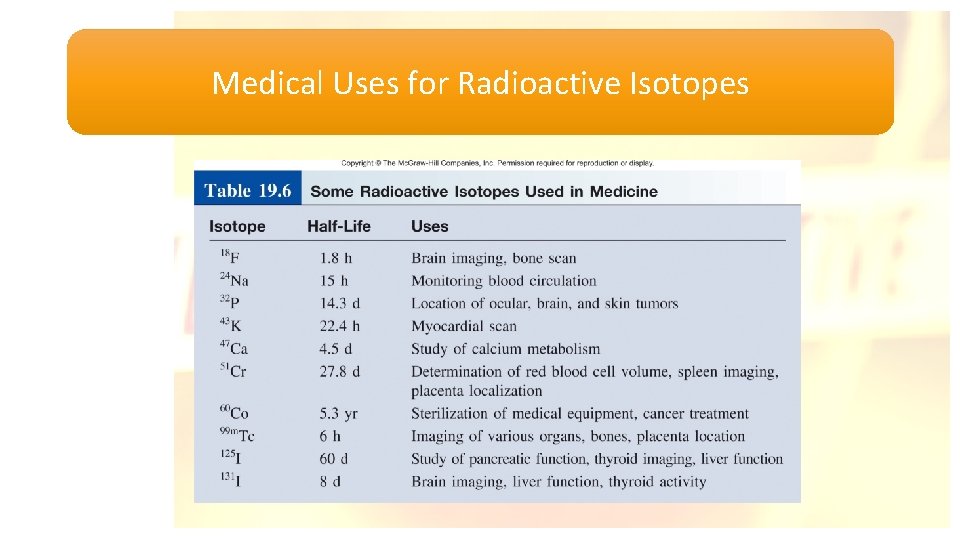
Medical Uses for Radioactive Isotopes

Nuclear Weapons • Use nuclear reactions to create an intense explosion. • The Manhattan Project • Project the ran from 1942 -46 to develop atomic bombs.

Nuclear Disasters • Three Mile Island (1979) • Partial Meltdown in Pennsylvania • Chernobyl (1986) • Reactor 4 meltdown in Ukraine • Time 3: 06 • Fukushima (2011) • Three of six nuclear reactors melted down in Japan as a result of a tsunami

What Are Your Questions?
- Slides: 30