Nuclear Chemistry Bravo 15 000 kilotons Standard SPS

Nuclear Chemistry Bravo – 15, 000 kilotons

Standard SPS 3. Students will distinguish the characteristics and components of radioactivity. a. Differentiate among alpha and beta particles and gamma radiation. b. Differentiate between fission and fusion. c. Explain the process half-life as related to radioactive decay. d. Describe nuclear energy, its practical application as an alternative energy source, and its potential problems.

Essential Questions What makes an element radioactive? What are the different types of radioactive decay? How are they similar/different?

Learning Targets • You should know what makes an • element radioactive. You should know the different types of radioactive decay and be able to compare/contrast them.

What do we mean by Radioactivity? Radioactive decay is the process in which an unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. Elements with atomic numbers greater than 83 are all radioactive. A nucleus is unstable if the ratio of protons and neutrons is unbalanced. There are numerous types of radioactive decay. KEY IDEA: An unstable nucleus releases energy to become more stable. Science Park HS -- Honors Chemistry

Nuclear Symbols Mass number (p+ + no) Atomic number (number of p+) Element symbol

Types of Radioactive Decay v alpha production (a, He): helium nucleus v beta production (b, e): v gamma ray production (g):

Nuclear Radiation • Some nuclei are unstable. • They can change into isotopes or different elements. • Types of nuclear radiation: alpha particles, betaparticles, gamma rays
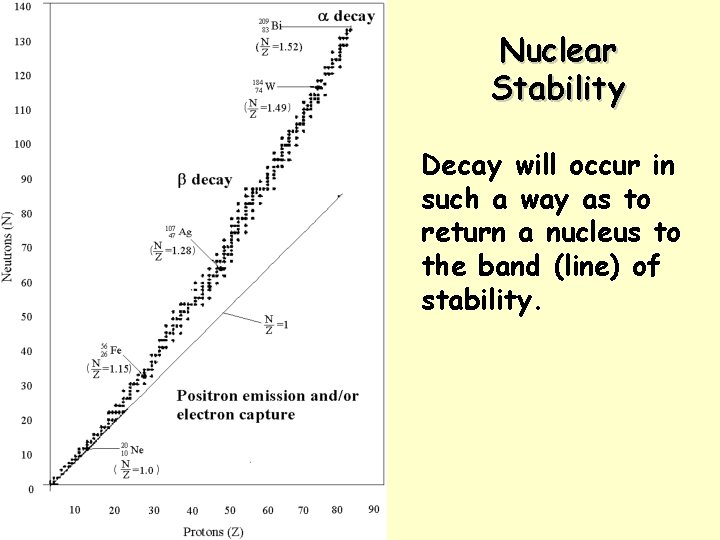
Nuclear Stability Decay will occur in such a way as to return a nucleus to the band (line) of stability.
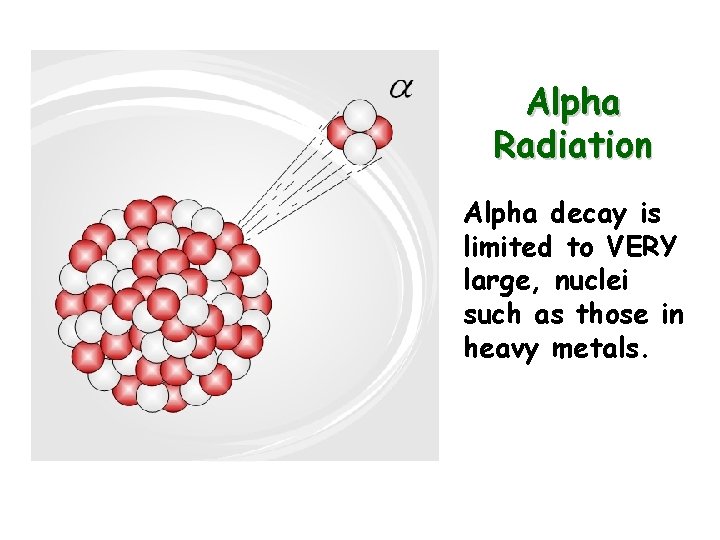
Alpha Radiation Alpha decay is limited to VERY large, nuclei such as those in heavy metals.

Alpha Particles (a) Alpha particles are made of a helium nucleus and contain 2 protons and 2 neutrons. (+)positively charged Massive, weigh 4 a. m. u. Stopped by paper or skin In alpha decay the atomic mass decreases by 4 and the atomic number decreases by 2 The products should add up to the reactants

Beta Radiation Beta decay converts a neutron into a proton.

Beta Particles (ß) Beta particles are formed from break down of a nucleus into 1 eand 1 proton. The beta particle is a fast moving electron. (-) negatively charged, insiginificant mass. The e- is thrown from nucleus. Faster than alpha particles Stopped by a thin sheet of metal Because a neutron is changed into a proton, the atomic number goes up by 1, but since they both weigh the same the atomic mass stays the same.

Gamma Rays/Radiation(ß) Part of the electromagnetic spectrum It is an emission of energy, NOT a particle like alpha and beta No charge, no mass Fastest, most penetrative, most energy of all types of decay Stopped by lead or thick concrete Most dangerous The nucleus is unaffected by gamma radiation so the atomic mass and number stay the same

Three Common Types of Radioactive Emissions Penetrability Alpha particles may be completely stopped by a sheet of paper, beta particles by aluminum shielding. Gamma rays, however, can only be reduced by much more substantial obstacles, such as a very thick piece of lead. Science Park HS -- Honors Chemistry
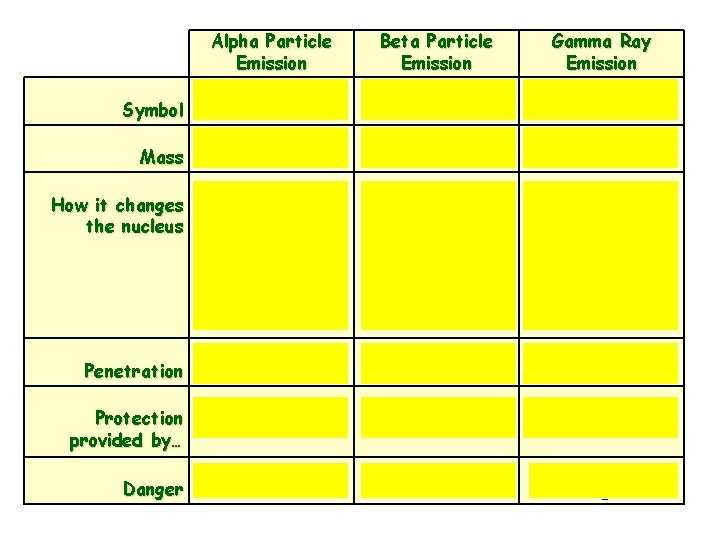
Symbol Mass How it changes the nucleus Alpha Particle Emission Beta Particle Emission or or Heavy Light Decreases the mass number by 4 Decreases the atomic number by 2 Converts a neutron into a proton Increases atomic number by 1 Gamma Ray Emission No Mass No change to the nucleus Penetration Low Medium High Protection provided by… Skin Paper, clothing Lead Danger Low Medium High

CA Standards Students know protons and neutrons in the nucleus are held together by nuclear forces that overcome the electromagnetic repulsion between the protons. Students know the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions. The change in mass (calculated by E = mc 2) is small but significant in nuclear reactions.

Fission - Splitting a heavy nucleus into two nuclei with smaller mass numbers.
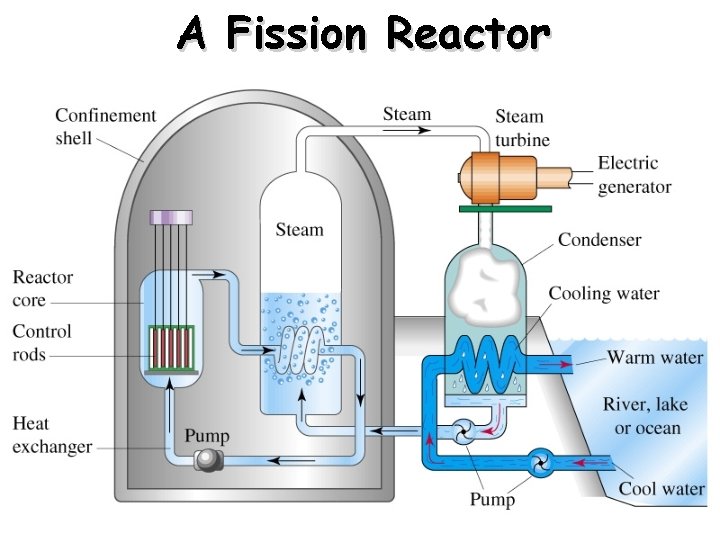
A Fission Reactor

Deuterium – Tritium Fusion Reaction Fusion - Combining two light nuclei to form a heavier, more stable nucleus. The sun makes its energy by fusion.
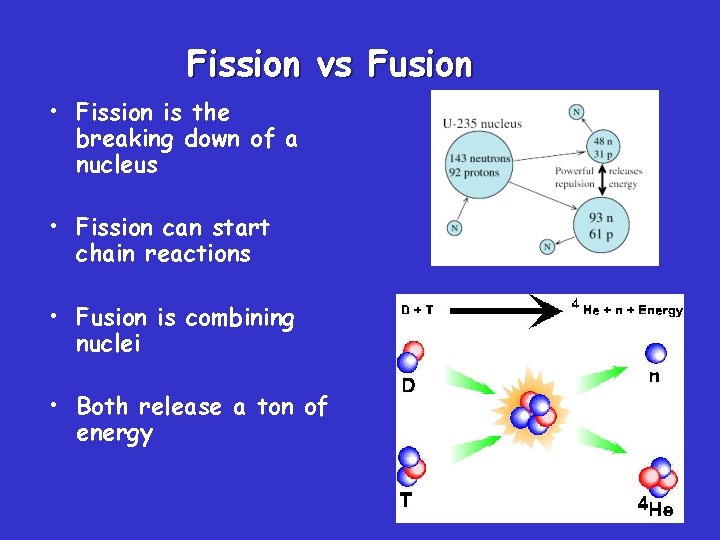
Fission vs Fusion • Fission is the breaking down of a nucleus • Fission can start chain reactions • Fusion is combining nuclei • Both release a ton of energy

Radiation in our world Helpful radiation Smoke alarms Treat cancer Tracers in medicine Tracers in geology Nuclear power-fission Harmful Radiation • Can be naturalsoil, sun, water, plants • Our sun emits solar radiation • Can actually change DNA and cause birth defects
- Slides: 22