November 14 2013 Once youre in youre in

November 14, 2013 Once you’re in, you’re in. When the bell rings, electronics and headphones away working on the DO NOW. HAVE OUT ON YOUR DESK YOUR VOCAB HW. TURN IN HW #10. TURN IN ANY LATE STEM PROJECT ASSIGNMENTS AND ANY LATE HW. DO NOW: In the entrance way to the classroom your phase mini-posters are up. On loose-leaf paper record the information from these posters – 4 bullets of info for all 3 phases (Solid, liquid, gas). You have 12 minutes to get this done.

Tutoring • Thursday afterschool in 710 (2: 30 -3: 30 pm)

Homework 11 -14 -13 • Vocabulary column #2 ** • HW assignment #11 **graded

Week of 11 -11 -13 11 -11: Reaction Stoichiometry Review HW: Anticipation Guide 11 -12: Solutions HW: Vocabulary column #1 , assignment #10 11 -13: Solutions HW: Vocabulary colonm #2, assignment #11 11 -14: Reaction Stoich test, Solutions Lab HW: Post-Lab work; study for solutions quiz Monday 11 -17

Objectives • SWBAT • • Infer the quantitative nature of a solution (molarity and dilution) Summarize properties of solutions Interpret Solutions diagrams Explain the solution process
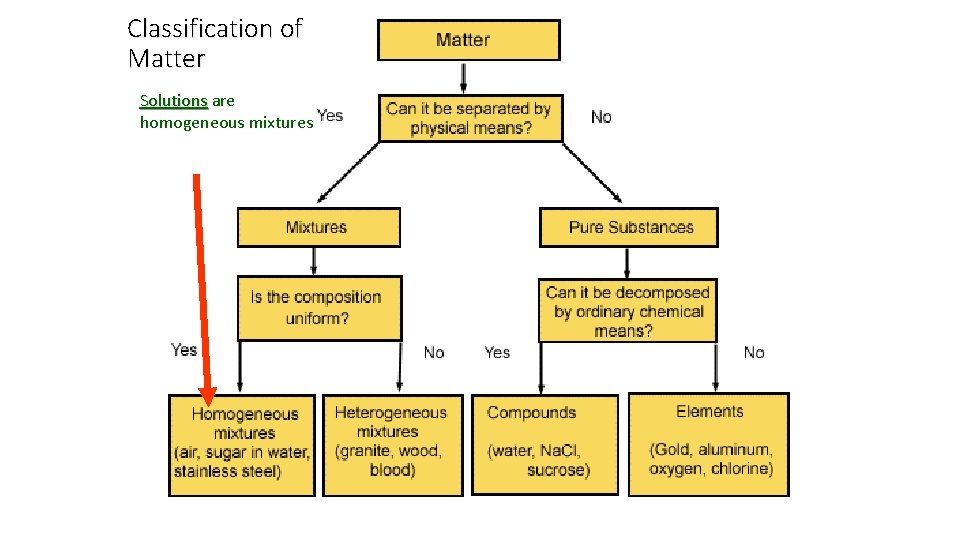
Classification of Matter Solutions are Solutions homogeneous mixtures

Solute A solute is the dissolved substance in a solution. Salt in salt water Sugar in soda drinks Carbon dioxide in soda drinks Solvent A solvent is the dissolving medium in a solution. Water in salt water Water in soda

“Like Dissolves Like” Nonpolar solutes dissolve best in nonpolar solvents Fats Steroids Benzene Hexane Waxes Toluene Polar and ionic solutes dissolve best in polar solvents Inorganic Salts Sugars Water Small alcohols Acetic acid

Solubility Trends § The solubility of MOST solids increases with temperature. § The rate at which solids dissolve increases with increasing surface area of the solid. § The solubility of gases decreases with increases in temperature. § The solubility of gases increases with the pressure above the solution.

Therefore… Solids tend to dissolve best when: o Heated o Stirred o Ground into small particles Gases tend to dissolve best when: o The solution is cold o Pressure is high
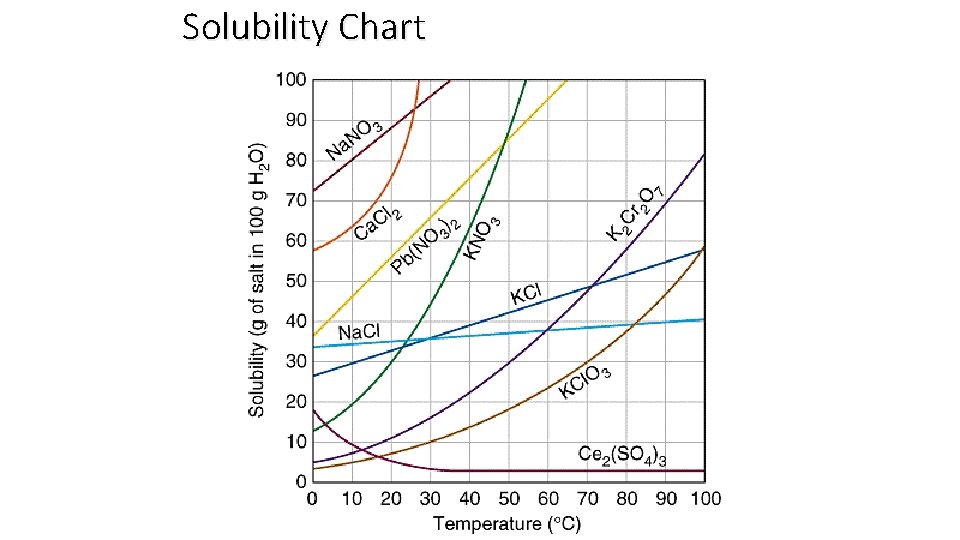
Solubility Chart

Definition of Electrolytes and Nonelectrolytes An electrolyte is: A substance whose aqueous solution conducts an electric current. A nonelectrolyte is: A substance whose aqueous solution does not conduct an electric current.
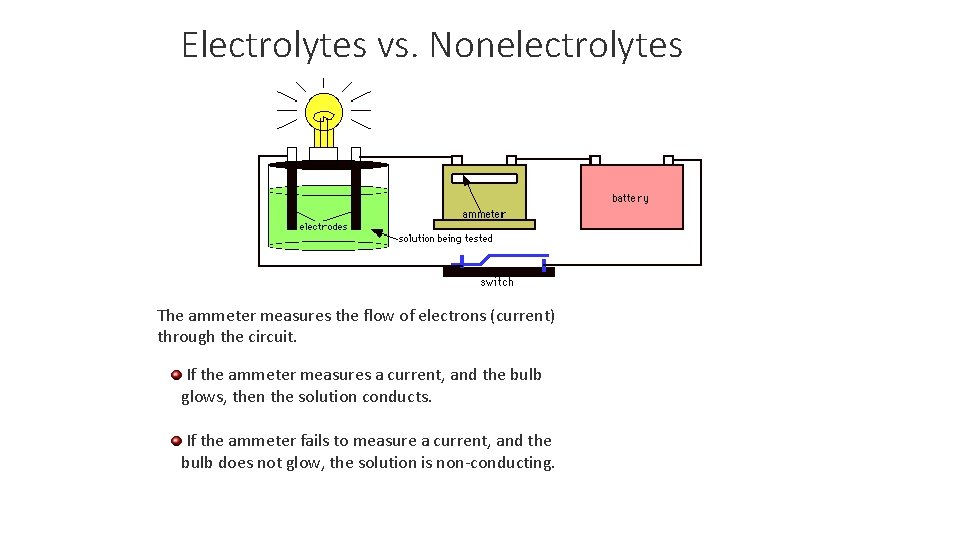
Electrolytes vs. Nonelectrolytes The ammeter measures the flow of electrons (current) through the circuit. If the ammeter measures a current, and the bulb glows, then the solution conducts. If the ammeter fails to measure a current, and the bulb does not glow, the solution is non-conducting.

Try to classify the following substances as electrolytes or nonelectrolytes… 1. 2. 3. 4. 5. 6. 7. 8. Pure water Tap water Sugar solution Sodium chloride solution Hydrochloric acid solution Lactic acid solution Ethyl alcohol solution Pure sodium chloride

Answers to Electrolytes ELECTROLYTES: NONELECTROLYTES: Tap water (weak) Pure water Na. Cl solution Sugar solution HCl solution Ethanol solution Lactate solution (weak) Pure Na. Cl

Foldable: Solution = Solute + Solvent • Solute - gets dissolved • Solvent - does the dissolving • • Aqueous (water) Tincture (alcohol) Amalgam (mercury) Organic • Polar • Non-polar Nightmare on White Street Chem Matters, December 1996

Calculations of Solution Concentration: Molarity is the ratio of moles of solute to Molarity liters of solution

Diluting Solutions • When you add solvent, you increase the number of solvent particles among which the solute particles move Moles of solute in stock solution = moles of solute after dilution M 1 V 1 = M 2 V 2

Mr. Causey Video Guide http: //www. youtube. com/watch? v=F 2 BRp. Fih. X 3 M

Colligative Properties of Solutions Colligative properties are properties that depend only on the number of solute particles in solution and not on the nature of the solute particles. • Vapor Pressure Lowering • Boiling Point Elevation • Freezing Point Depression • Osmotic Pressure

FOLDABLE 4 squares Colligative Properties • Vapor Pressure Lowering is due to the number of solute particles in solution • The greater the number of solute particles in a solvent, the lower the resulting vapor pressure. • Boiling Point Elevation • Temperature difference between as solution’s boiling point and a pure solvent’s boiling point • The greater the number of solute particles in the solution, the greater the boiling point elevation.

Colligative Properties • Freezing Point Depression • The difference in temperature between its freezing point and the freezing point of its pure solvent. • The freezing point of a solution is always lower than that of a pure solvent. • Depends on the number of solute particles in a given volume of solution. • Osmotic Pressure • Amount of additional pressure caused by the water molecules that moved into the solution. • Depends on the number of solute particles in a given volume of solution.

Complete the following using your notes and your book by Monday 11 -18 • Solubility Curve Worksheet • Stoich using molarity worksheet (you will receive tomorrow) • Net ionic equation handout

Exit Ticket 1. What volume, in milliliters of 2. 00 M calcium chloride (Ca. CL 2) stock solution would you use to make. 50 L of. 300 M calcium chloride solution? 2. A 100. 5 m. L intravenous solution contains 5. 10 g of glucose (C 6 H 12 O 6). What is the molarity of the solution? Molar mass of glucose is 180 g.
- Slides: 24