Novel Stent Design Bio Mimicry in DES Sigmund

Novel Stent Design & Bio. Mimicry in DES Sigmund Silber, MD, Ph. D FESC, FACC, FAHA Professor of Medicine Heart Center at the Isar Munich, Germany

Disclosures �None Disclaimer – All the technologies, trade marks presented herein are property of Meril Life Sciences Pvt. Ltd. Copying and/or distributing in any form without written permission from Meril Life Sciences Pvt. Ltd. is strictly prohibited.
Ups and Downs of Innovations Unbridled enthusiasm Efficacy Harsh realities ST Ultimate applicability Innovation Saf ety Thoughtful adaptations
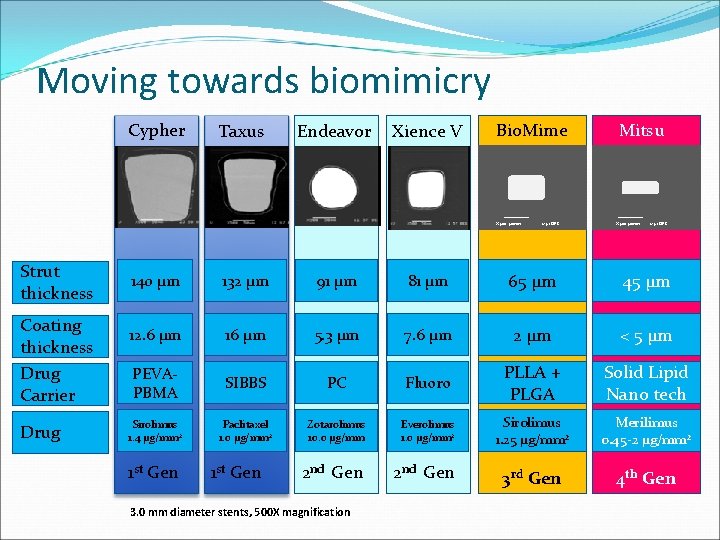
Moving towards biomimicry Cypher Taxus Endeavor Xience V Bio. Mime X 500 50 mm Strut thickness Coating thickness Drug Carrier Drug 12 57 DES Mitsu X 500 50 mm 12 57 DES 140 µm 132 µm 91 µm 81 µm 65 µm 45 µm 12. 6 µm 16 µm 5. 3 µm 7. 6 µm 2 µm < 5 µm PEVAPBMA SIBBS PC Fluoro PLLA + PLGA Solid Lipid Nano tech Sirolimus 1. 4 µg/mm 2 Paclitaxel 1. 0 µg/mm 2 Zotarolimus 10. 0 µg/mm Everolimus 1. 0 µg/mm 2 Sirolimus 1. 25 µg/mm 2 Merilimus 0. 45 -2 µg/mm 2 2 nd Gen 3 rd Gen 4 th Gen 1 st Gen 3. 0 mm diameter stents, 500 X magnification
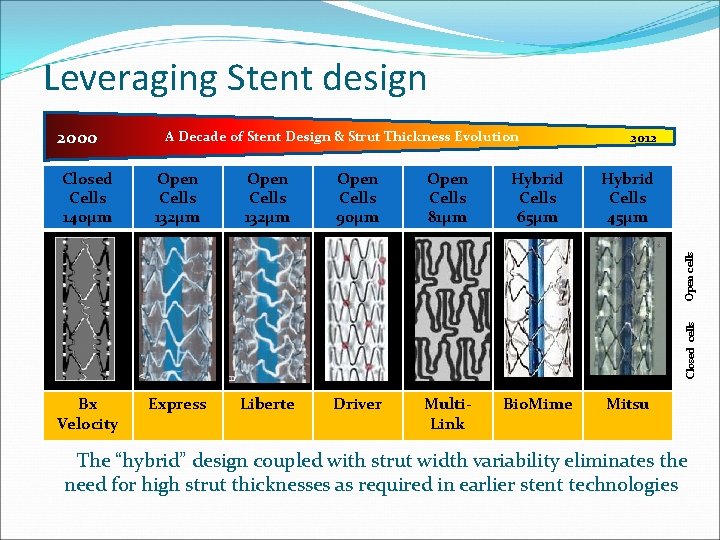
Leveraging Stent design 2000 A Decade of Stent Design & Strut Thickness Evolution 2012 Open Cells 132µm Open Cells 90µm Open Cells 81µm Hybrid Cells 65µm Hybrid Cells 45µm Bx Velocity Express Liberte Driver Multi. Link Bio. Mime Mitsu Closed cells Open cells Closed Cells 140µm The “hybrid” design coupled with strut width variability eliminates the need for high strut thicknesses as required in earlier stent technologies
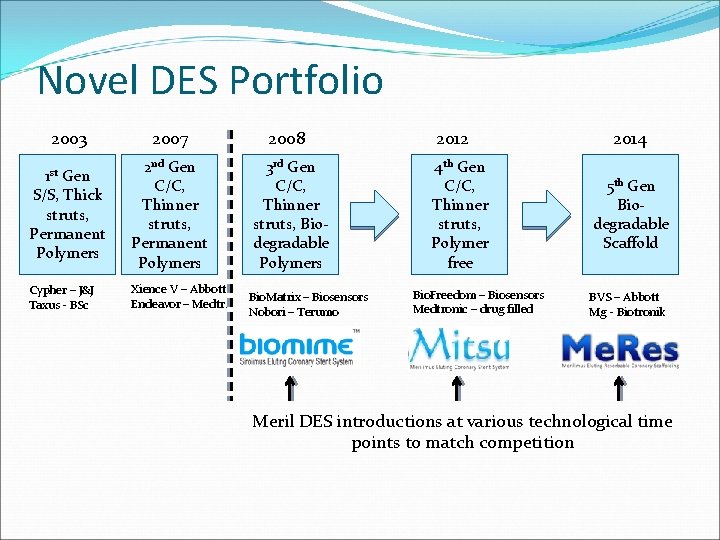
Novel DES Portfolio 2003 2007 2008 Gen S/S, Thick struts, Permanent Polymers 2 nd Gen C/C, Thinner struts, Permanent Polymers 3 rd Gen C/C, Thinner struts, Biodegradable Polymers Cypher – J&J Taxus - BSc Xience V – Abbott Endeavor – Medtr. 1 st Bio. Matrix – Biosensors Nobori – Terumo 2012 4 th Gen C/C, Thinner struts, Polymer free Bio. Freedom – Biosensors Medtronic – drug filled 2014 5 th Gen Biodegradable Scaffold BVS – Abbott Mg - Biotronik Meril DES introductions at various technological time points to match competition

Bio. Mime Stent Architecture � Cobalt chromium (L 605) platform with 65µm strut thickness. � Hybrid cell design comprising of an intelligent mix of open and close cells resulting in excellent radial strength with a high flexibility. � Unique strut width variability that ensure a <3% recoil and 0. 29% foreshortening � Special electro-polishing technique eliminates surface nickel oxides SEM image of crimped Bio. Mime SES at 50 x Closed cell at edges Open cell in mid - segment Hybrid design
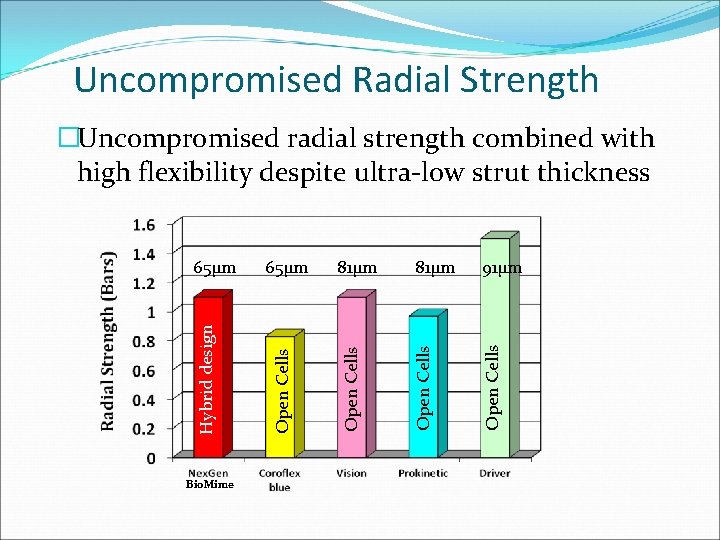
Uncompromised Radial Strength Bio. Mime 81µm 91µm Open Cells 65µm Open Cells Hybrid design 65µm Open Cells �Uncompromised radial strength combined with high flexibility despite ultra-low strut thickness
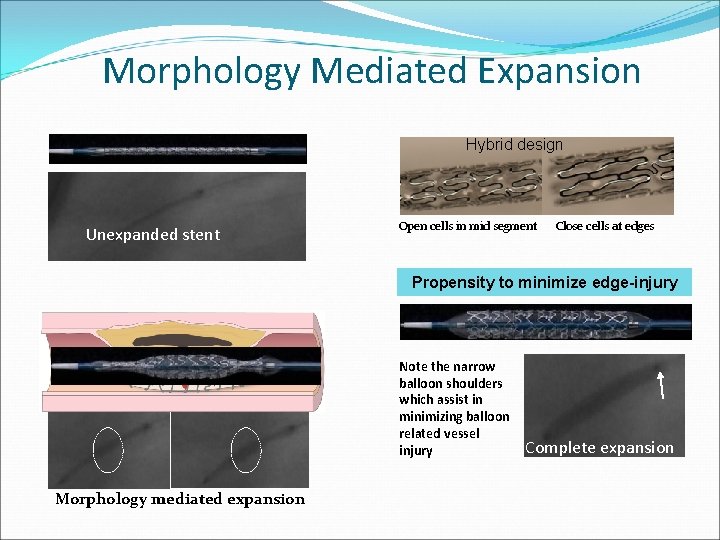
Morphology Mediated Expansion Hybrid design Unexpanded stent Open cells in mid segment Close cells at edges Propensity to minimize edge-injury Note the narrow balloon shoulders which assist in minimizing balloon related vessel injury Morphology mediated expansion Complete expansion

Excellent Side Branch Access The area of the largest circle circumscribable in the cell of the stent expanded to the nominal diameter: Tc = 0. 71 mm 2 The expanded BIOMIME 3. 0 x 16 mm stent after side branch expansion Expanded cell perimeter that ensures side branch access: KSBA = 11. 29 mm Expanded cell area that ensures side branch access: TSBA = 8. 00 mm 2 Data on file with Meril Life Sciences.

The Drug: Sirolimus � Sirolimus is a good choice considering that it acts on the common final pathway of cell division cycle without exceptional risk of necrosis induction 1. 25µgm/mm 2 of Sirolimus loading (Cypher has 1. 4µgm/mm 2) Data on file with Meril Life Sciences.
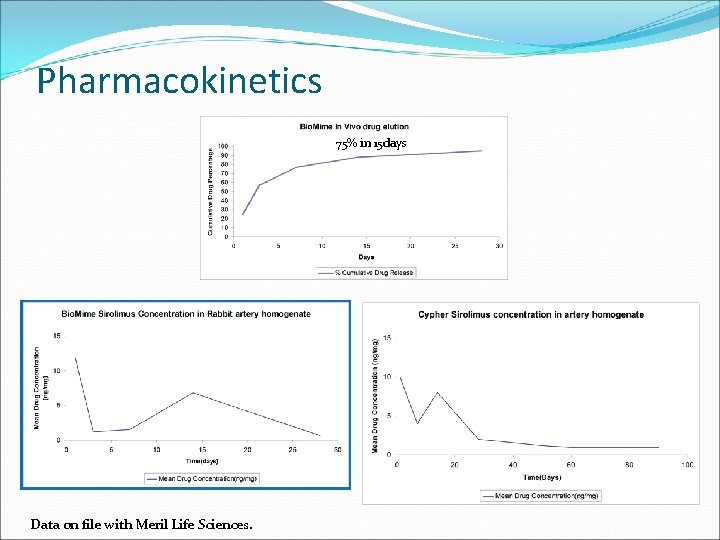
Pharmacokinetics 75% in 15 days Data on file with Meril Life Sciences.

PLGA PLLA About Bio. Poly. TM � Bio. Poly is Meril’s propriety bio-degradable co- polymer formulation. � The principle mode of Bio. Poly. TM degradation is hydrolysis via mass loss & by products are excreted as CO 2 and H 2 O via Kreb’s cycle Before inflation � The material offers uniformity in stent coating & thin coating of <2µm is possible � Does-not web, crack, lump or on stent or balloon surface After inflation
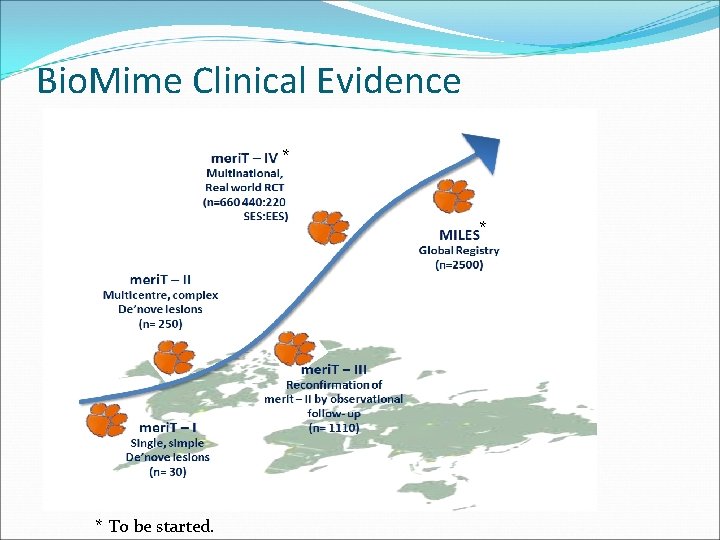
Bio. Mime Clinical Evidence * * * To be started.
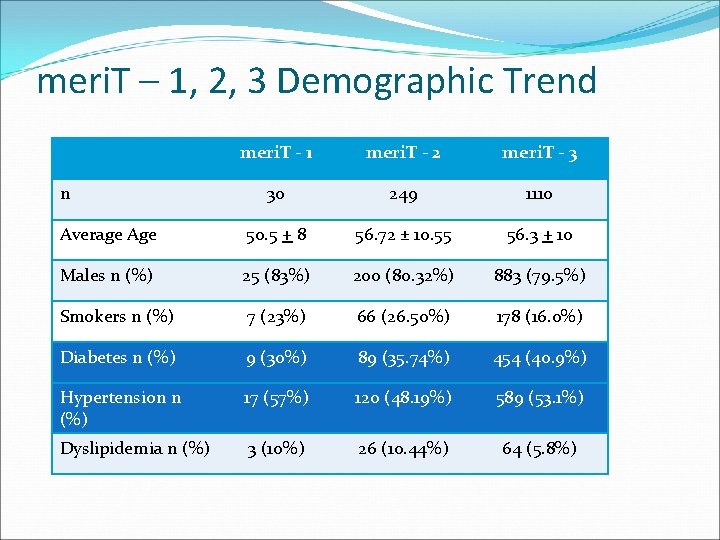
meri. T – 1, 2, 3 Demographic Trend meri. T - 1 meri. T - 2 meri. T - 3 30 249 1110 Average Age 50. 5 + 8 56. 72 ± 10. 55 56. 3 + 10 Males n (%) 25 (83%) 200 (80. 32%) 883 (79. 5%) Smokers n (%) 7 (23%) 66 (26. 50%) 178 (16. 0%) Diabetes n (%) 9 (30%) 89 (35. 74%) 454 (40. 9%) Hypertension n (%) 17 (57%) 120 (48. 19%) 589 (53. 1%) Dyslipidemia n (%) 3 (10%) 26 (10. 44%) 64 (5. 8%) n
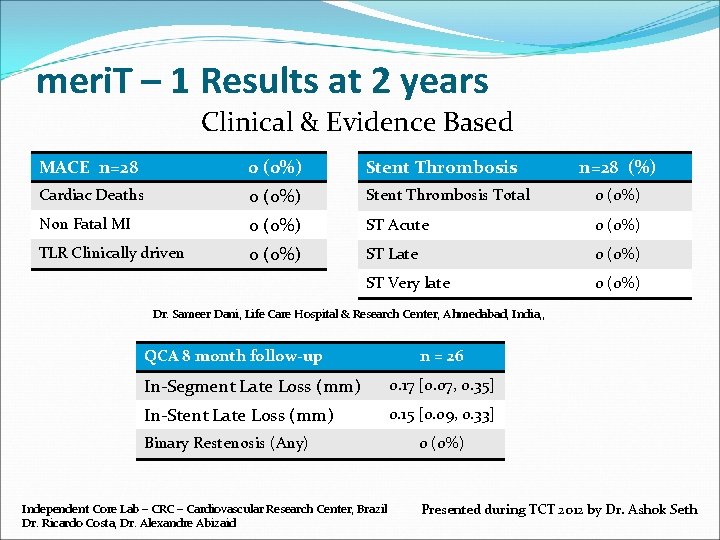
meri. T – 1 Results at 2 years Clinical & Evidence Based MACE n=28 0 (0%) Stent Thrombosis Cardiac Deaths 0 (0%) Stent Thrombosis Total 0 (0%) Non Fatal MI 0 (0%) ST Acute 0 (0%) TLR Clinically driven 0 (0%) ST Late 0 (0%) ST Very late 0 (0%) n=28 (%) Dr. Sameer Dani, Life Care Hospital & Research Center, Ahmedabad, India, , QCA 8 month follow-up n = 26 In-Segment Late Loss (mm) 0. 17 [0. 07, 0. 35] In-Stent Late Loss (mm) 0. 15 [0. 09, 0. 33] Binary Restenosis (Any) Independent Core Lab – CRC – Cardiovascular Research Center, Brazil Dr. Ricardo Costa, Dr. Alexandre Abizaid 0 (0%) Presented during TCT 2012 by Dr. Ashok Seth
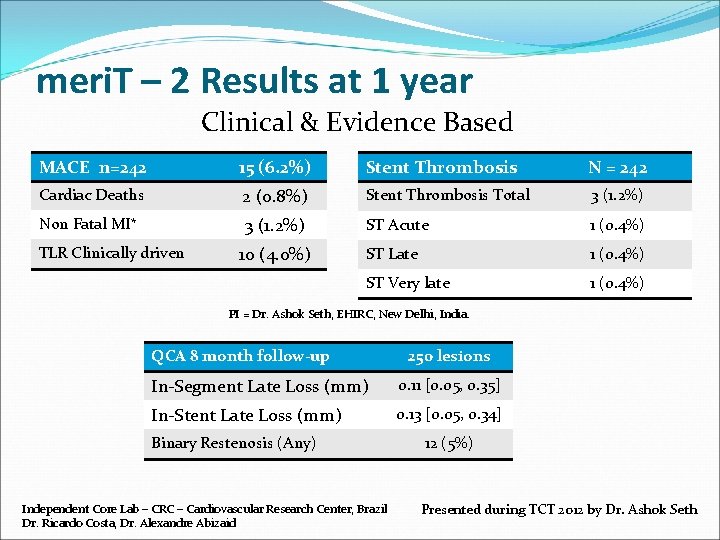
meri. T – 2 Results at 1 year Clinical & Evidence Based MACE n=242 15 (6. 2%) Stent Thrombosis N = 242 Cardiac Deaths 2 (0. 8%) Stent Thrombosis Total 3 (1. 2%) Non Fatal MI* 3 (1. 2%) ST Acute 1 (0. 4%) ST Late 1 (0. 4%) ST Very late 1 (0. 4%) TLR Clinically driven 10 (4. 0%) PI = Dr. Ashok Seth, EHIRC, New Delhi, India. QCA 8 month follow-up 250 lesions In-Segment Late Loss (mm) 0. 11 [0. 05, 0. 35] In-Stent Late Loss (mm) 0. 13 [0. 05, 0. 34] Binary Restenosis (Any) Independent Core Lab – CRC – Cardiovascular Research Center, Brazil Dr. Ricardo Costa, Dr. Alexandre Abizaid 12 (5%) Presented during TCT 2012 by Dr. Ashok Seth
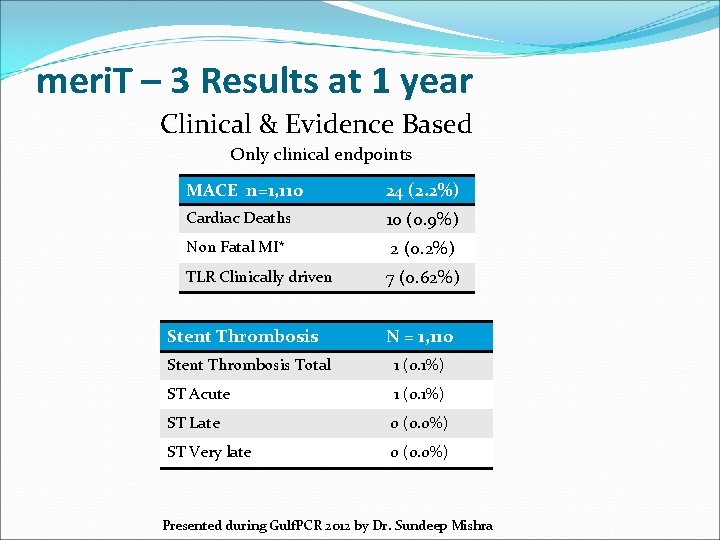
meri. T – 3 Results at 1 year Clinical & Evidence Based Only clinical endpoints MACE n=1, 110 24 (2. 2%) Cardiac Deaths 10 (0. 9%) Non Fatal MI* 2 (0. 2%) TLR Clinically driven Stent Thrombosis 7 (0. 62%) N = 1, 110 Stent Thrombosis Total 1 (0. 1%) ST Acute 1 (0. 1%) ST Late 0 (0. 0%) ST Very late 0 (0. 0%) Presented during Gulf. PCR 2012 by Dr. Sundeep Mishra

Beyond Bio. Mime…
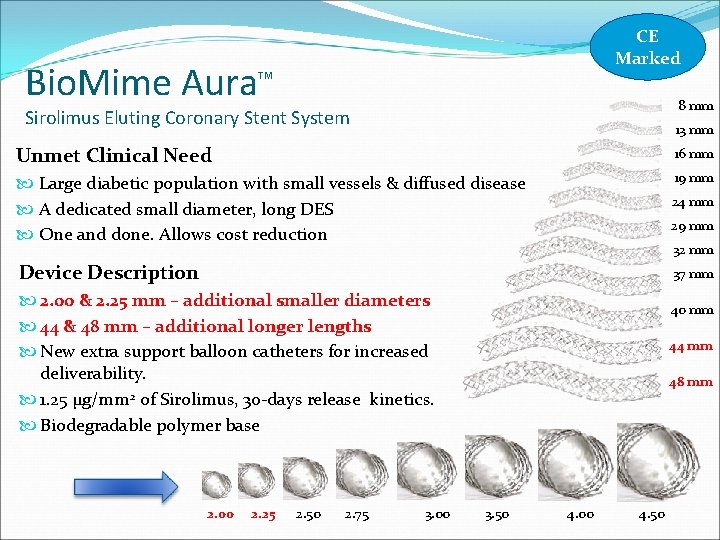
Bio. Mime Aura CE Marked TM 8 mm Sirolimus Eluting Coronary Stent System 13 mm Unmet Clinical Need 16 mm Large diabetic population with small vessels & diffused disease A dedicated small diameter, long DES One and done. Allows cost reduction 19 mm 24 mm 29 mm 32 mm Device Description 37 mm 2. 00 & 2. 25 mm – additional smaller diameters 44 & 48 mm – additional longer lengths New extra support balloon catheters for increased deliverability. 1. 25 µg/mm 2 of Sirolimus, 30 -days release kinetics. Biodegradable polymer base 2. 00 2. 25 2. 50 2. 75 3. 00 40 mm 44 mm 48 mm 3. 50 4. 00 4. 50

Bio. Mime Morph TM Sirolimus Eluting Coronary Stent System Unmet Clinical Need A stent that matches the diameter differentials from distal to proximal in long diffused lesions. One & done. Device Description Unique tapered coronary stent. Tapering diameters (1/2 sized) Prox. to Distal – 3. 50→ 3. 00 mm & 3. 00→ 2. 50 mm Longest lengths – 50, 60 mm Mounted on a newly created extra support Rx balloon catheter with tapered diameters 1. 25 µg/mm 2 of Sirolimus, 30 -days release kinetics. Biodegradable polymer base = D 1 = D 2 D 1 > D 2 Ø 3. 50 Ø 3. 00 Ø 2. 50 Proximal l – 50, 60 mm Distal Ø 3. 50 Ø 3. 00 Actual device photographs. Data on file Meril Life Sciences. Ø 3. 00 Ø 2. 50
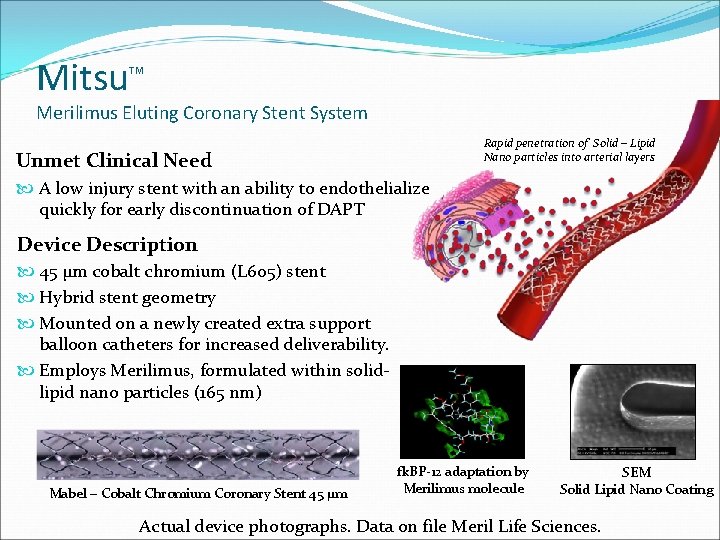
Mitsu TM Merilimus Eluting Coronary Stent System Rapid penetration of Solid – Lipid Nano particles into arterial layers Unmet Clinical Need A low injury stent with an ability to endothelialize quickly for early discontinuation of DAPT Device Description 45 µm cobalt chromium (L 605) stent Hybrid stent geometry Mounted on a newly created extra support balloon catheters for increased deliverability. Employs Merilimus, formulated within solidlipid nano particles (165 nm) Mabel – Cobalt Chromium Coronary Stent 45 µm fk. BP-12 adaptation by Merilimus molecule SEM Solid Lipid Nano Coating Actual device photographs. Data on file Meril Life Sciences.

Me. Res TM Sirolimus Eluting Bio. Resorbable Coronary Scaffold Unmet Clinical Need A completely resorbable scaffold which allows for restoration of normal vascular function, eliminates need for long term DAPT and ensures freedom from MACE/ST. Device Description New PLA formulation for enhanced radial strength Hybrid stent geometry Mounted on a newly created extra support balloon catheters for increased deliverability. Employs 1. 25 µg/mm 2 of Sirolimus, timed to elute over 90 -days from non-inflammatory, biodegradable polymers. Expanded Open-cell Actual device photographs. Data on file Meril Life Sciences. Expanded Close-cell

Thank you for your attention!
- Slides: 24