Notes SPI 0807 9 9 Periodic Table Science

Notes SPI 0807. 9. 9 Periodic Table Science Tennessee SPI Objective: Instructions Use the click periodic tablefrom to determine 1) First on "View" the top-menuthe bar. properties Then click on "Slide Show" from the drop-down menu. of 2)an element. 3) Then use the right-arrow button on your keyboard to Check for Understanding advance through this presentation. Identify the atomic number, atomic mass, number of protons, neutrons, and electrons in atoms using the periodic table. Essential Question How do I use the periodic table to determine the atomic number, the atomic mass, the number of sub-atomic particles, & other properties in an atom?
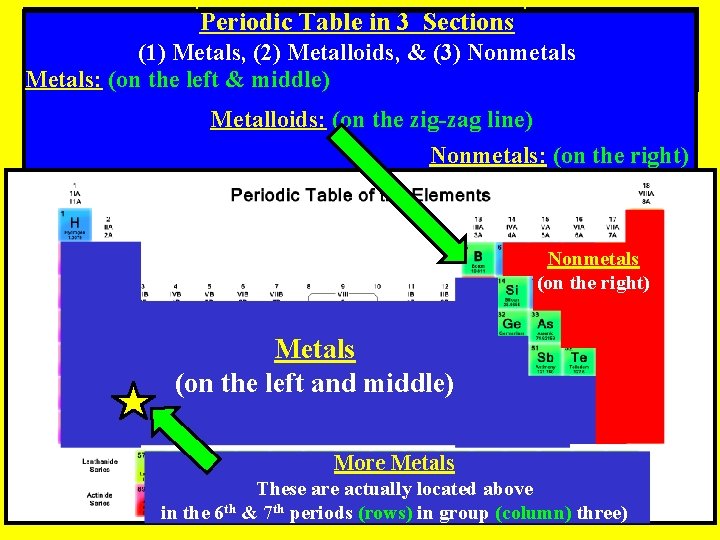
Periodic Table in 3 Sections (1) Metals, (2) Metalloids, & (3) Nonmetals Metals: (on the left & middle) Metalloids: (on the zig-zag line) Nonmetals: (on the right) Nonmetals (on the right) Metals (on the left and middle) More Metals These are actually located above in the 6 th & 7 th periods (rows) in group (column) three)

Periodic Table in 3 Sections (1) Metals, (2) Metalloids, & (3) Nonmetals Metals: (on the left of the periodic table) Ømetal elements tend to be shiny, malleable, ductile, and are good conductors of electricity and thermal energy (heat)

Periodic Table in 3 Sections (1) Metals, (2) Metalloids, & (3) Nonmetals: (on the right side of the periodic table) ØNonmetal elements are generally not shiny, not malleable, not ductile, and are not good conductors of electricity or thermal energy (heat)

Periodic Table in 3 Sections (1) Metals, (2) Metalloids, & (3) Nonmetals Metalloids: (on the zig-zag line) ØHave some properties of metals and some properties of nonmetals, are called semi-conductors
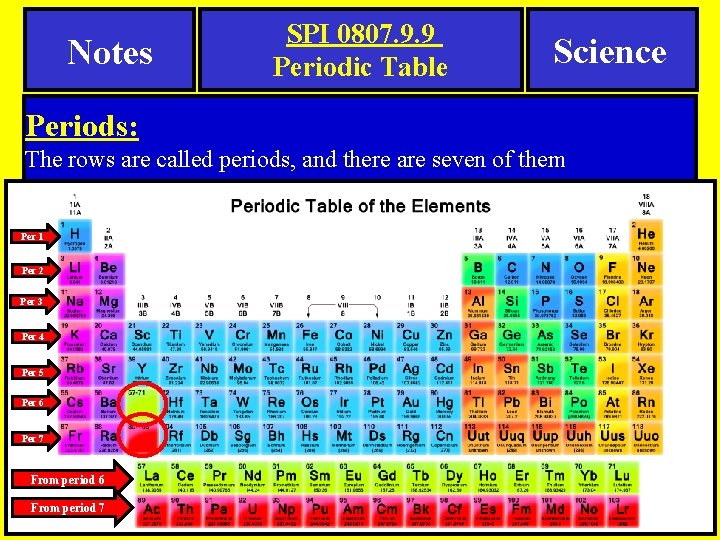
Notes SPI 0807. 9. 9 Periodic Table Science Periods: The rows are called periods, and there are seven of them Per 1 Per 2 Per 3 Per 4 Per 5 Per 6 Per 7 From period 6 From period 7
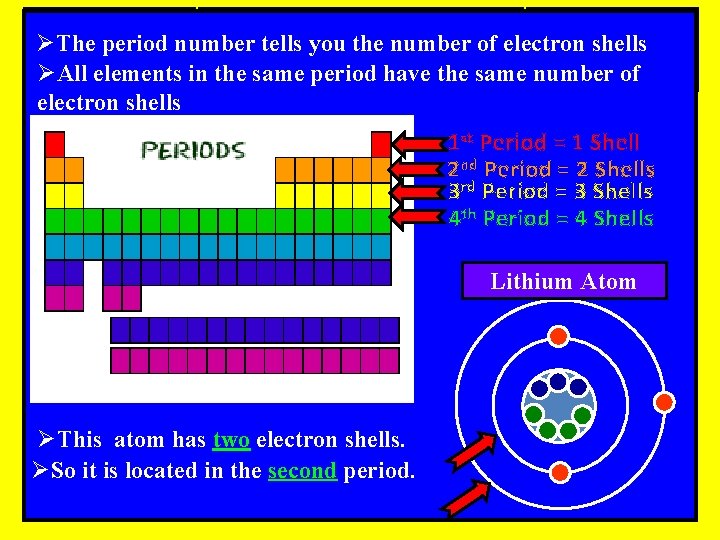
ØThe period number tells you the number of electron shells ØAll elements in the same period have the same number of electron shells 1 st Period = 1 Shell 2 nd Period = 2 Shells 3 rd Period = 3 Shells 4 th Period = 4 Shells Lithium Atom ØThis atom has two electron shells. ØSo it is located in the second period.
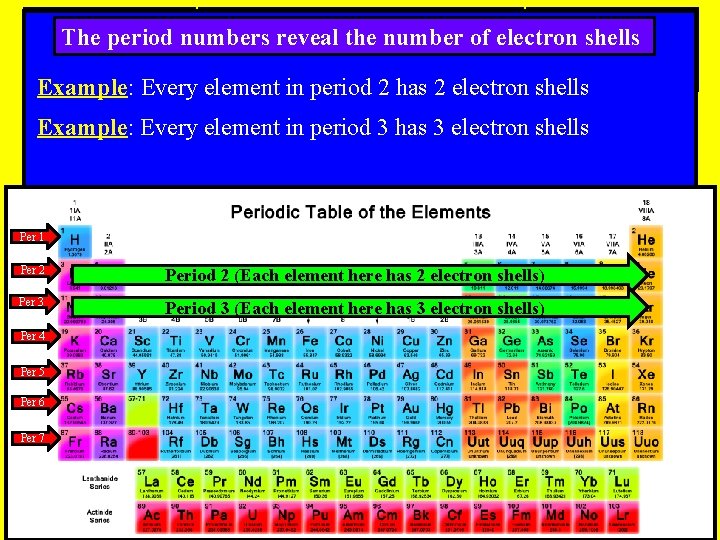
The period numbers reveal the number of electron shells Example: Every element in period 2 has 2 electron shells Example: Every element in period 3 has 3 electron shells Per 1 Per 2 Period 2 (Each element here has 2 electron shells) Per 3 Period 3 (Each element here has 3 electron shells) Per 4 Per 5 Per 6 Per 7

. Break Time! SPI 0807. 9. 9 Periodic Table Break Time! Stand Stretch! Ø You have been sitting for a mighty long time. Ø Let's take a 1 minute break to stand stretch.
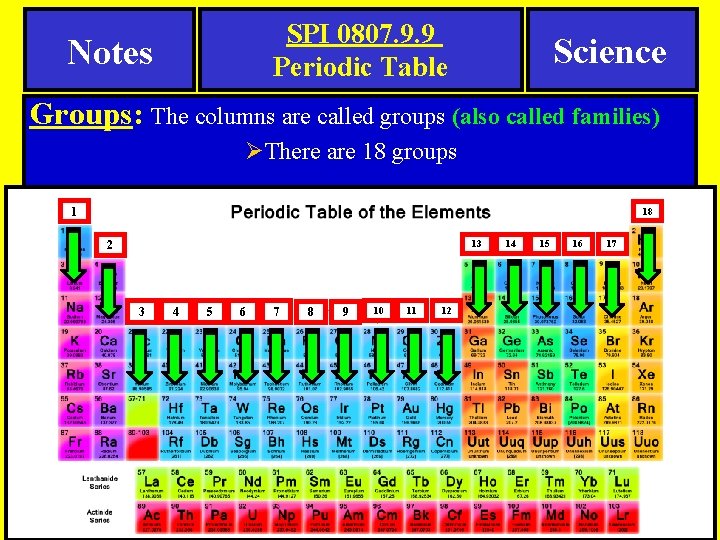
SPI 0807. 9. 9 Periodic Table Notes Science Groups: The columns are called groups (also called families) ØThere are 18 groups 1 18 13 2 3 4 5 6 7 8 9 10 11 12 14 15 16 17
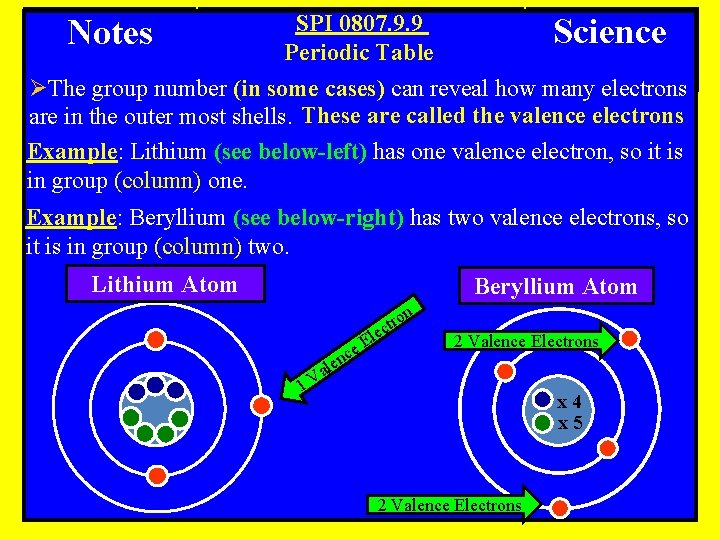
SPI 0807. 9. 9 Science Periodic Table ØThe group number (in some cases) can reveal how many electrons are in the outer most shells. These are called the valence electrons Example: Lithium (see below-left) has one valence electron, so it is in group (column) one. Notes Example: Beryllium (see below-right) has two valence electrons, so it is in group (column) two. Lithium Atom Beryllium Atom n o ctr e c l e. E n ale 2 Valence Electrons 1 V x 4 x 5 2 Valence Electrons
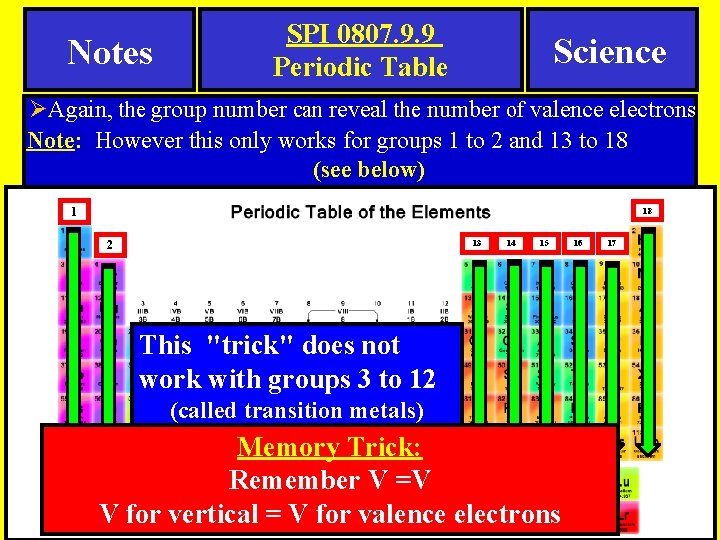
Notes SPI 0807. 9. 9 Periodic Table Science ØAgain, the group number can reveal the number of valence electrons Note: However this only works for groups 1 to 2 and 13 to 18 (see below) 1 18 13 2 14 15 This "trick" does not work with groups 3 to 12 (called transition metals) Memory Trick: Remember V =V V for vertical = V for valence electrons 16 17

Notes SPI 0807. 9. 9 Periodic Table Science ØAgain, the group number can reveal the number of valence electrons Reminder: This "trick" only works for groups 1 to 2 and 13 to 18 (see below) 1 2 Example: Every element in group one has only one valence electron. Example: Every element in group two has exactly two valence electrons.

Notes SPI 0807. 9. 9 Periodic Table Science ØAgain, the group number can reveal the number of valence electrons Reminder: This "trick" only works for groups 1 to 2 and 13 to 18 (see below) For Groups 13 to 18: Simply subtract ten from each number Example: For group 13 subtract ten, and you get three. So every element in group 13 has three valence electrons Example: For group 14 subtract ten, and you get four. So every element in group 14 has four valence electrons 18 13 14 15 16 17

Reactivity in Elements For groups 1 to 2: ØReactivity increases from right to left For groups 13 to 17: (Leave out group 18…they are non-reactive!) ØReactivity increases from left to right Groups 1 to 2 Groups 13 to 17 1 Again, ignore groups 3 to 12 for now (called transition metals) 14 15 16 17 Non-reactive 13 2

Questions SPI 0807. 9. 9 Periodic Table Science Periodic Table-Question Here is an example of a question you might see on a quiz or test.
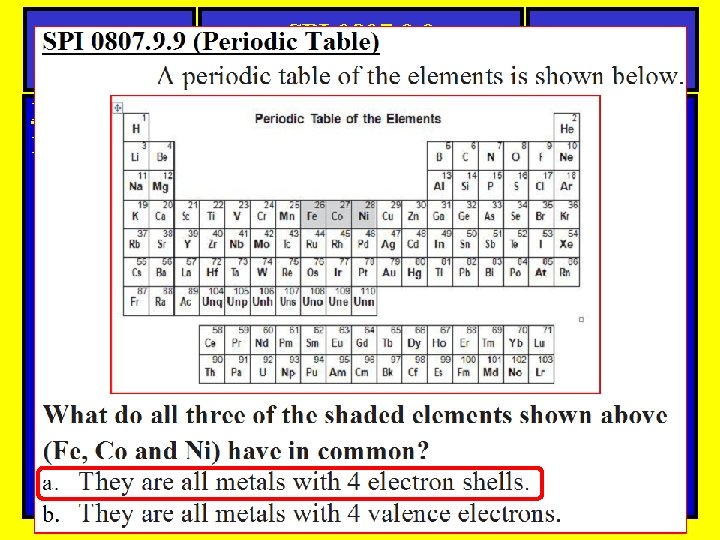
Questions SPI 0807. 9. 9 Periodic Table Science Periodic Table-Question Here is an example of a question you might see on a quiz or test.
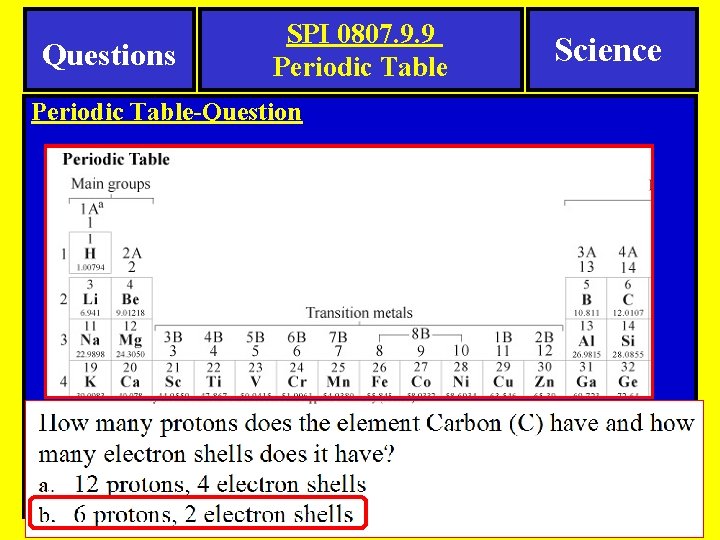
Questions SPI 0807. 9. 9 Periodic Table-Question Science

Questions SPI 0807. 9. 9 Periodic Table Science Periodic Table-Question Here is an example of a question you might see on a quiz or test.
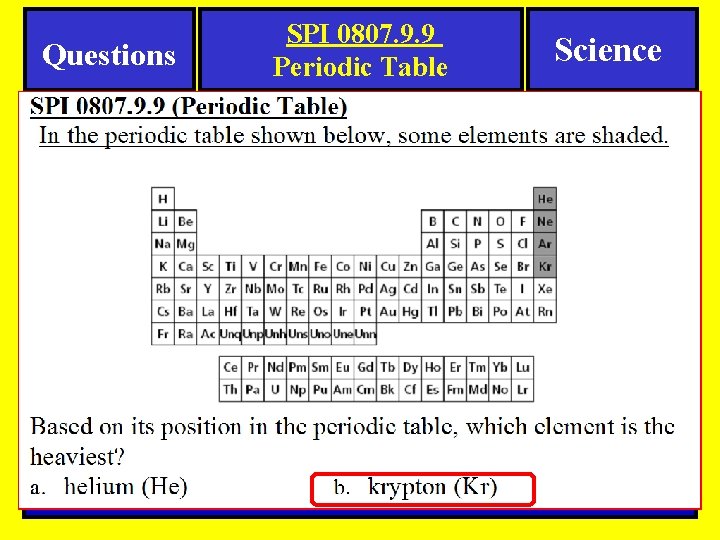
Questions SPI 0807. 9. 9 Periodic Table Science Periodic Table-Question Here is an example of a question you might see on a quiz or test.
- Slides: 20