Notes SPI 0807 9 10 Reactants Products Science










- Slides: 14

Notes SPI 0807. 9. 10 Reactants & Products Science Tennessee SPI Objective: Identify the reactants and products of a chemical reaction. Essential Question(s) How do I recognize reactants and products in chemical equations? I Can Statement I can identify reactants and products in a chemical equation. Success Criteria I can prove that I understand this topic by showing you the reactants and products in a chemical equation.

Notes SPI 0807. 9. 10 Reactants & Products Science Chemical Reactions ØAre represented by chemical equations Chemical Equations ØHave two parts: (1) reactants and (2) products ØReactants: are the "ingredients" of a chem reaction. These are things you are combining… the things you start with. ØProducts: are things that you produce in a chemical reaction… the things you end up with. Product Reactants (Hydrogen & Oxygen) (water) Example: 2 H 2 + O 2 2 H 2 O

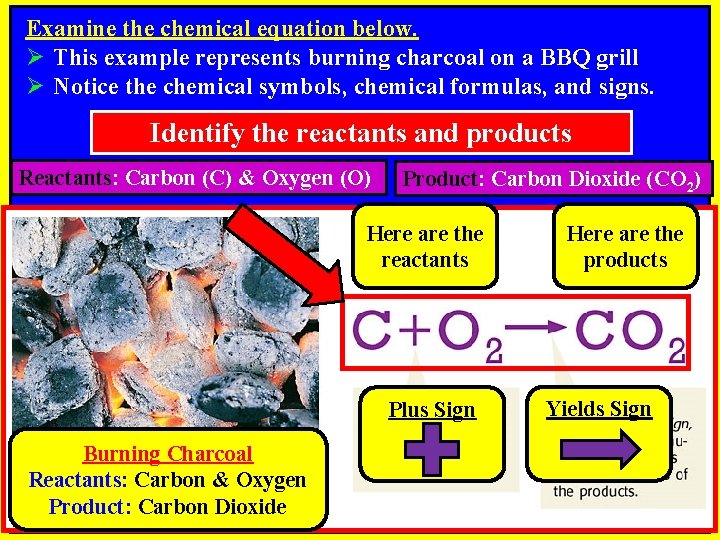
Examine the chemical equation below. Ø This example represents burning charcoal on a BBQ grill Ø Notice the chemical symbols, chemical formulas, and signs. Identify the reactants and products Reactants: Carbon (C) & Oxygen (O) Product: Carbon Dioxide (CO 2) Here are the reactants Plus Sign Burning Charcoal Reactants: Carbon & Oxygen Product: Carbon Dioxide Here are the products Yields Sign

Notes SPI 0807. 9. 10 Reactants & Products Science Examine the reactants Reactants- starting materials in a chemical reaction Remember: the arrow ALWAYS points away from the reactants! Here are the reactants

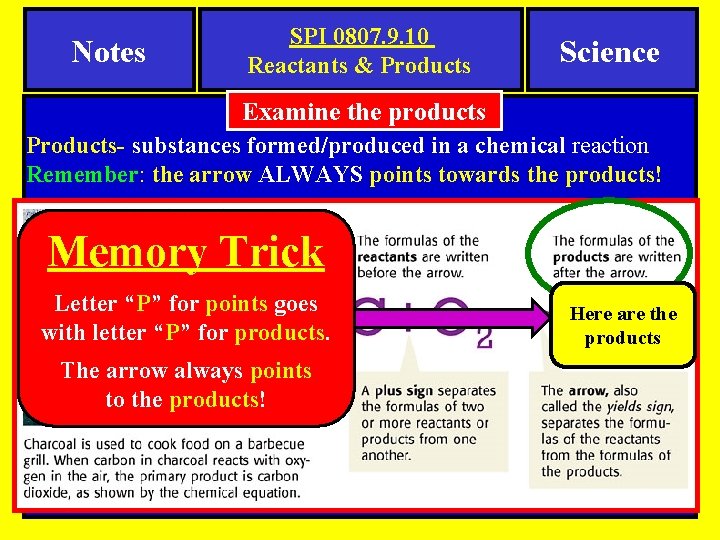
Notes SPI 0807. 9. 10 Reactants & Products Science Examine the products Products- substances formed/produced in a chemical reaction Remember: the arrow ALWAYS points towards the products! Memory Trick Letter “P” for points goes with letter “P” for products. The arrow always points to the products! Here are the products

Review SPI 0807. 9. 10 Reactants & Products 2 H 2 + O 2 Science 2 H 2 O Identify the REACTANTS in this equation. Reminder: The arrow always points towards the product(s)! Reactants 2 H 2 and O 2 …or you could simply identify the reactants as H (hydrogen) and O (oxygen)

Review SPI 0807. 9. 10 Reactants & Products N 2 + 3 H 2 Science 2 NH 3 Identify the PRODUCT in this equation. Reminder: The arrow always points towards the product(s)! Product 2 NH 3 …or you could simply identify the product as NH 3 (ammonia)

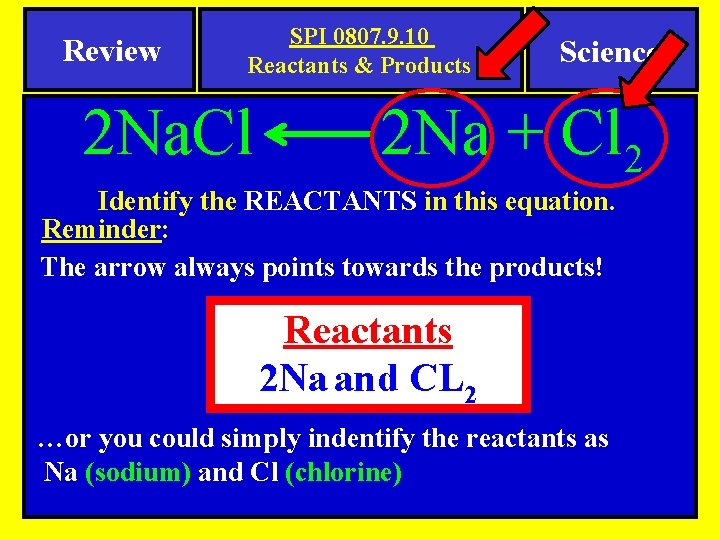
Review SPI 0807. 9. 10 Reactants & Products 2 Na. Cl Science 2 Na + Cl 2 Identify the REACTANTS in this equation. Reminder: The arrow always points towards the products! Reactants 2 Na and CL 2 …or you could simply indentify the reactants as Na (sodium) and Cl (chlorine)

Review SPI 0807. 9. 10 Reactants & Products Sn + 2 H 2 O Science Sn. O 2 + 2 H 2 Identify the PRODUCTS in this equation. Reminder: The arrow always points towards the products! Products Sn and 2 H 2 O …or you could simply indentify the products as Sn (tin) and H 2 O (water)

Review SPI 0807. 9. 10 Science Reactants & Products Questions The arrow always points towards the product(s)!

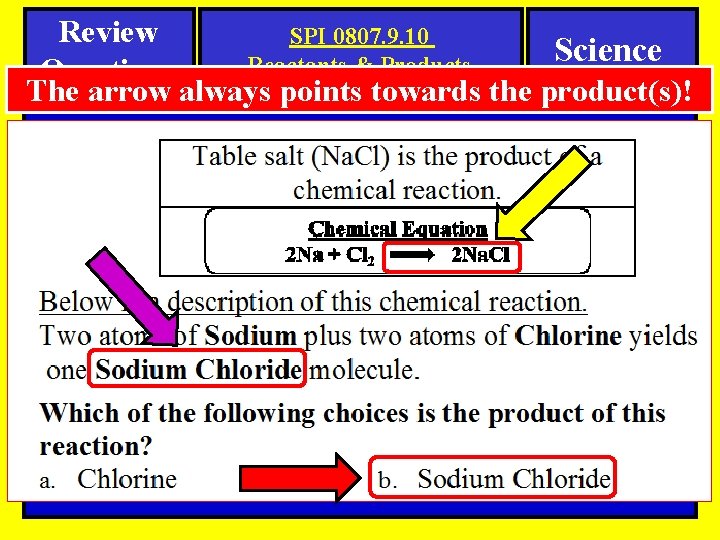
Review Questions SPI 0807. 9. 10 Reactants & Products Science The arrow always points towards the product(s)! Reactants Products

Review Questions SPI 0807. 9. 10 Reactants & Products Science The arrow always points towards the product(s)!

Review Questions SPI 0807. 9. 10 Reactants & Products Science The arrow always points towards the product(s)! Product Reactants

Review Questions SPI 0807. 9. 10 Reactants & Products Science The arrow always points towards the product(s)!