Notes One Unit Two Pages 308 317 Balancing

Notes One Unit Two Pages 308 -317 Balancing chemical equations

Making Tricycles • Tricycles are constructed from two components… • Frame…. F • Wheel… W • FW 3 F 2 W 4
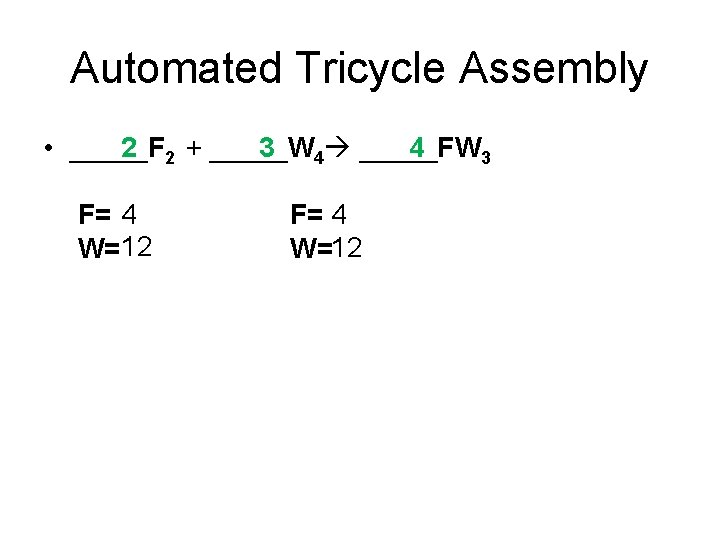
Automated Tricycle Assembly • _____F 2 2 + _____W 3 4 _____FW 4 3 F= 4 W=12
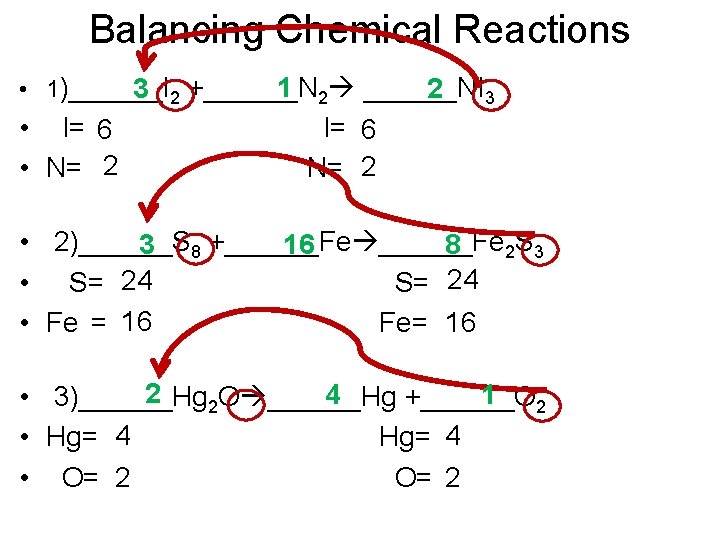
Balancing Chemical Reactions • 1)______I 1 2 ______NI 3 2 +______N 2 3 • I= 6 • N= 2 I= 6 N= 2 • 2)______S 3 8 +______Fe 16 8 2 S 3 • S= 24 • Fe = 16 Fe= 16 2 4 1 • 3)______Hg 2 O ______Hg +______O 2 • Hg= 4 • O= 2

Notes Two Unit Two Writing Chemical Equations Pages 318 -324 Qualitative Information Quantitative Information Reactants and products Writing and Balancing Chemical Equations

Qualitative Information Solid, Liquid, gaseous or dissolved Chemicals. Cl 2(g) gas S 8(g) gas HCl(g) gas Cu(l) liquid Br 2(l) liquid HCl(aq) dissolved O 2(g) gas H 2(g)gas Fe+2(aq)dissolved N 2(g)gas I 2(c) Solid Fe 2 O 3(c) Solid P 4(s) Solid Fe(s)Solid Na. Cl(aq)dissolved

Quantitative Information Subscripts How many atoms of an element that are in a compound. Al= Two ? O = Nine ? Al 2(CO 3)3 C = Three ?
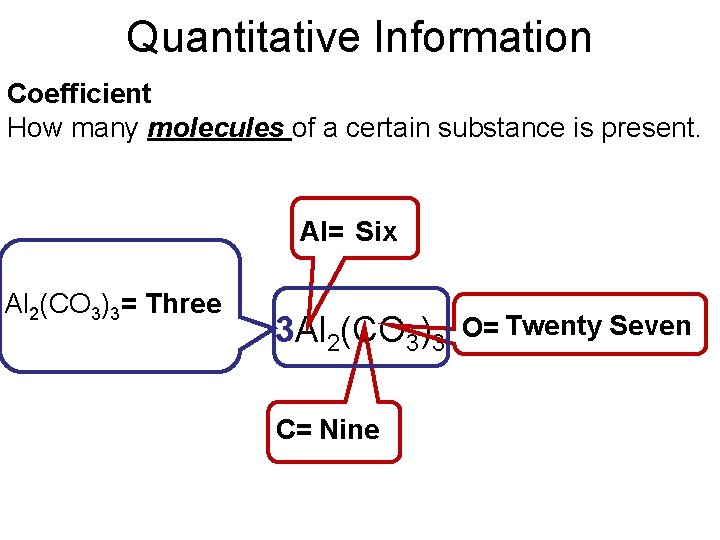
Quantitative Information Coefficient How many molecules of a certain substance is present. Al= Six ? Al 2(CO 3)3= Three ? 3 Al 2(CO 3)3 C= Nine ? Seven O= Twenty ?
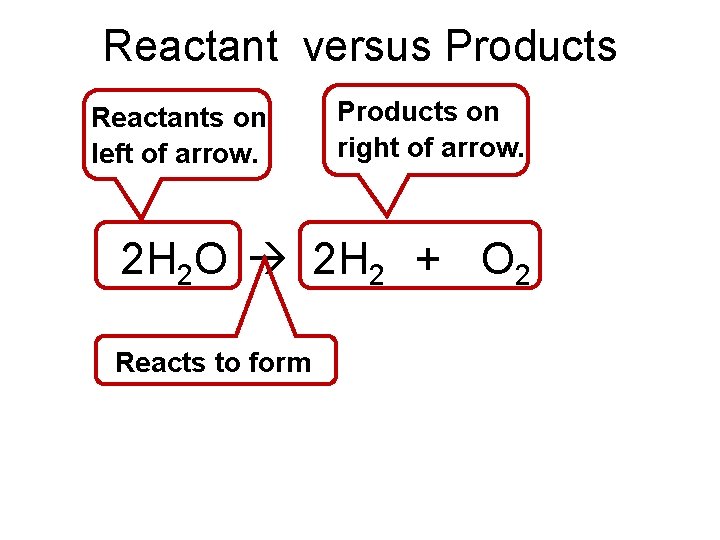
Reactant versus Products Reactants on left of arrow. Products on right of arrow. 2 H 2 O 2 H 2 + O 2 Reacts to form

Things to Remember When making formulas from names… 1) The endings –ide, –ate, –ite cation and anion lists Sodium Dichromate Lead(IV) Sulfate (Na+1)_( 1 2 Cr O -2 )_ 2 7 (Pb+4)_( 4 2 SO -2)_ 4 2) The endings –ous acid or –ic acid Na 2 Cr 2 O 7 Pb(SO 4)2 acid list 3) common sense names: water H 2 O sulfur trioxide SO 3 carbon dioxide CO 2 dinitrogen trioxide N 2 O 3 mono, di, tri, tetra, penta, hexa, octa, nona, deca 4) Otherwise I) Special Elementals II) Or periodic table

Where Do We Find The Information For… oxygen… O 2(g)…No -ide, -ite, -ate, -ous Special elements, then Periodic table oxygen difluoride…OF 2…Common Sense, mono, di, … iron(II) chloride… Fe. Cl 2… ends in ide…Cation/Anion Sulfur hexafluoride. . . SF 6…Common Sense, mono, di, … hydrochloric acid…HCl… ends in -ic acid or -ous acid Iron metal…. Fe(s)…No -ide, -ite, -ate, -ous Special elements, then Periodic table nitrogen…. N 2(g)…No -ide, -ite, -ate, -ous Special elements, then Periodic table

Writing and Balancing Chemical Equations • Step 1 – Word Equation • Step 2 – Make Correct Formulas • Step 3 – Balance the Equation

Reaction Types 1) Combustion: hydrocarbon+ oxygen carbon dioxide + water C 5 H 12(l) + 8 O 2(g) 5 CO 2(g) + 6 H 2 O(l) more to one 2) Synthesis: A + B AB 4 Sn(s) + S 8(c) 4 Sn. S 2(s) one to more 3) Decomposition: AB A + B 2 H 2 O(l) 2 H 2(g) + O 2(g) Single element 4) Single displacement: A + BC AC + B 2 Al(s) + 6 H 2 O(l) 2 Al(OH)3(aq) + 3 H 2(g) 5) Double displacement: AB + CD AD + CB Ag. NO 3(aq) + KI(aq) Ag. I(s) + KNO 3(aq) 6) Acid-base: acid + Base Water + Salt 6 HCl(aq) + 2 Al(OH)3(s) 6 H 2 O(l) + 2 Al. Cl 3(aq)
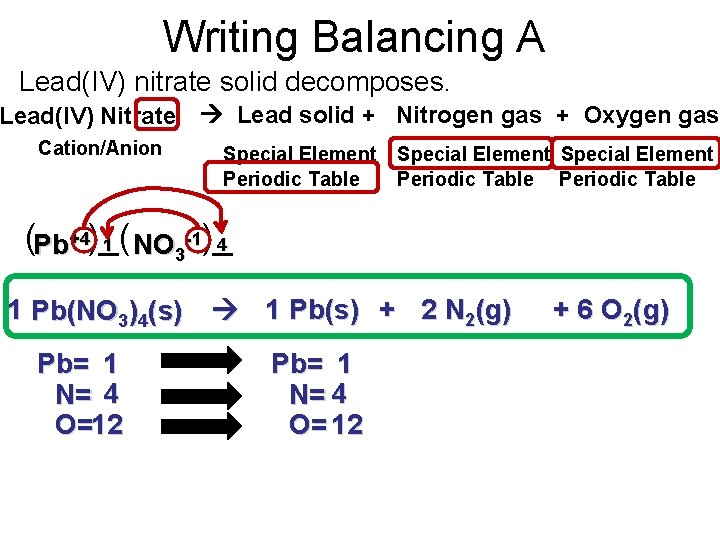
Writing Balancing A Lead(IV) nitrate solid decomposes. Lead(IV) Nitrate Lead solid + Nitrogen gas + Oxygen gas Cation/Anion Special Element Periodic Table (Pb+4)_( 4 1 NO -1)_ 3 1 Pb(NO 3)4(s) 1 Pb(s) + 2 N 2(g) Pb= 1 N= 4 O=12 Pb= 1 N= 4 O= 12 + 6 O 2(g)
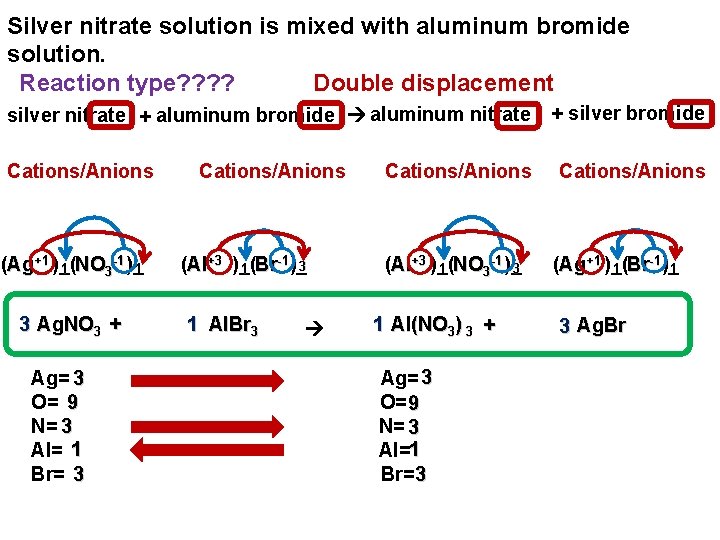
Silver nitrate solution is mixed with aluminum bromide solution. Reaction type? ? Double displacement silver nitrate + aluminum bromide aluminum nitrate + silver bromide Cations/Anions -1 (Ag+1 )_( 1 1 NO 3 )_ 3 Ag. NO 3 + Ag= 3 O= 9 N= 3 Al= 1 Br= 3 Cations/Anions -1 3 (Al+3 )_( 1 Br )_ 1 Al. Br 3 Cations/Anions -1 (Al+3 )_( 1 NO 3 )_ 3 1 Al(NO 3) 3 + Ag= 3 O= 9 N= 3 Al=1 Br=3 Cations/Anions -1 (Ag+1 )_( 1 1 Br )_ 3 Ag. Br
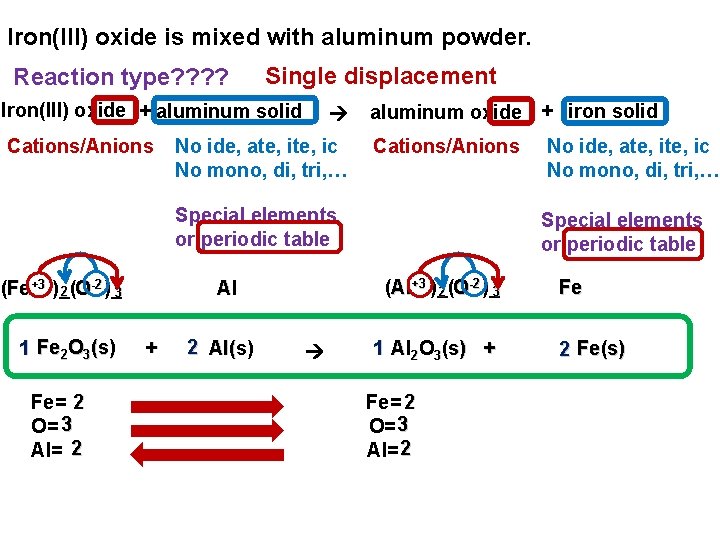
Iron(III) oxide is mixed with aluminum powder. Reaction type? ? Single displacement Iron(III) oxide + aluminum solid Cations/Anions No ide, ate, ic No mono, di, tri, … aluminum oxide + iron solid Cations/Anions Special elements or periodic table -2 (Fe+3 )_( 2 O )_ 3 1 Fe 2 O 3(s) Fe= 2 O= 3 Al= 2 + 2 Al(s) Special elements or periodic table -2 (Al+3 )_( 2 O )_ 3 Al No ide, ate, ic No mono, di, tri, … 1 Al 2 O 3(s) + Fe= 2 O= 3 Al= 2 Fe(s)
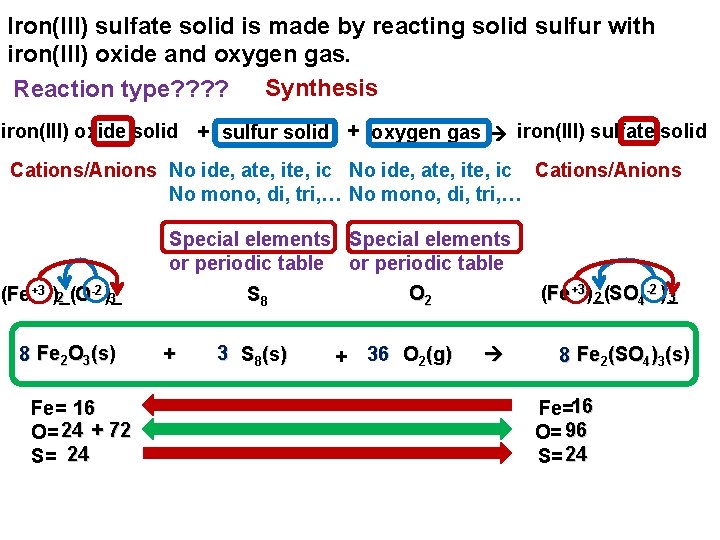
Iron(III) sulfate solid is made by reacting solid sulfur with iron(III) oxide and oxygen gas. Synthesis Reaction type? ? iron(III) oxide solid + sulfur solid + oxygen gas iron(III) sulfate solid Cations/Anions No ide, ate, ite, ic Cations/Anions No mono, di, tri, … Special elements or periodic table -2 (Fe+3 )_( 2 O )_ 3 8 Fe 2 O 3(s) Fe= 16 O= 24 + 72 S= 24 + 3 S 8(s) -2 (Fe+3 )_( 3 2 SO 4 )_ O 2 S 8 + 36 O 2(g) 8 Fe 2(SO 4)3(s) Fe=16 O= 96 S= 24
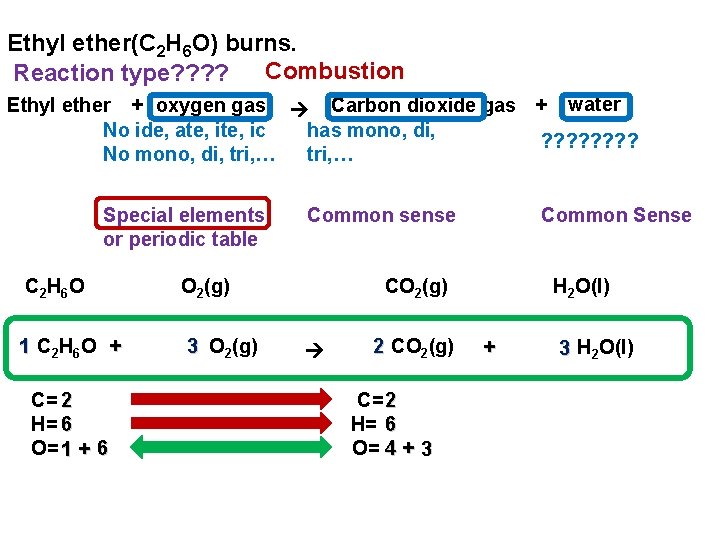
Ethyl ether(C 2 H 6 O) burns. Combustion Reaction type? ? Ethyl ether + oxygen gas Carbon dioxide gas + water No ide, ate, ic has mono, di, ? ? ? ? No mono, di, tri, … Special elements or periodic table C 2 H 6 O O 2(g) 1 C 2 H 6 O + 3 O 2(g) C= 2 H= 6 O= 1 + 6 Common sense Common Sense CO 2(g) 2 CO 2(g) C= 2 H= 6 O= 4 + 3 H 2 O(l)

Anthracene burns. anthracene + oxygen gas Carbon dioxide gas + water C 14 H 10 1 C 14 H 10 CO 2(g) + 16. 5 O 2(g) C= 14 H= 10 O= 33 2 C 14 H 10 14 CO 2(g) + H 2 O(l) 5 H 2 O(l) C= 14 H= 10 O= 28 +5 + 33 O 2(g) 28 CO 2(g) + 10 H 2 O(l)

Reaction Type…. ? Mg(s) + 2 HCl(aq) Mg. Cl 2(aq)+ H 2(g) Single Displacement 4 Fe(c) + 3 O 2(g) 2 Fe 2 O 3(c) Synthesis 2 Ag 2 O(c) 4 Ag(c) + O 2(g) Decomposition Ca(OH)2(aq)+ H 2 SO 4(aq) Ca. SO 4(c)+ 2 H 2 O(l) Double Displacement Cu(c) + 2 Ag. NO 3(aq) Cu(NO 3)2(aq) + 2 Ag(c) Single Displacement Mg(OH)2(aq) + H 3 PO 4(aq) H 2 O(l) + Mg 3(PO 4)2(aq) Double Displacement Zn(s)+ Ag. NO 3(aq) Zn(NO 3)2(aq) + Ag(s) Single Displacement HNO 3(aq)+ Ni (s) Ni(NO 3)2(aq)+ H 2(g) Single Displacement C 5 H 12(l)+ O 2(g) CO 2(g)+ H 2 O(l) Combustion

Review Lab and Quiz Notes Pages 325 -330
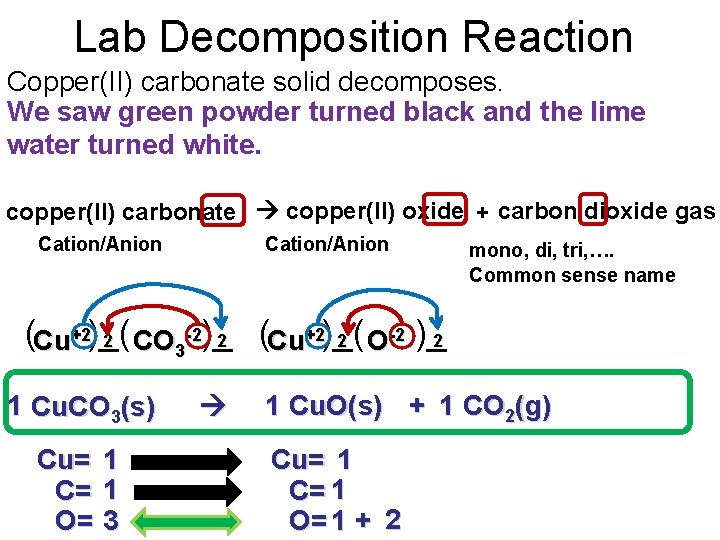
Lab Decomposition Reaction Copper(II) carbonate solid decomposes. We saw green powder turned black and the lime water turned white. copper(II) carbonate copper(II) oxide + carbon dioxide gas Cation/Anion mono, di, tri, …. Common sense name +2)_( (Cu+2)_( ( 2 2 2 CO -2)_ 2 O-2 )_ Cu 3 1 Cu. CO 3(s) Cu= 1 C= 1 O= 3 1 Cu. O(s) + 1 CO 2(g) Cu= 1 C= 1 O= 1 + 2
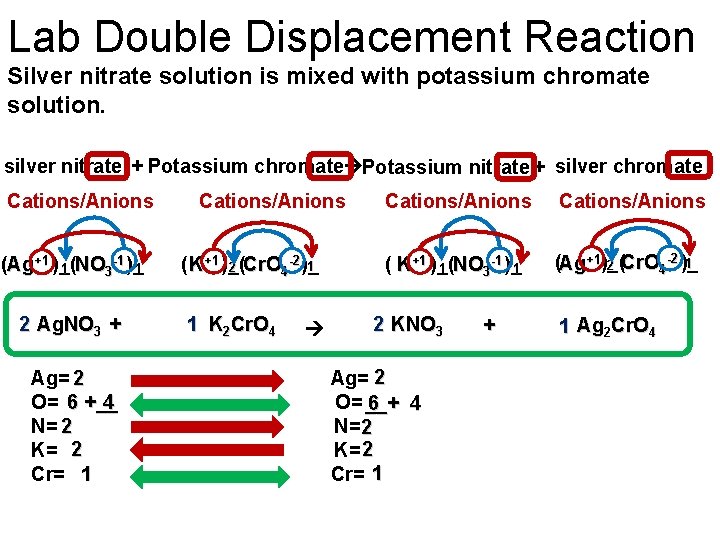
Lab Double Displacement Reaction Silver nitrate solution is mixed with potassium chromate solution. silver nitrate + Potassium chromate Potassium nitrate + silver chromate Cations/Anions -1 (Ag+1 )_( 1 1 NO 3 )_ 2 Ag. NO 3 + Ag= 2 4 O= 6 +__ N= 2 K= 2 Cr= 1 Cations/Anions -2 1 ( K+1 )_( 2 Cr. O 4 )_ 1 K 2 Cr. O 4 Cations/Anions -1 ( K+1 )_( 1 NO 3 )_ 1 -2 1 (Ag+1)_( 2 Cr. O 4 )_ 2 KNO 3 Ag= 2 O= __+ 6 4 N= 2 K= 2 Cr= 1 + 1 Ag 2 Cr. O 4
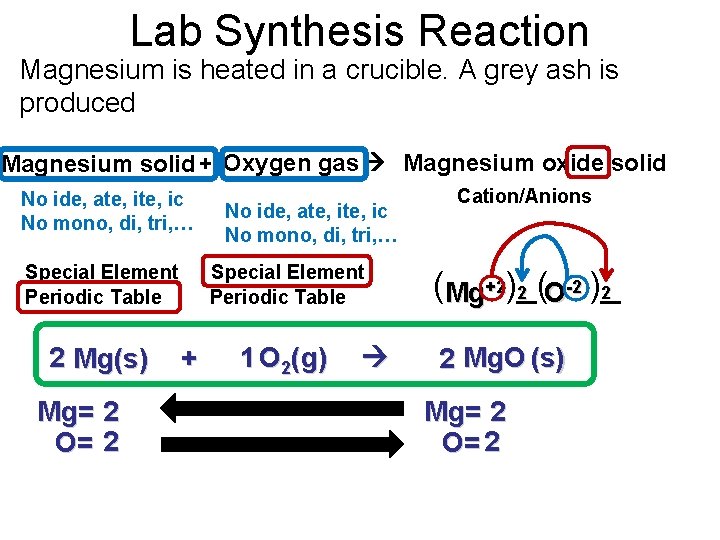
Lab Synthesis Reaction Magnesium is heated in a crucible. A grey ash is produced Magnesium solid + Oxygen gas Magnesium oxide solid No ide, ate, ic No mono, di, tri, … Special Element Periodic Table 2 Mg(s) Mg= 2 O= 2 No ide, ate, ic No mono, di, tri, … Special Element Periodic Table + 1 O 2(g) Cation/Anions (Mg+2)_( 2 2 O-2 )_ 2 Mg. O (s) Mg= 2 O= 2
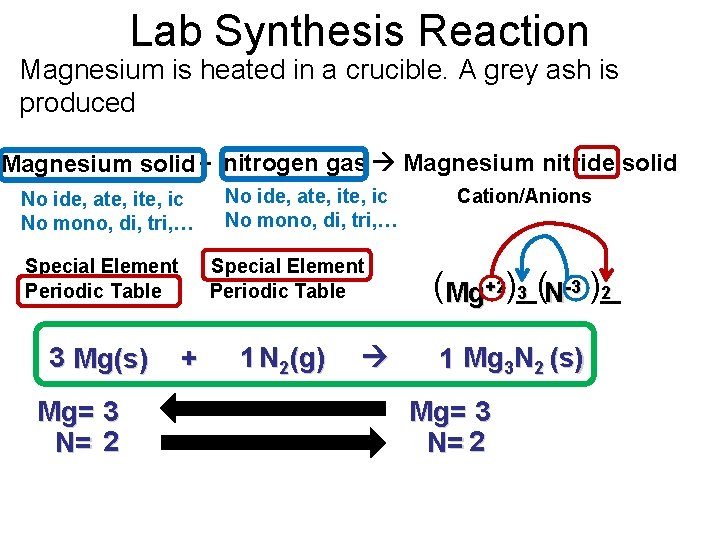
Lab Synthesis Reaction Magnesium is heated in a crucible. A grey ash is produced Magnesium solid + nitrogen gas Magnesium nitride solid No ide, ate, ic No mono, di, tri, … Special Element Periodic Table 3 Mg(s) Mg= 3 N= 2 No ide, ate, ic No mono, di, tri, … Special Element Periodic Table + 1 N 2(g) Cation/Anions (Mg+2)_( 2 3 N-3 )_ 1 Mg 3 N 2 (s) Mg= 3 N= 2
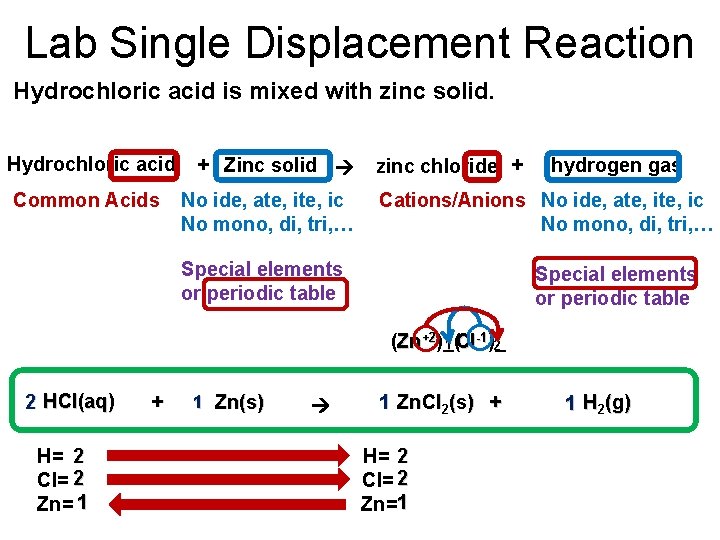
Lab Single Displacement Reaction Hydrochloric acid is mixed with zinc solid. Hydrochloric acid Common Acids + Zinc solid No ide, ate, ic No mono, di, tri, … zinc chloride + hydrogen gas Cations/Anions No ide, ate, ic No mono, di, tri, … Special elements or periodic table -1 (Zn+2)_( 1 Cl )_ 2 2 HCl(aq) H= 2 Cl= 2 Zn= 1 + 1 Zn(s) 1 Zn. Cl 2(s) + H= 2 Cl= 2 Zn=1 1 H 2(g)

CONCLUSIONS AND QUESTIONS: 1. Why did the mass increase during the burning of the magnesium in part A? 2. What is the difference between a physical reaction and a chemical reaction?
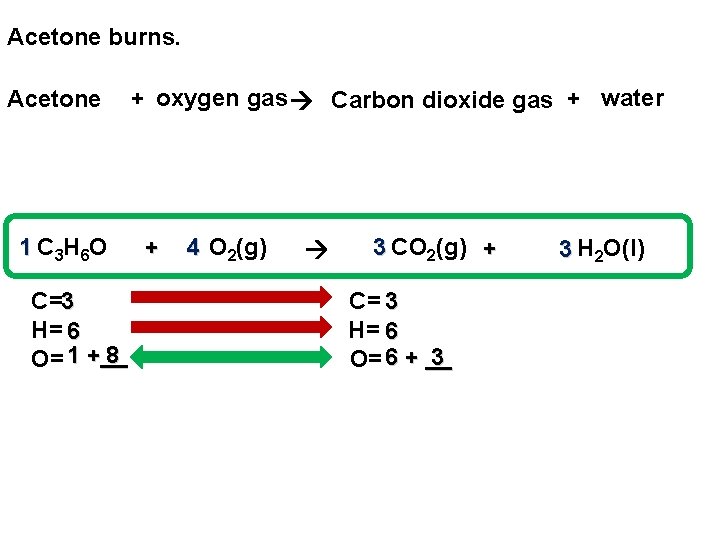
Acetone burns. Acetone 1 C 3 H 6 O C=3 H= 6 8 O= 1 +__ + oxygen gas Carbon dioxide gas + water + 4 O 2(g) 3 CO 2(g) + C= 3 H= 6 3 O= 6 + __ 3 H 2 O(l)

Reaction Types Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2(g) 4 Fe(c) + 3 O 2(g) 2 Fe 2 O 3(c) 2 Ag 2 O(c) 4 Ag(c) + O 2(g) Ca(OH)2(aq) + H 2 SO 4(aq) Ca. SO 4(c) + 2 H 2 O(l) Cu(c)+ 2 Ag. NO 3(aq) Cu(NO 3)2(aq)+ 2 Ag(c) C 5 H 12(l) + 8 O 2(g) 5 CO 2(g) + 6 H 2 O(l) Ba. CO 3(s) + C(s) + H 2 O(l) 2 CO(g) + Ba(OH)2(s)
- Slides: 29