Notes Ionic Bonds 1 Key Concept Ionic bonds

Notes: Ionic Bonds 1. Key Concept: Ionic bonds form when electrons are transferred _____ from one atom to another atom. ØLike all chemical bonds, ionic bonds form so that the outermost ______ energy _____ levels of ____ the atoms in the bonds are filled. ØIn an ionic bond, one atom has lost electrons. ____ gained Ø The other atom has ______ electrons.
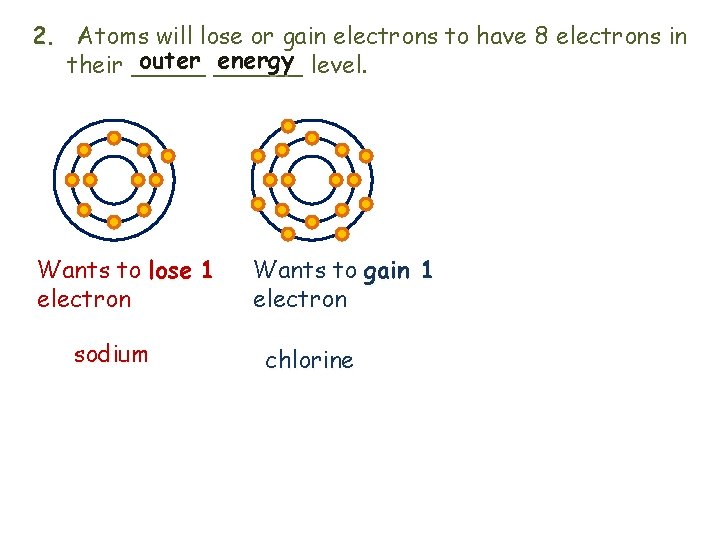
2. Atoms will lose or gain electrons to have 8 electrons in outer ______ energy level. their _____ Wants to lose 1 electron sodium Wants to gain 1 electron chlorine
REMEMBER, This is a happy atom!
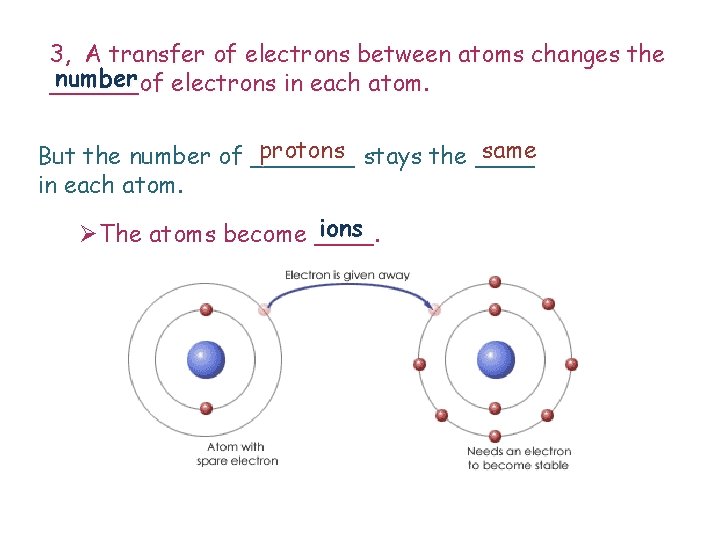
3, A transfer of electrons between atoms changes the number electrons in each atom. ______of protons stays the ____ same But the number of _______ in each atom. ions ØThe atoms become ____.
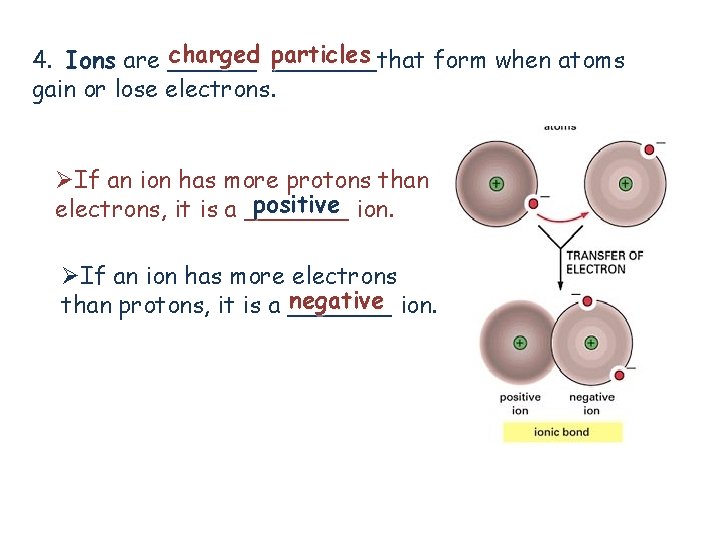
charged particles 4. Ions are _______that form when atoms gain or lose electrons. ØIf an ion has more protons than positive ion. electrons, it is a _______ ØIf an ion has more electrons negative ion. than protons, it is a _______
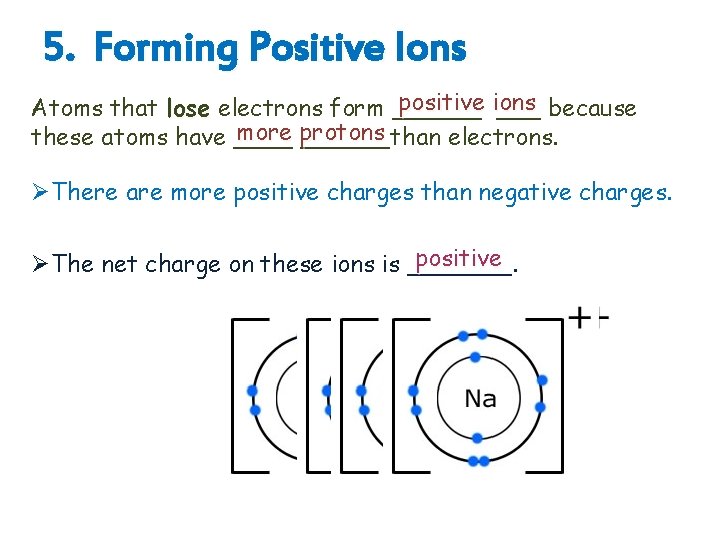
5. Forming Positive Ions positive ions Atoms that lose electrons form ______ because more protons these atoms have ______than electrons. ØThere are more positive charges than negative charges. positive ØThe net charge on these ions is _______.
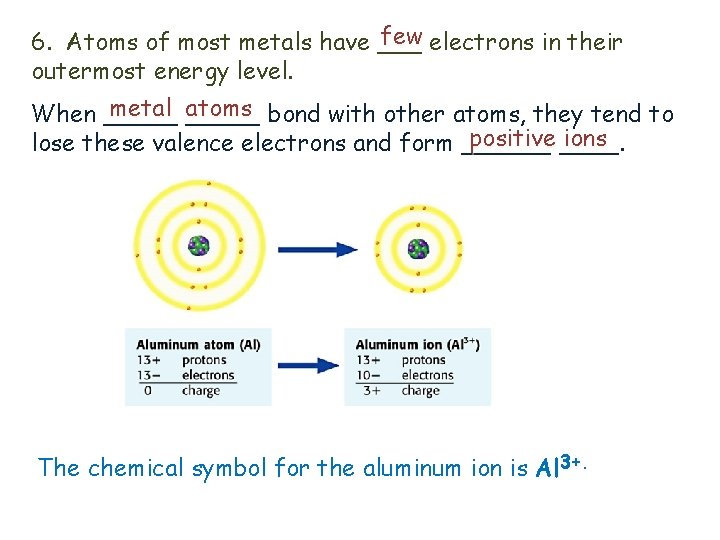
few electrons in their 6. Atoms of most metals have ___ outermost energy level. metal _____ atoms bond with other atoms, they tend to When _____ positive ____. ions lose these valence electrons and form ______ The chemical symbol for the aluminum ion is Al 3+.
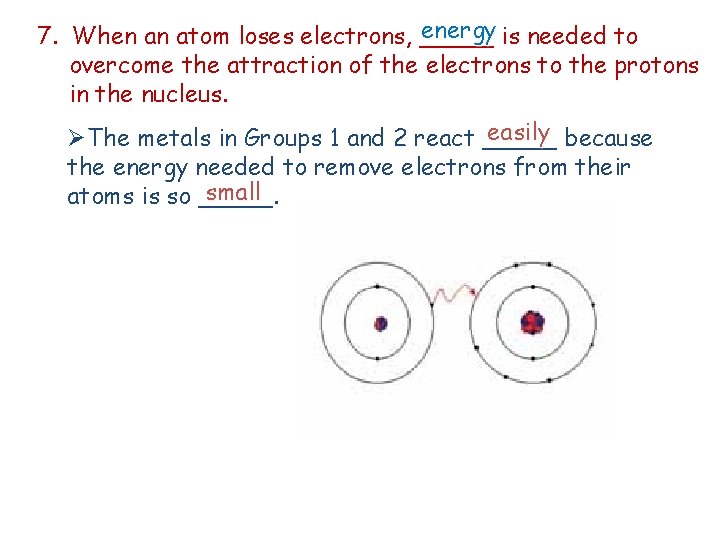
energy is needed to 7. When an atom loses electrons, _____ overcome the attraction of the electrons to the protons in the nucleus. easily because ØThe metals in Groups 1 and 2 react _____ the energy needed to remove electrons from their small atoms is so _____.

8. Forming Negative Ions negative ___ ions because Atoms that gain electrons form ______ more electrons these atoms have _______ than protons. ØThere are more negative charges than positive charges. negative ØThe net charge on these ions is _______.
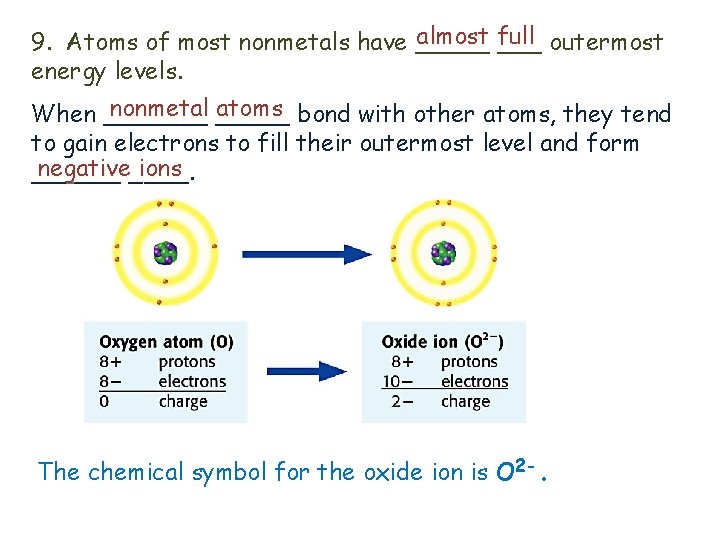
almost full 9. Atoms of most nonmetals have _____ outermost energy levels. nonmetal _____ atoms bond with other atoms, they tend When _______ to gain electrons to fill their outermost level and form negative____. ions ______ The chemical symbol for the oxide ion is O 2 -.

fill their outermost energy 10. Atoms of most nonmetals ___ gaining _______. electrons level by ______ given ___ off by most nonmetal atoms when ØEnergy is _____ gain electrons. they ____ more easily ØThe _____ an atom gains an electron, the more _____ energy the atom releases. ____ halogens such as fluorine and chlorine, are ØThe _______, very reactive because they release _______ amount of energy. a large ______

11. Forming Ionic Compounds ionic ____ bond forms because of the strong forces of An ____ attraction between the ______ positive metal ions and the ____ negative nonmetal ions _______ ___. opposite charges The _______of the ions causes the ions to ____ together _______. stick
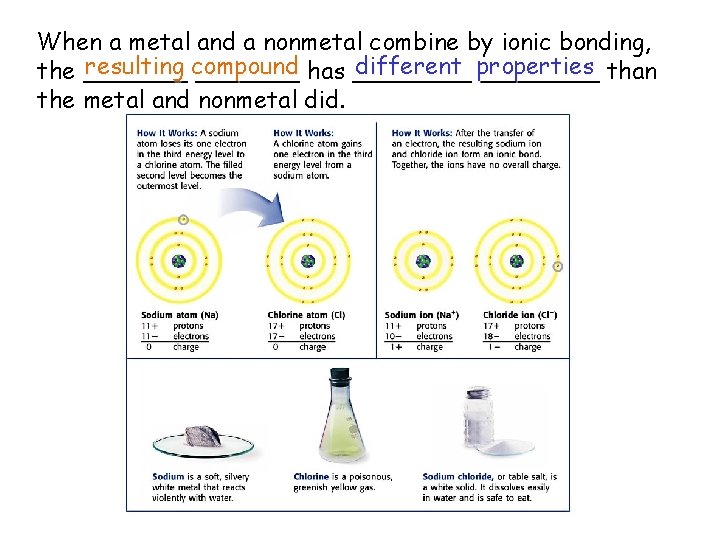
When a metal and a nonmetal combine by ionic bonding, resulting compound different properties the _______ has ________ than the metal and nonmetal did.
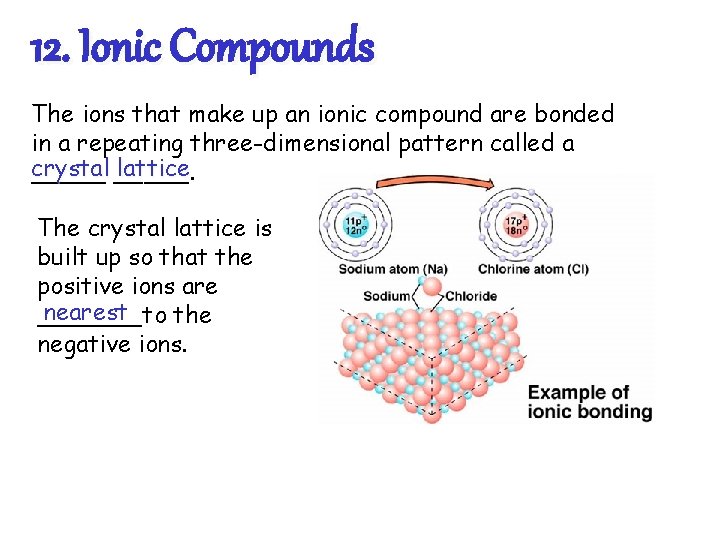
12. Ionic Compounds The ions that make up an ionic compound are bonded in a repeating three-dimensional pattern called a crystal lattice _____. The crystal lattice is built up so that the positive ions are nearest _______to the negative ions.

13. Properties of Ionic Compounds Ø brittle solids at room temperature • break apart when hit with a hammer Ø high melting points • magnesium oxide melts at 2, 800ºC Ø very high boiling points Ø high solubility in water • sea water tastes salty because it has sodium chloride and many other ionic compounds dissolved in it
- Slides: 15