Notes Chapter 2 3 Carbon Compounds Chemistry of

Notes: Chapter 2. 3 Carbon Compounds
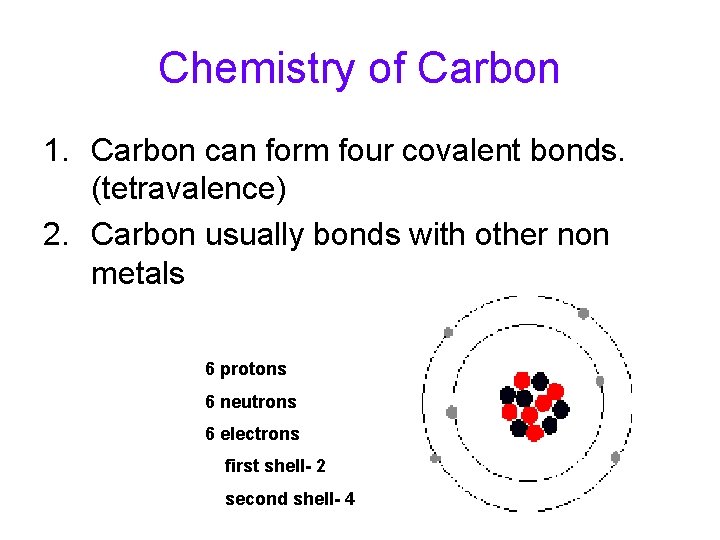
Chemistry of Carbon 1. Carbon can form four covalent bonds. (tetravalence) 2. Carbon usually bonds with other non metals 6 protons 6 neutrons 6 electrons first shell- 2 second shell- 4
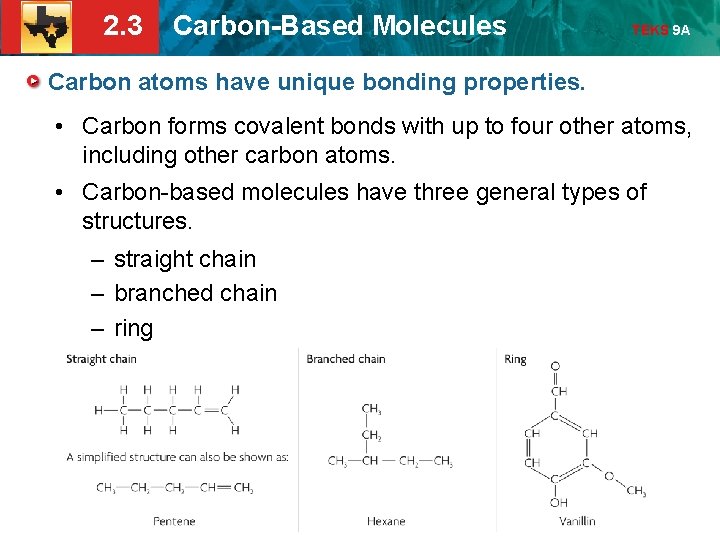
2. 3 Carbon-Based Molecules TEKS 9 A Carbon atoms have unique bonding properties. • Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. • Carbon-based molecules have three general types of structures. – straight chain – branched chain – ring
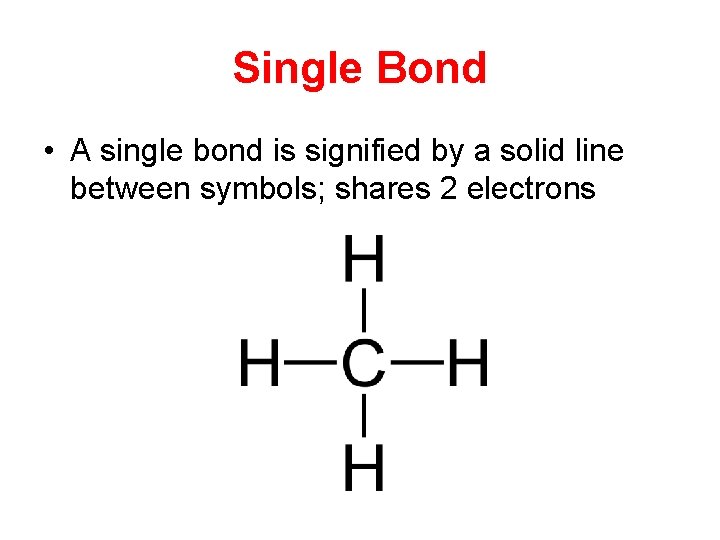
Single Bond • A single bond is signified by a solid line between symbols; shares 2 electrons
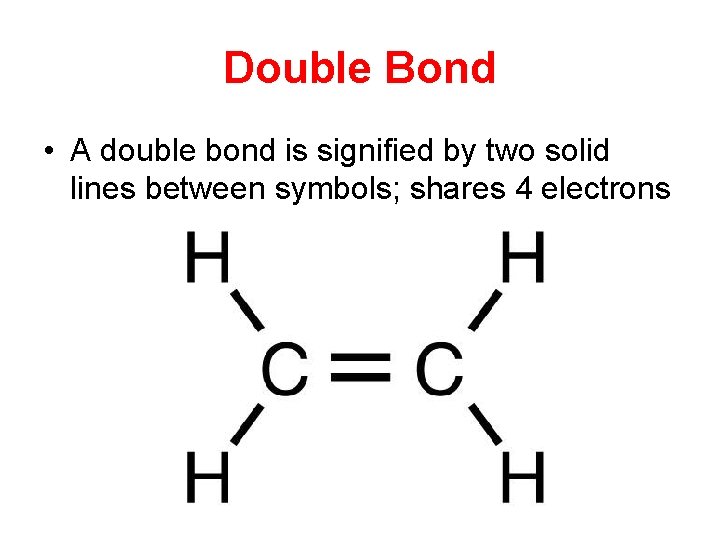
Double Bond • A double bond is signified by two solid lines between symbols; shares 4 electrons
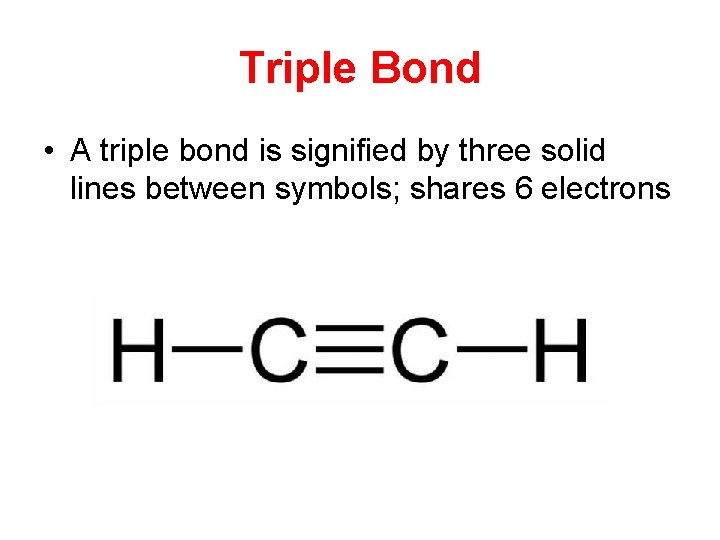
Triple Bond • A triple bond is signified by three solid lines between symbols; shares 6 electrons

Macromolecules 1. Macromolecules are “Giant molecules” 2. Consist of monomers (smaller units) that join to form polymers (large molecules of repeating units – monomers- bonded together by covalent bonds.
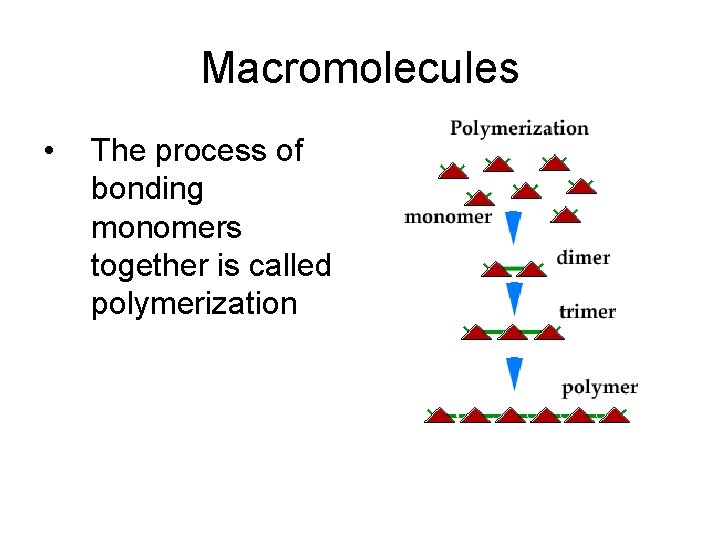
Macromolecules • The process of bonding monomers together is called polymerization
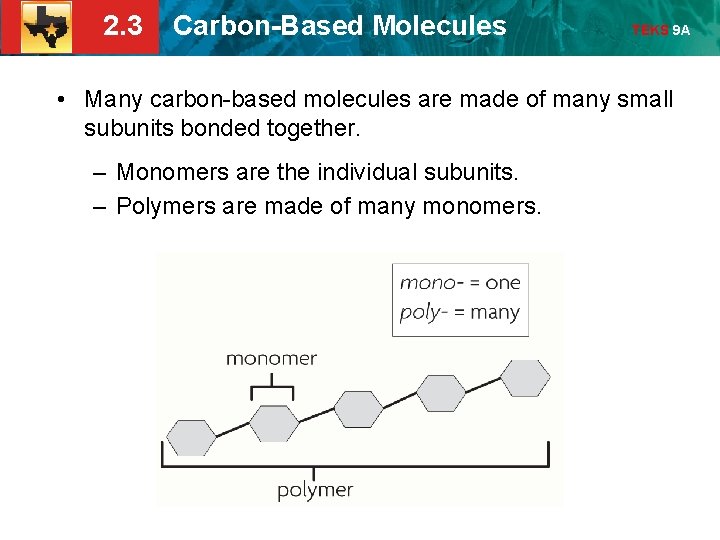
2. 3 Carbon-Based Molecules TEKS 9 A • Many carbon-based molecules are made of many small subunits bonded together. – Monomers are the individual subunits. – Polymers are made of many monomers.
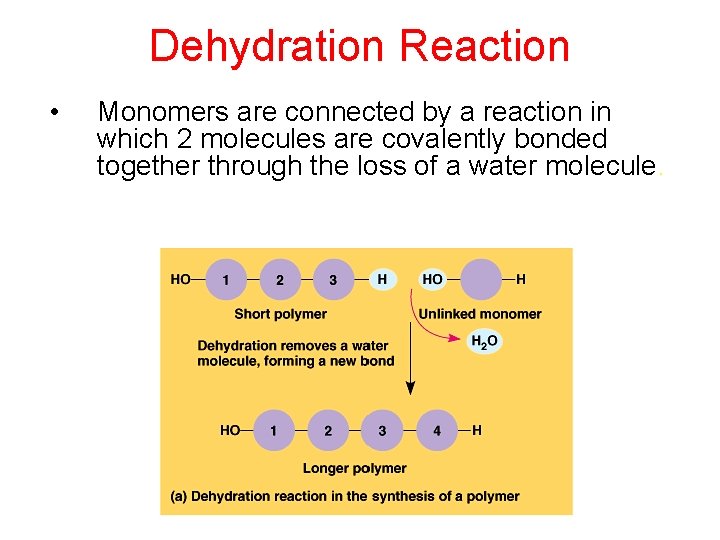
Dehydration Reaction • Monomers are connected by a reaction in which 2 molecules are covalently bonded together through the loss of a water molecule.
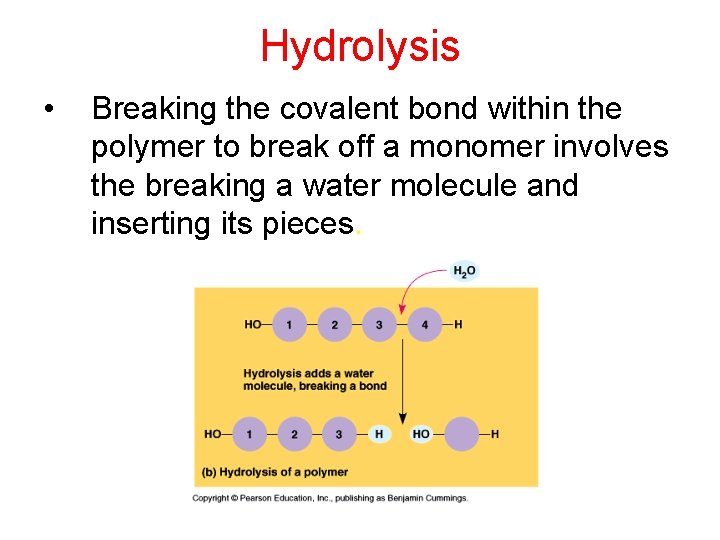
Hydrolysis • Breaking the covalent bond within the polymer to break off a monomer involves the breaking a water molecule and inserting its pieces.

Types of macromolecules 1. 2. 3. 4. Carbohydrates Lipids Nucleic Acids Proteins

Carbohydrates 1. Composition: made of C, H and O atoms, usually in a 1: 2: 1 ratio 2. Monomer- monosaccharide 3. Polymer – polysaccharide 4. Uses: Main source of energy for organisms, structural purpose in cell membrane, and exoskeleton of insects.
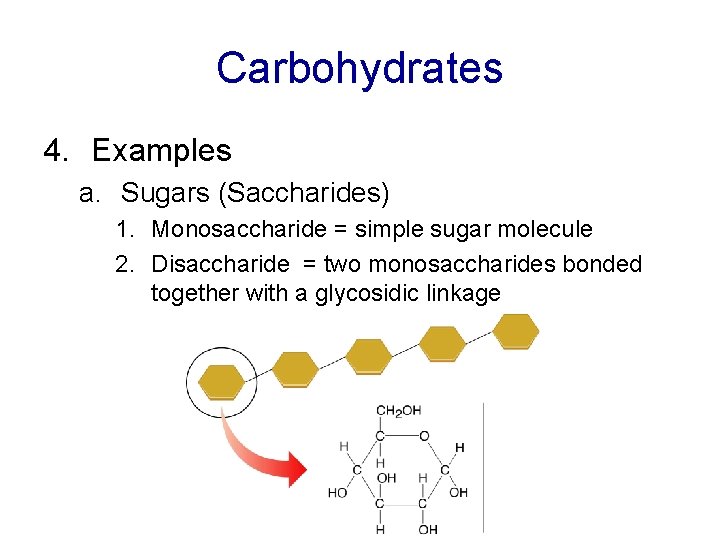
Carbohydrates 4. Examples a. Sugars (Saccharides) 1. Monosaccharide = simple sugar molecule 2. Disaccharide = two monosaccharides bonded together with a glycosidic linkage

Carbohydrates b. Starches: Complex carbohydrates (polysaccharides) that store extra sugar 1. In animals, starch is called glycogen, structural is called chitin o Chitin is used by arthropods to build their exoskeletons.

Carbohydrates 2. In plants, “plant starch”, structural is called cellulose o Cellulose makes up the cell wall of plant cells. o Humans cannot breakdown cellulose during digestion. o Known as “insoluble fiber”
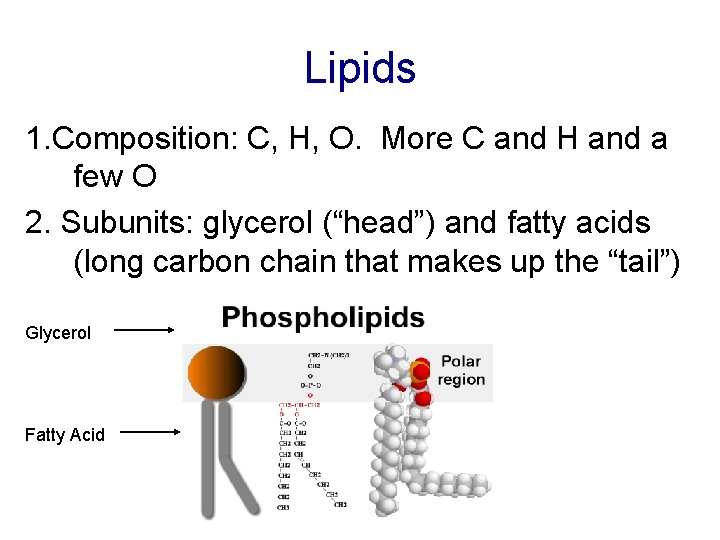
Lipids 1. Composition: C, H, O. More C and H and a few O 2. Subunits: glycerol (“head”) and fatty acids (long carbon chain that makes up the “tail”) Glycerol Fatty Acid
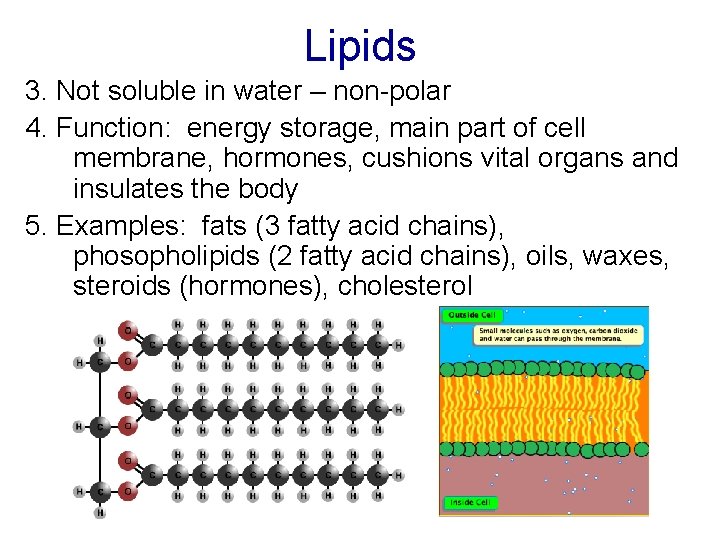
Lipids 3. Not soluble in water – non-polar 4. Function: energy storage, main part of cell membrane, hormones, cushions vital organs and insulates the body 5. Examples: fats (3 fatty acid chains), phosopholipids (2 fatty acid chains), oils, waxes, steroids (hormones), cholesterol
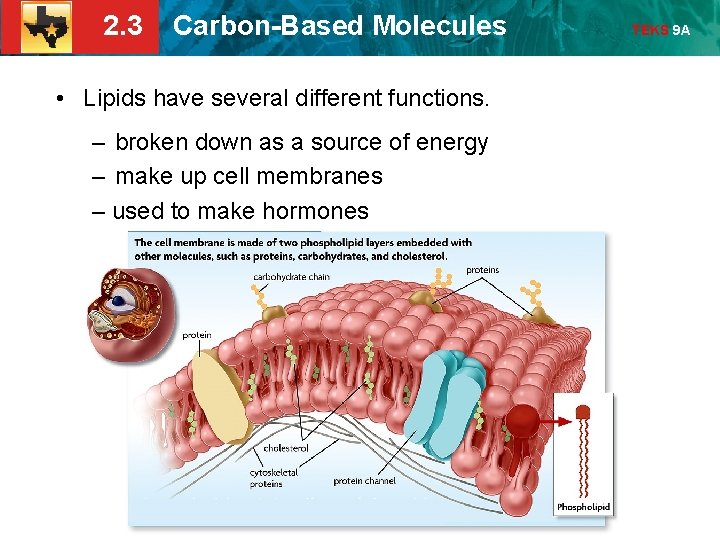
2. 3 Carbon-Based Molecules • Lipids have several different functions. – broken down as a source of energy – make up cell membranes – used to make hormones TEKS 9 A

Lipids 6. Types of fatty acids: a. Unsaturated fatty acids are found in lipids that are liquid at room temperature, C=C bonds • • The double bond creates a kink in the tails that keeps them from packing closely together. Example: Olive oil Double bond

Lipids b. Saturated fatty acids are found in lipids that are solids at room temperature, no C=C bonds • Example: Shortening, butter

Types of Lipids Saturated = only single bonds, maximum number of H atoms Unsaturated = at least on C = C double bond lipid
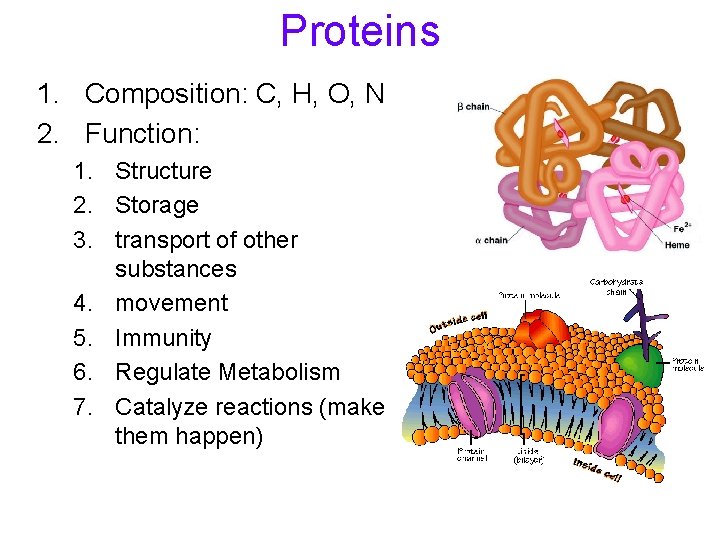
Proteins 1. Composition: C, H, O, N 2. Function: 1. Structure 2. Storage 3. transport of other substances 4. movement 5. Immunity 6. Regulate Metabolism 7. Catalyze reactions (make them happen)
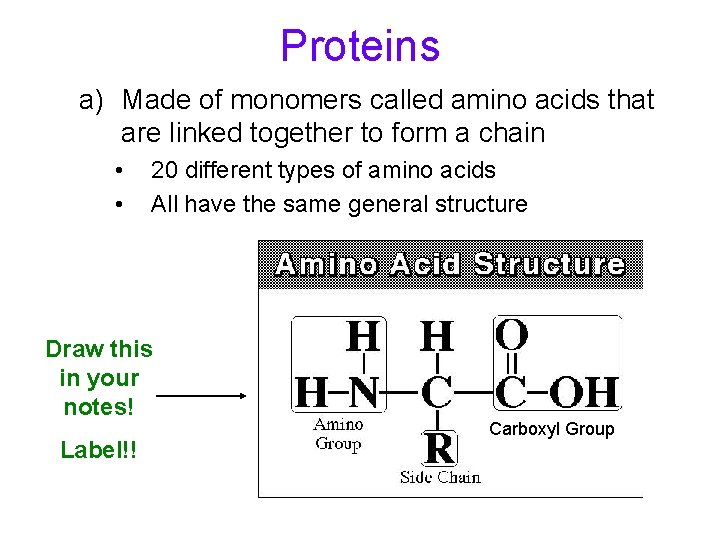
Proteins a) Made of monomers called amino acids that are linked together to form a chain • • 20 different types of amino acids All have the same general structure Draw this in your notes! Label!! Carboxyl Group
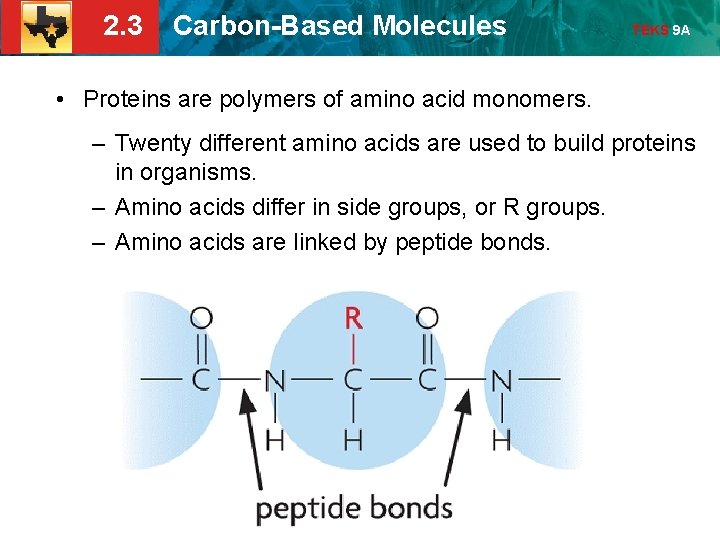
2. 3 Carbon-Based Molecules TEKS 9 A • Proteins are polymers of amino acid monomers. – Twenty different amino acids are used to build proteins in organisms. – Amino acids differ in side groups, or R groups. – Amino acids are linked by peptide bonds.
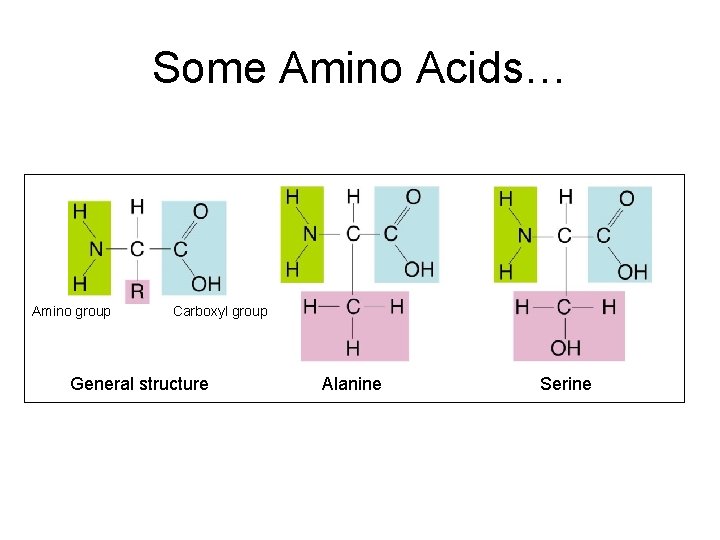
Some Amino Acids… Amino group Carboxyl group General structure Alanine Serine
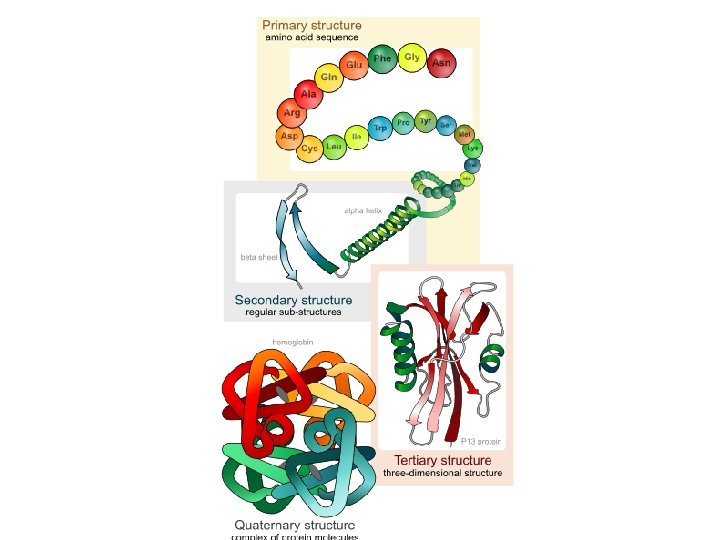
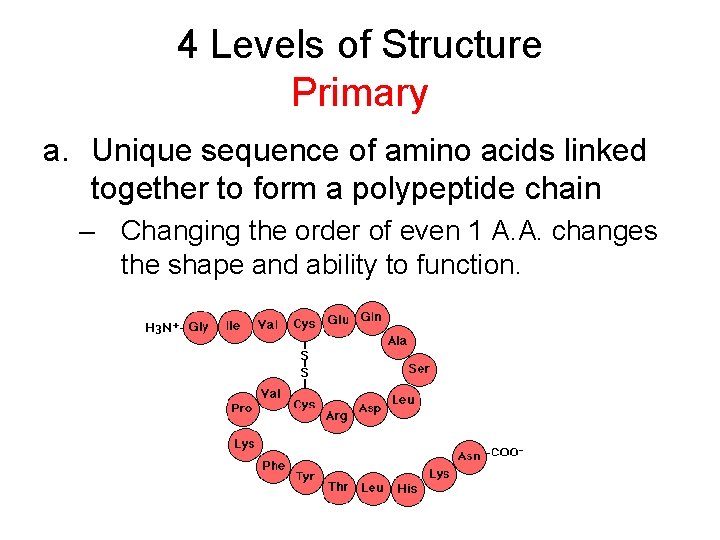
4 Levels of Structure Primary a. Unique sequence of amino acids linked together to form a polypeptide chain – Changing the order of even 1 A. A. changes the shape and ability to function.
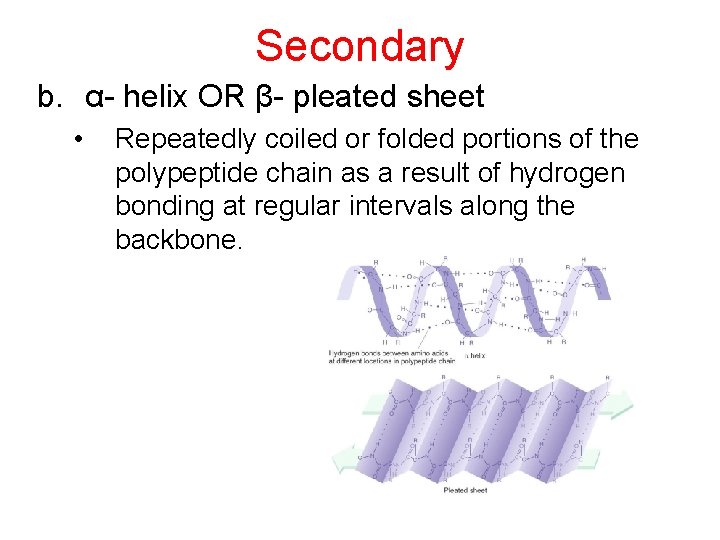
Secondary b. α- helix OR β- pleated sheet • Repeatedly coiled or folded portions of the polypeptide chain as a result of hydrogen bonding at regular intervals along the backbone.
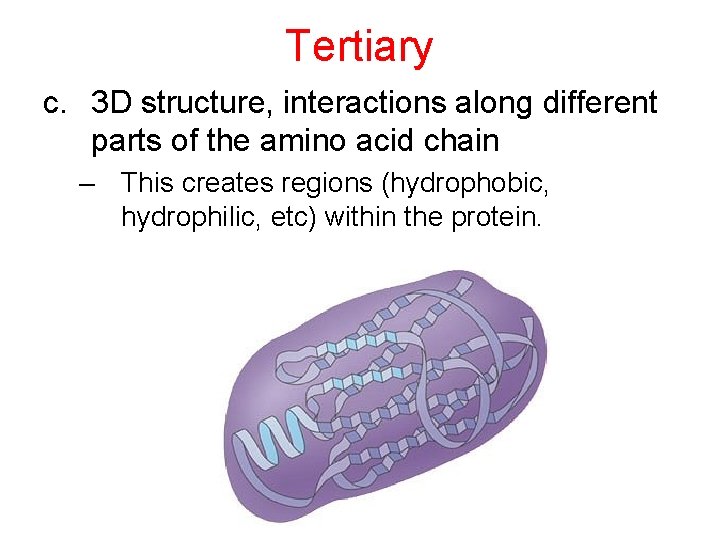
Tertiary c. 3 D structure, interactions along different parts of the amino acid chain – This creates regions (hydrophobic, hydrophilic, etc) within the protein.
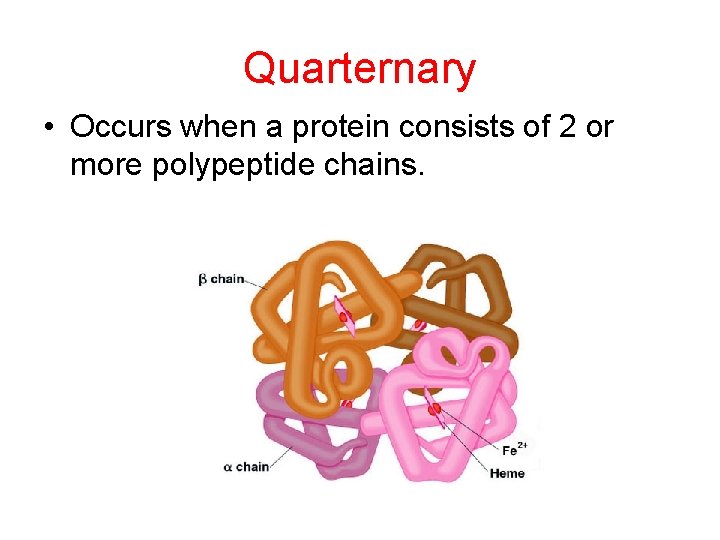
Quarternary • Occurs when a protein consists of 2 or more polypeptide chains.
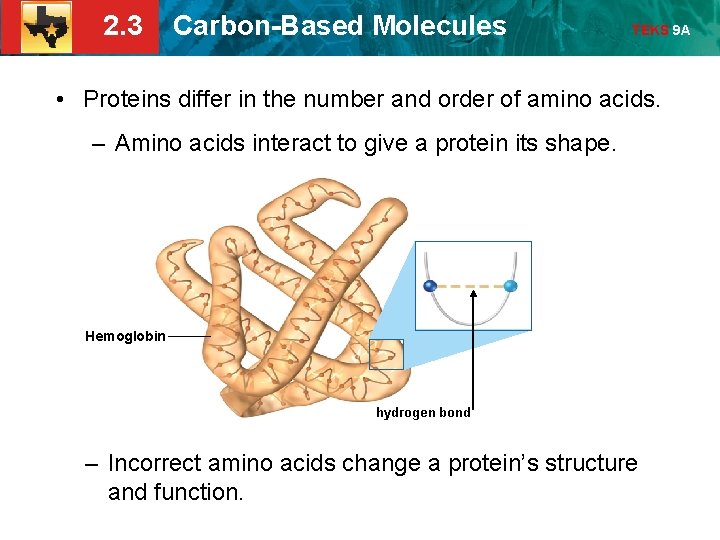
2. 3 Carbon-Based Molecules TEKS 9 A • Proteins differ in the number and order of amino acids. – Amino acids interact to give a protein its shape. Hemoglobin hydrogen bond – Incorrect amino acids change a protein’s structure and function.
Nucleic Acids 1. Composition: C, H, O, N, P 2. Function: Store and transmit genetic information by determining the amino acid sequence in proteins 3. Two types a. DNA (deoxyribonucleic acid) b. RNA (ribonucleic acid)
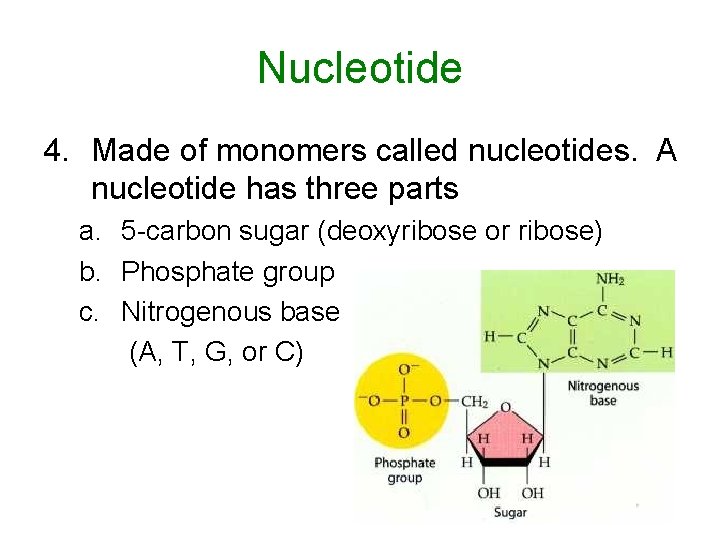
Nucleotide 4. Made of monomers called nucleotides. A nucleotide has three parts a. 5 -carbon sugar (deoxyribose or ribose) b. Phosphate group c. Nitrogenous base (A, T, G, or C)
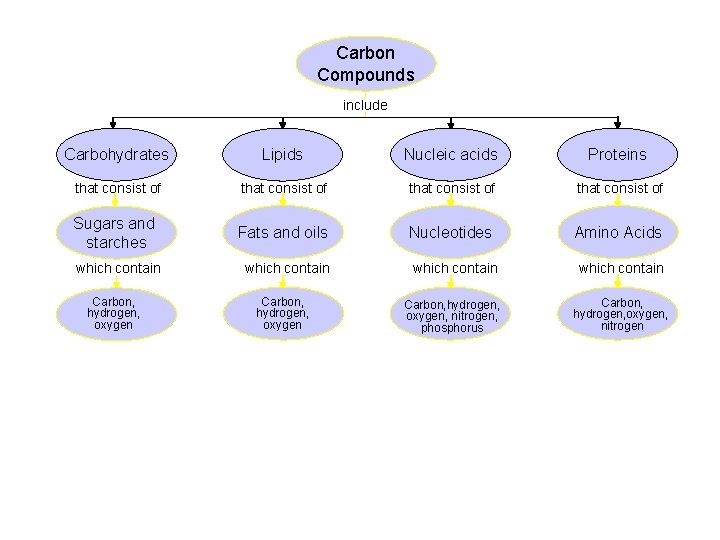
Carbon Compounds include Carbohydrates Lipids Nucleic acids Proteins that consist of Sugars and starches Fats and oils Nucleotides Amino Acids which contain Carbon, hydrogen, oxygen, nitrogen, phosphorus Carbon, hydrogen, oxygen, nitrogen
- Slides: 35