Notes Acids and Bases Acids taste sour Note

Notes: Acids and Bases

Acids • taste sour – Note: NEVER taste an acid in class! • conduct electricity • react with indicators to produce predictable changes in color • corrode metals (HCl Acid corrodes Mg ribbon)

Common Acids • • Sulfuric Acid Hydrochloric Acid Nitric Acid Citric Acid

Bases • Bitter taste • In solution, feel slippery (like soap) • corrode metals • conduct electricity • React with indicators

Common Bases • Aluminum Hydroxide • Calcium Hydroxide • Ammonia

What is the p. H scale? • 1 -6 for an Acid (1 is the strongest acid) • 7 - Neutral (water is a neutral substance) • 8 -14 for bases (14 is the strongest base).
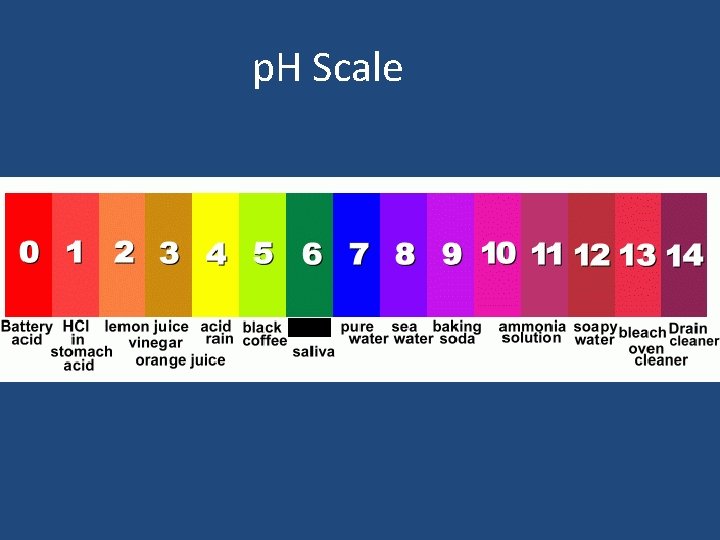
p. H Scale

Vocabulary • Indicators such as litmus, p. H paper, and cabbage juice are used to detect the presence of acids and bases by changes in color. • Reactivity is the tendency of a substance to undergo chemical reaction, either by itself or with other materials. • Combustibility, or flammability, means that a substance is capable of igniting and burning.

What is a neutralization reaction? • Neutralization: a chemical reaction between an acid and a base which produces water and salt • During a neutralization reaction… – hydronium ion, H+ (acid) + hydroxide ion, OH- (base) = water – negative ions (acid) + positive ions (base) = salt Acid + Base = Water + Salt
- Slides: 9