Note to teachers This presentation is made available

Note to teachers ► This presentation is made available for use in your classroom. ► At mini. PCR, we are committed to providing an engaging classroom environment for your students and provide this material to help you achieve that goal. Please feel free to modify this presentation for your needs as you feel necessary. ► Along with many of these slides are notes with added information and classroom tips. ► This presentation remains the property of mini. PCR. Please do not redistribute. © 2017 by Amplyus, all rights reserved

blue. Gel TM Electrophoresis Rainbow Lab Release v 4. 1: October 2016 © 2017 by Amplyus, all rights reserved

Goals for today’s lab: ► Separate molecules based on size and charge-to-mass ratio using gel electrophoresis. ► Separate molecules based on polarity using paper chromatography. ► Learn to use a micropipette. ► Learn to use a gel electrophoresis system.
Gel electrophoresis is at the heart of DNA analysis

Gel electrophoresis is a method for separating molecules Gel electrophoresis separates molecules based on 2 main properties: size and charge-to-mass ratio. ► It is most often used with DNA or proteins, but can be used with any charged molecule. ► A scientist views protein separation results on a polyacrylamide gel

Types of gels ► The two main substrates used in gel electrophoresis are agarose and polyacrylamide. ► Polyacrylamide gels are typically used for separating proteins. Polyacrylamide is made of long chains (polymers) of the molecule acrylamide, a known neurotoxin. ► Agarose gels are typically used for separating DNA. Agarose is a polysaccharide extracted from seaweed and is safe to handle. ► The material used to make the two types of gels is different, but how the gel works is similar. Today we will be using agarose gels

Gel Electrophoresis – agarose gels The gel is a matrix, similar to a web-like obstacle course, through which molecules are pulled by electric force. § Smaller molecules have an easier time making it through the matrix, so they move faster. § Larger molecules have more trouble making it through the obstacles, so they migrate slower. § Molecules with a positive charge are pulled in one direction. § Molecules with a negative charge are pulled the opposite way. § The force is driven by an electric field, so a molecule’s charge determines how hardcharge-to-mass it is pulled. A molecule’s ratio determines the speed at which it travels
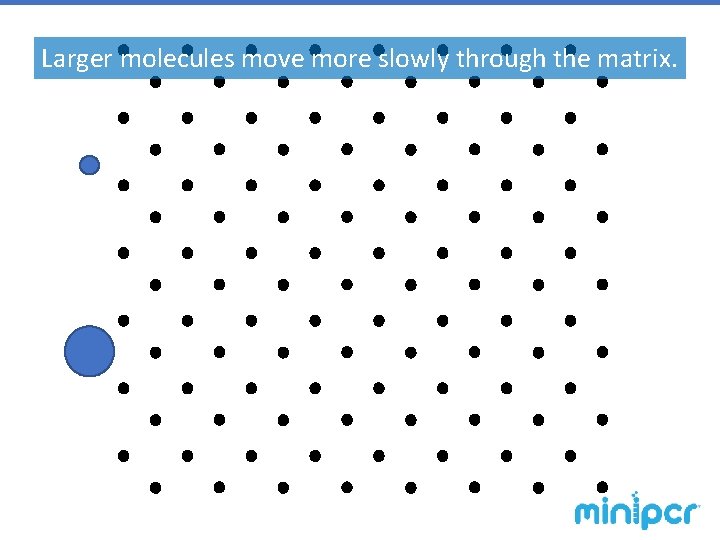
Larger molecules move more slowly through the matrix.
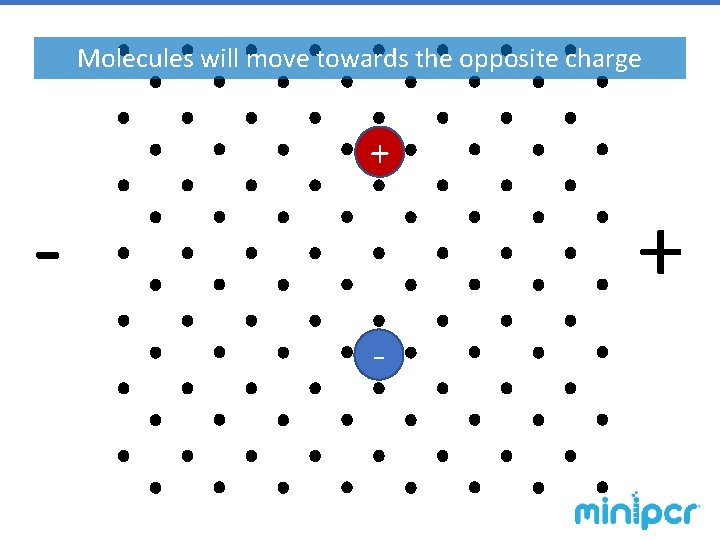
Molecules will move towards the opposite charge + -
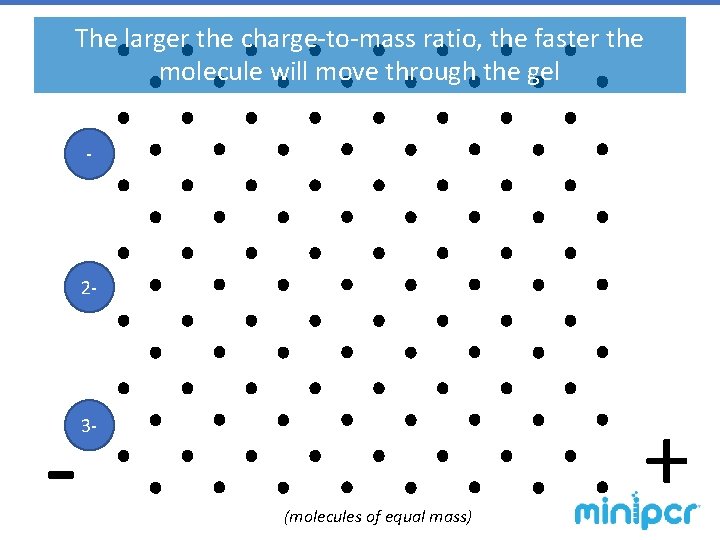
The larger the charge-to-mass ratio, the faster the molecule will move through the gel - 2 - - 3 - (molecules of equal mass) +

Paper chromatography separates molecules based on solubility in a solvent ► Polar molecules are those whose charge is not spread evenly over the molecule. While the molecule as a whole may have no charge, one part of it may be more positive and another part may be more negative. ► Water and ethanol (rubbing alcohol) are polar solvents. ► Molecules that very polar will dissolve easily in them. ► Molecules that are not very polar will not dissolve as easily. Water Ethanol

Polar molecules will travel farther in paper chromatography In paper chromatography, the solvent will travel up the filter paper by capillary action. ► Molecules that are very well dissolved will move easily with the solvent and travel far up the paper. ► Molecules that are not as well dissolved will not move as easily with the solvent and will not travel as far up the paper. ►
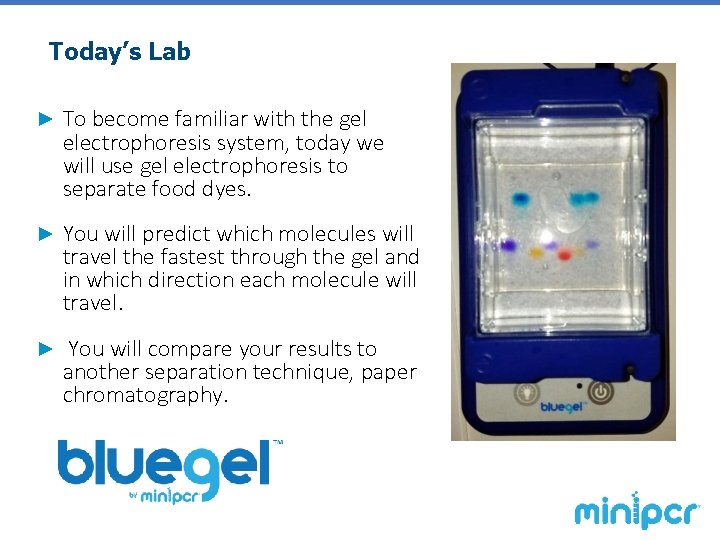
Today’s Lab ► To become familiar with the gel electrophoresis system, today we will use gel electrophoresis to separate food dyes. ► You will predict which molecules will travel the fastest through the gel and in which direction each molecule will travel. ► You will compare your results to another separation technique, paper chromatography.

Like everything, the food you eat is a collection of different molecules. ► Many foods you eat are the color they are because dyes have been added. Dyes are small chemical molecules that are safe.
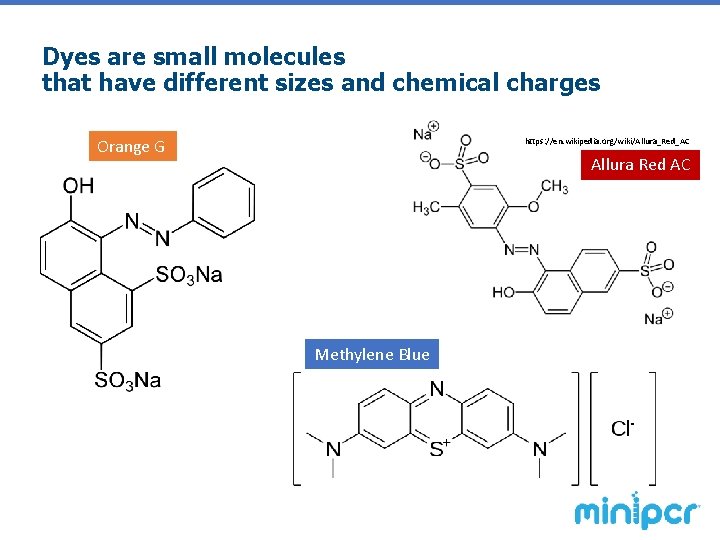
Dyes are small molecules that have different sizes and chemical charges Orange G https: //en. wikipedia. org/wiki/Allura_Red_AC Allura Red AC Methylene Blue

Let’s do it! Micropipetting Gel electrophoresis 45 min total Dye visualization

Warming up: Micropipetting practice

Micropipetting practice Steps to micropipetting 1. Identify volume range 2. Adjust volume 3. Press plunger to FIRST STOP 4. Slowly Release Plunger 5. Now press plunger to SECOND STOP

Now, transfer real liquids: 20, 10, and 5µl 1. Adjust volume 2. Get a tip 3. Press plunger to FIRST STOP 4. Slowly release plunger, collecting liquid 5. Transfer liquid 6. Press plunger to SECOND STOP 7. Remove tip from liquid 8. Release plunger 9. Eject tip

blue. Gel™: integrated electrophoresis AND visualization system Cast 0. 8% agarose gels (instructions are per gel): ► In an Erlenmeyer Flask add: • 25 ml 1 X TBE buffer • 0. 2 g agarose ► Microwave for 30 seconds or until liquid is clear. ► Cool for 1 -2 minutes before pouring. ► Allow gel to cool to harden (about 15 minutes) ► When cool, place the gel and casting tray in the blue. Gel electrophoresis system. Cover with 1 X TBE buffer
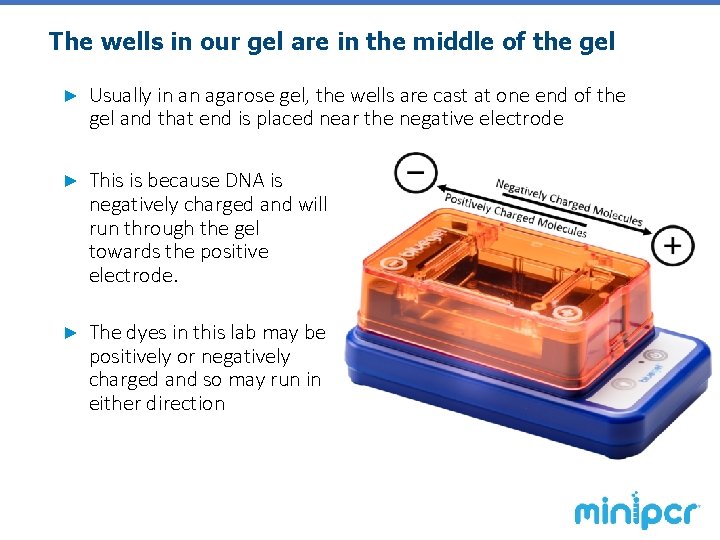
The wells in our gel are in the middle of the gel ► Usually in an agarose gel, the wells are cast at one end of the gel and that end is placed near the negative electrode ► This is because DNA is negatively charged and will run through the gel towards the positive electrode. ► The dyes in this lab may be positively or negatively charged and so may run in either direction

1. Draw up 15 µl of dye using a micropipette 2. Carefully load each dye into the correct well of the gel. Use a new tip each time to avoid contamination
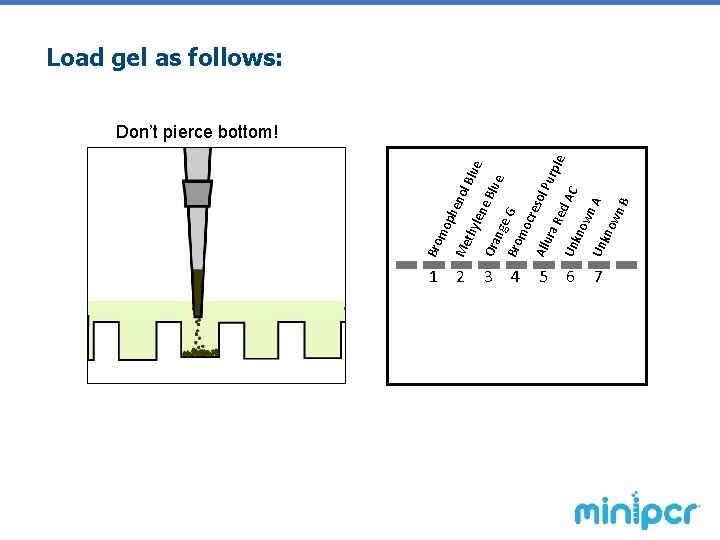
3 4 5 G sol nol Blu e e. B lue Pur ra R ple ed AC Un kno wn A Un kno wn B cre mo Allu nge 2 Bro Ora 1 len thy phe Me mo Bro Load gel as follows: Don’t pierce bottom! 6 7
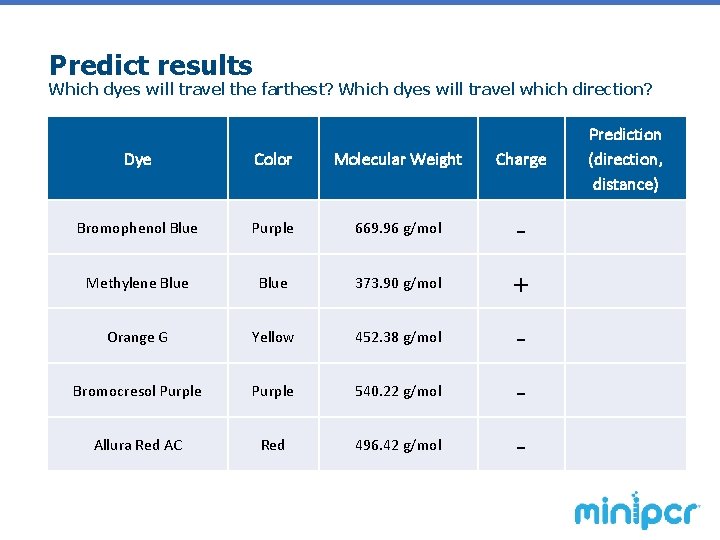
Predict results Which dyes will travel the farthest? Which dyes will travel which direction? Dye Color Molecular Weight Charge H Bromophenol Blue Purple 669. 96 g/mol - Methylene Blue 373. 90 g/mol + Orange G Yellow 452. 38 g/mol - Bromocresol Purple 540. 22 g/mol - Allura Red AC Red 496. 42 g/mol - Prediction (direction, distance)

Wrap up • How did the experiment turn out? • Which dyes ran in each direction? Can you explain to me why? • Which dye traveled the farthest? Which dye traveled the least distance? Can you explain why? • Compare gel electrophoresis and paper chromatography? • What are some ways that paper chromatography and gel electrophoresis are similar? • In what ways are they different from each other? • Can you think of a way that molecules may travel together in one technique, but separate in the other? Tweet out your results! @mini. PCR #Rainbow. Lab


Thank you! www. mini. PCR. com support@minipcr. com Facebook. com/mini. PCR

Appendix: additional resources Understanding gel electrophoresis • http: //www. yourgenome. org/facts/what-is-gel-electrophoresis • https: //www. khanacademy. org/science/biology/biotech-dnatechnology/dna-sequencing-pcr-electrophoresis/a/gel-electrophoresis Understanding chromatography • https: //www. khanacademy. org/test-prep/mcat/chemicalprocesses/separations-purifications/a/principles-of-chromatography • http: //www. chemguide. co. uk/analysis/chromatogrmenu. html History of the use of DNA in crime solving • http: //www. forensicmag. com/articles/2005/01/evolution-dna-evidencecrime-solving-judicial-and-legislative-history

The complete biotech toolkit: DNA Discovery System™ Micropipetting PCR Gel electrophoresis Visualization
- Slides: 29