Norplant Levonorgestrel implantable progesterone only contraception Jadelle Sinoimplant

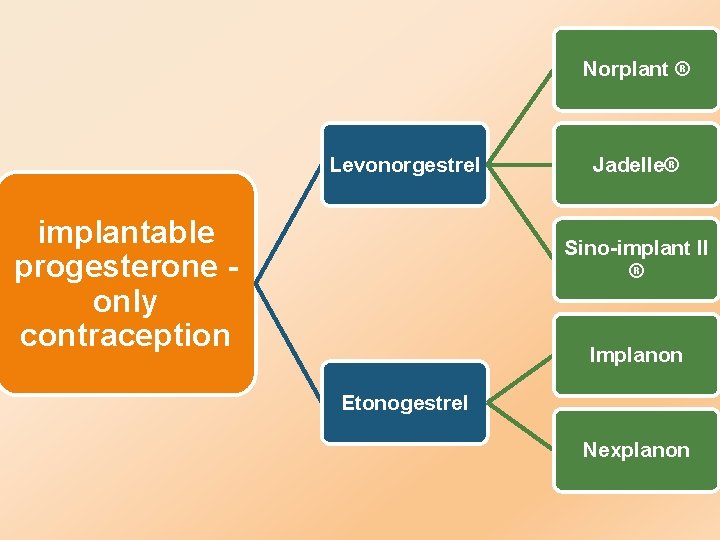
Norplant ® Levonorgestrel implantable progesterone only contraception Jadelle® Sino-implant II ® Implanon Etonogestrel Nexplanon

Norplant®: It consisted of six flexible rods • prevent pregnancy for 5 years • 1983 -1990 -2002 Jadelle® : it consists of two implantable rods • prevent pregnancy for 5 years • 2000 Sino-implant II ® : it consists of two implantable rods • prevent pregnancy fo 4 years

Etonogestrel is more potent than Levonorgestrel Serum Etonogestrel levels shows less individual variation over time as compared with Levonorgestrel levels
Nexplanon and Implanon both are etonogestrel The brand name product Implanon is no longer commercially available in the U. S. , since 2012 and has been replaced by Nexplanon
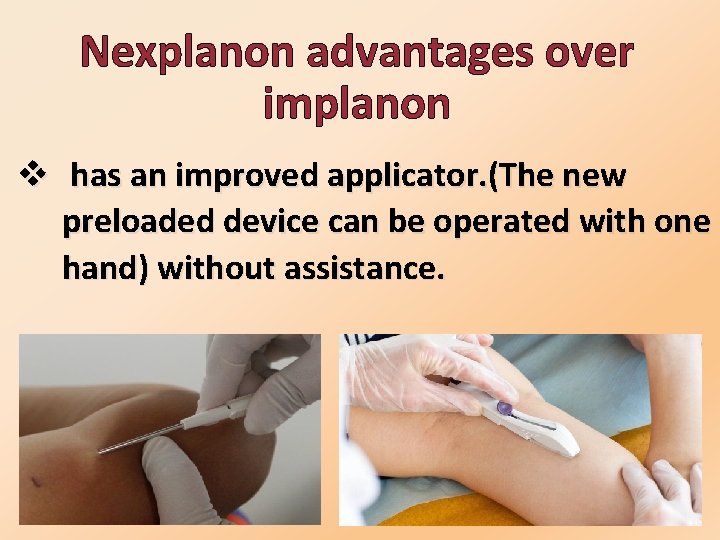
Nexplanon advantages over implanon v has an improved applicator. (The new preloaded device can be operated with one hand) without assistance.
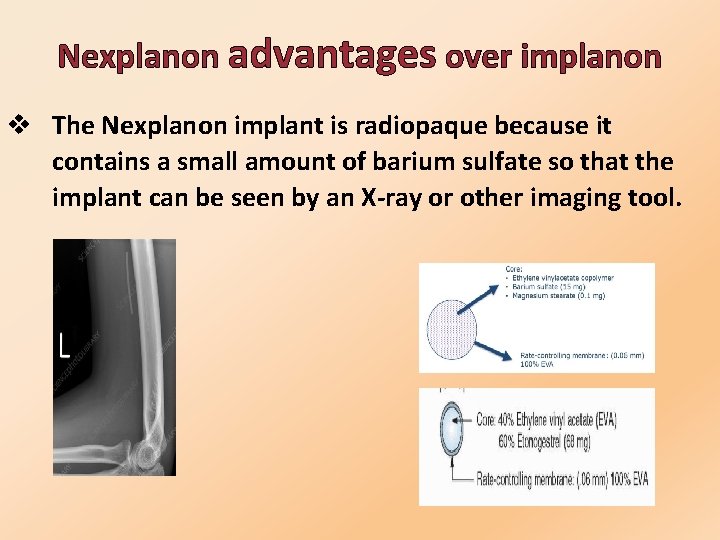
Nexplanon advantages over implanon v The Nexplanon implant is radiopaque because it contains a small amount of barium sulfate so that the implant can be seen by an X-ray or other imaging tool.
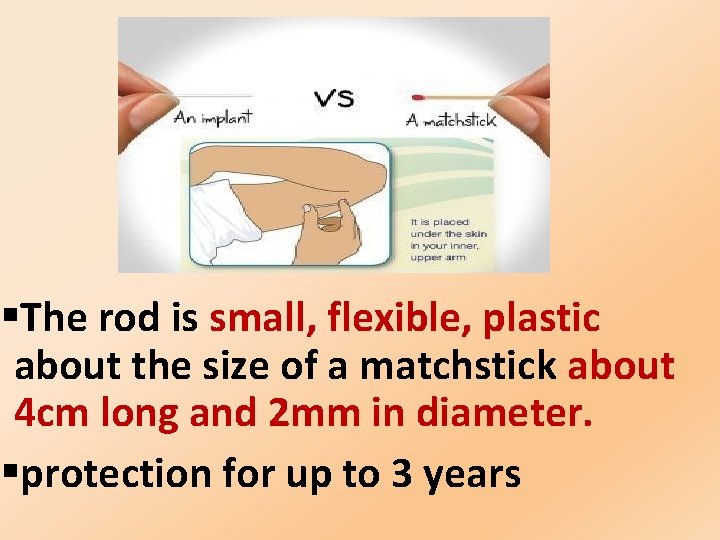
§The rod is small, flexible, plastic about the size of a matchstick about 4 cm long and 2 mm in diameter. §protection for up to 3 years

Mode of action?
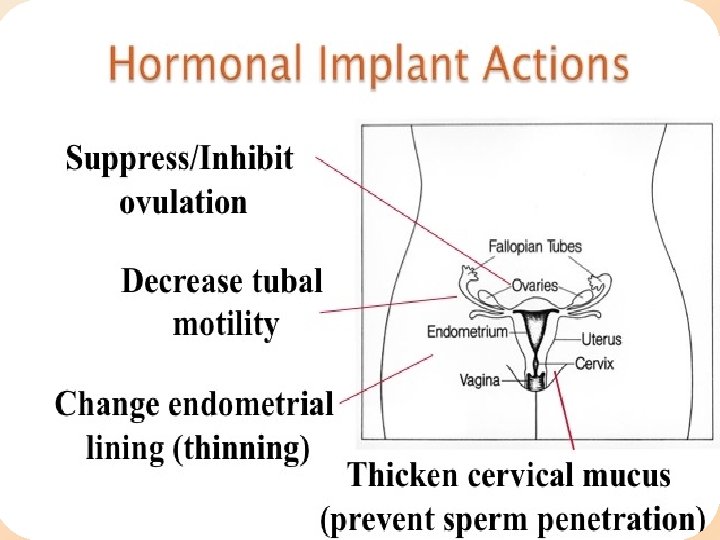

Advantages of implantable progesterone §Independence of user compliance §Rapid return to fertility: 90% of women ovulate within 30 days §pregnancies were observed to occur as early as 7 to 14 days after removal. § Efficacy not being affected by broad- spectrum antibiotics §Efficacy of implanon is extremely high with Pearl indices of 0. 01.

causes of failures of the method 1. improper placement 2. conception prior to method insertion. 3. drug interactions(enzyme-inducing drugs )

INSERTION When to insert it? ? ?

women without preceding hormone contraception • inserted within day 1 -5 of menstrual cycl first trimester abortion: • within 5 days second trimester abortion Postpartum(Not Breastfeeding): • within 21 days
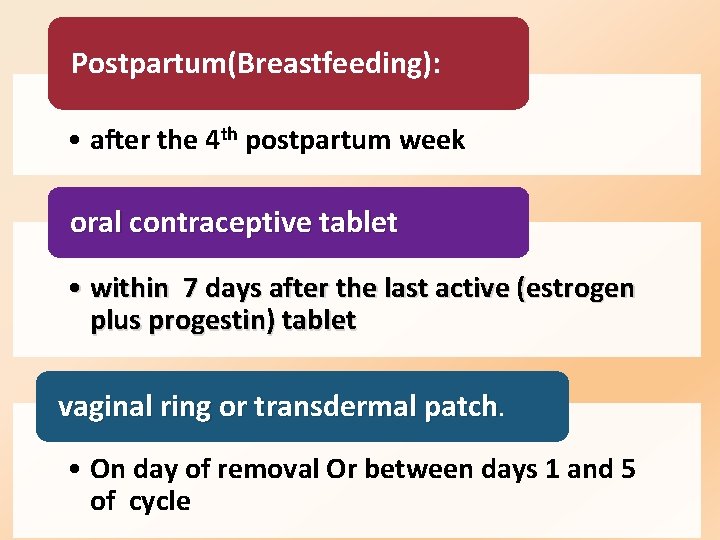
Postpartum(Breastfeeding): • after the 4 th postpartum week oral contraceptive tablet • within 7 days after the last active (estrogen plus progestin) tablet vaginal ring or transdermal patch. • On day of removal Or between days 1 and 5 of cycle

Injectable contraceptives • on the day the next injection is due Progesterone only pills • within 24 hours after taking the last tablet. previous implant or IUS. • on the same day the removal of them • using the same incision as long as the site is in the correct location

back-up contraception) use a barrier method until 7 days after insertion. (If intercourse has already occurred, pregnancy should be excluded.

Prior to insertion 1) Ensure there are no contra-indications to insertion contraindicated
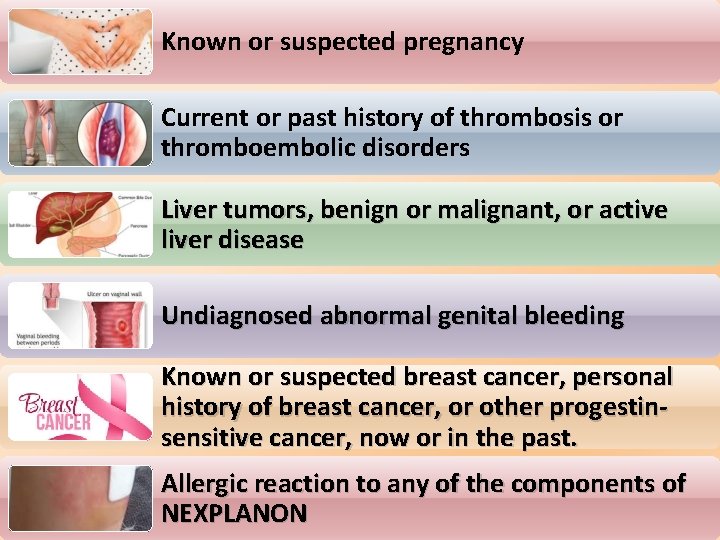
Known or suspected pregnancy Current or past history of thrombosis or thromboembolic disorders Liver tumors, benign or malignant, or active liver disease Undiagnosed abnormal genital bleeding Known or suspected breast cancer, personal history of breast cancer, or other progestinsensitive cancer, now or in the past. Allergic reaction to any of the components of NEXPLANON

counseling qside-effects q. Complications qinsertion/removal procedures qfollow-up

q. Obtain written consent for the procedure.

examination q. Blood pressure q. Height and weight to calculate the BMI.
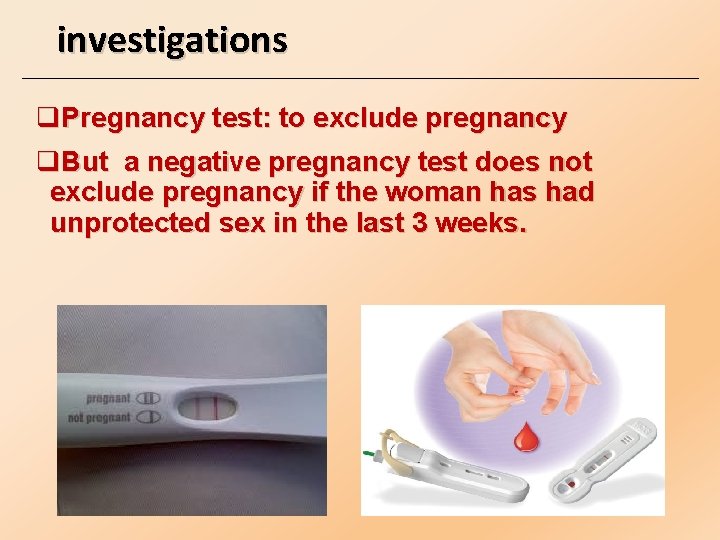
investigations q. Pregnancy test: to exclude pregnancy q. But a negative pregnancy test does not exclude pregnancy if the woman has had unprotected sex in the last 3 weeks.
§Clinicians should have attended a specific training programme, and ensure that the first 2 -3 insertions and removals are mentored by an experienced colleague.

Post procedure §Document procedure information in the woman’s hospital records §Provide the woman the supplied card and document: §site of Implanon §date of insertion §date for removal

§No routine follow-up is required. Review with her General Practitioner (GP) if there are 1. abnormalities of the insertion site eg. the implant is not palpable, pain 2. pregnancy test positive 3. presence of side-effects eg. change in menstrual pattern 4. develops a condition that contraindicates continuing with the implant

When we should remove it? qby the end of the third year q. When there is complications

What are the complications
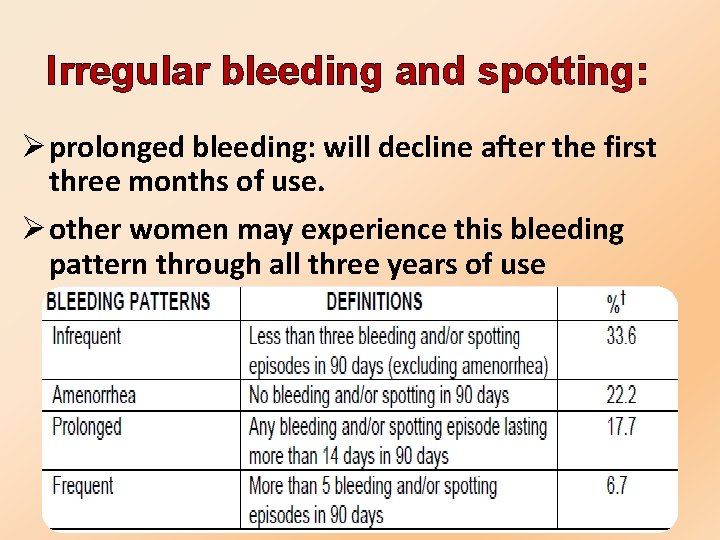
Irregular bleeding and spotting: Ø prolonged bleeding: will decline after the first three months of use. Ø other women may experience this bleeding pattern through all three years of use
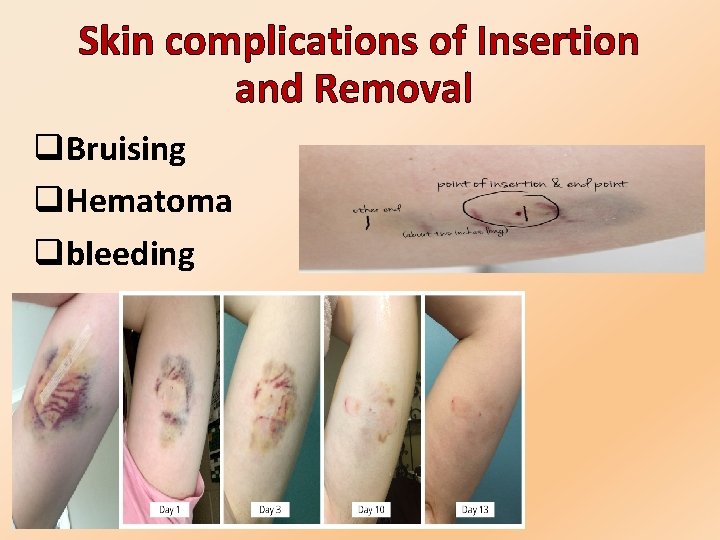
Skin complications of Insertion and Removal q. Bruising q. Hematoma qbleeding
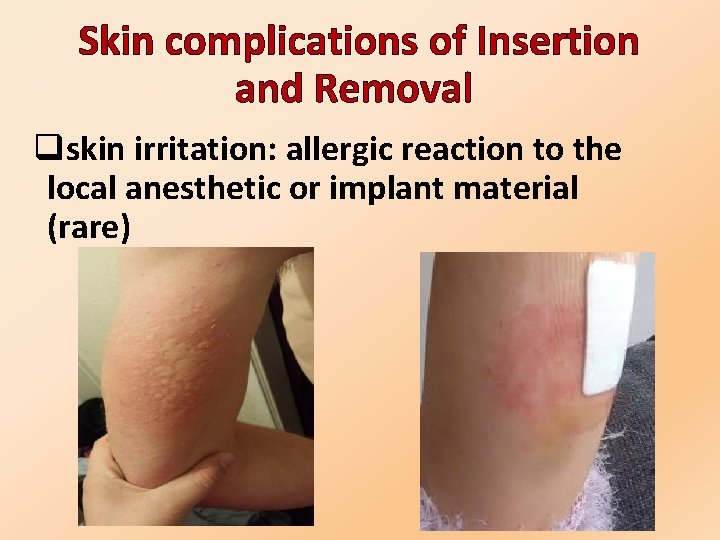
Skin complications of Insertion and Removal qskin irritation: allergic reaction to the local anesthetic or implant material (rare)
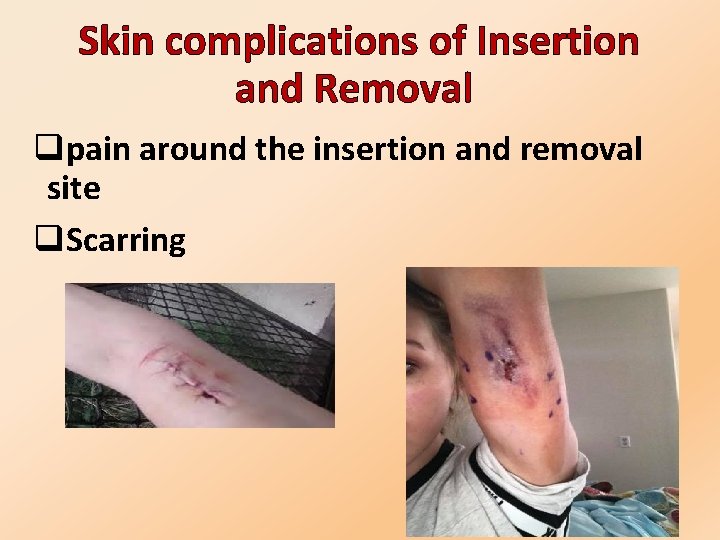
Skin complications of Insertion and Removal qpain around the insertion and removal site q. Scarring
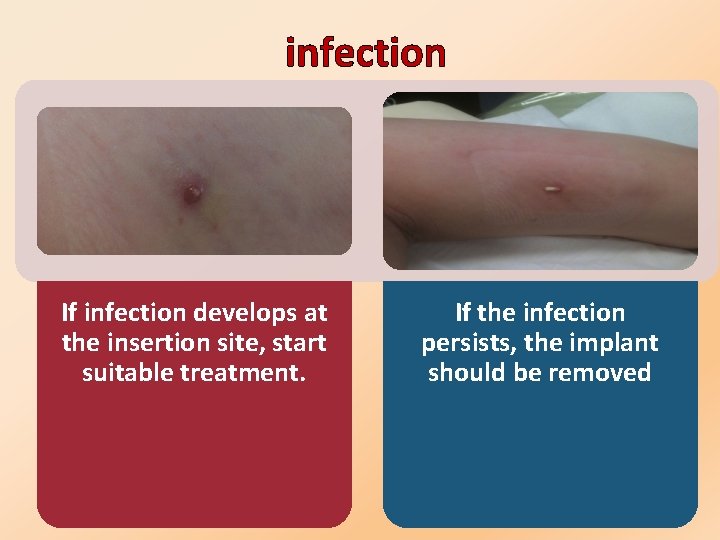
infection If infection develops at the insertion site, start suitable treatment. If the infection persists, the implant should be removed
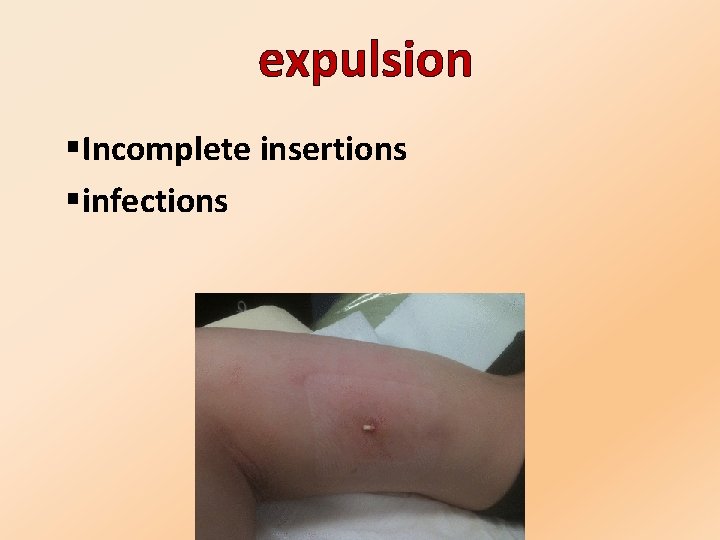
expulsion §Incomplete insertions §infections

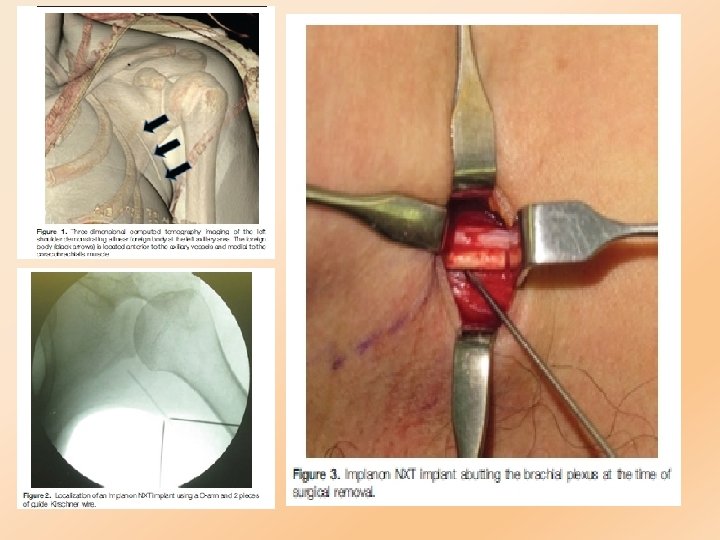
Deep insertion complications intramuscular fascial intravascular q neural injury: presented with paraesthesia q vascular injury. q migration
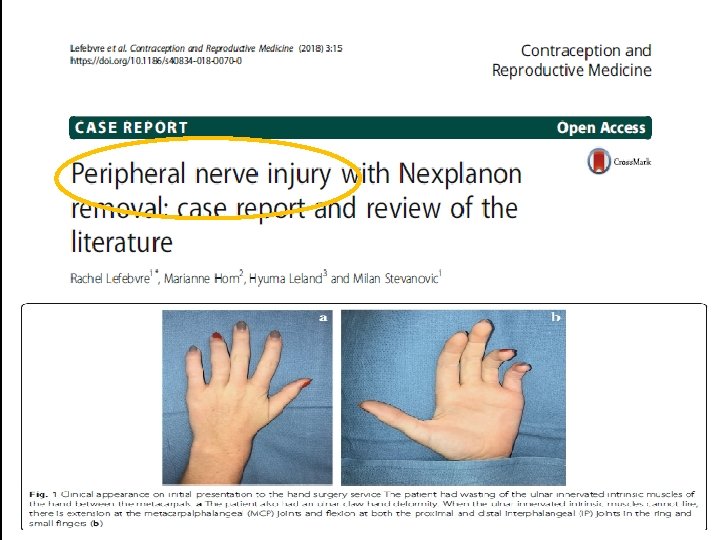
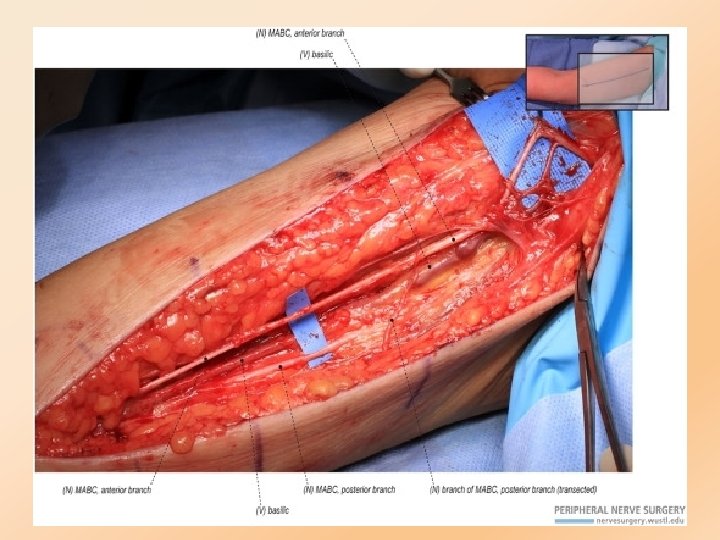

Migration §very rare, the rod can sometimes move slightly within the arm. §Nexplanon implants having reached the lung via the pulmonary artery


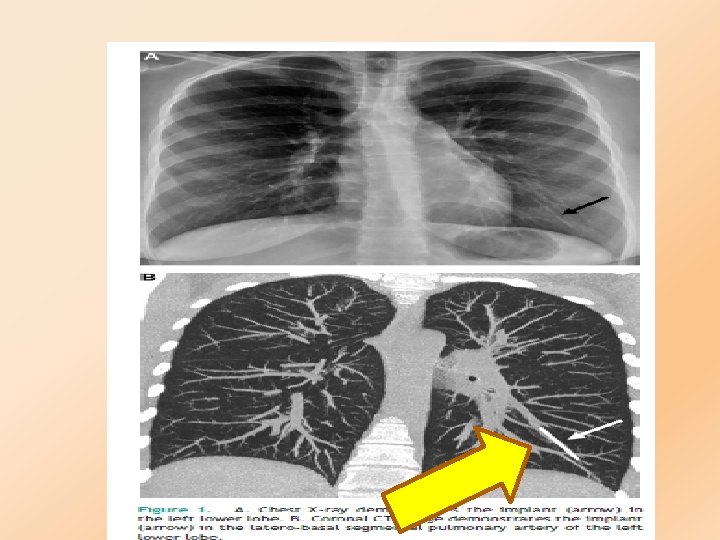
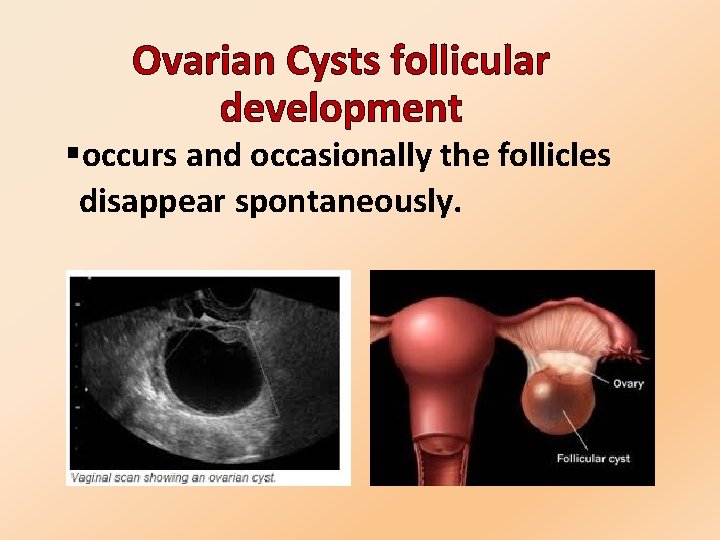
Ovarian Cysts follicular development §occurs and occasionally the follicles disappear spontaneously.

Possible weight gain §Some women may experience slight weight gain when using the implant. The average increase in body weight less than (2, 25 kg) over 2 years.

Thank you
- Slides: 46