Normal modes of vibration of a molecule An

Normal modes of vibration of a molecule – An Exercise by Dr. D. UTHRA Head Department of Physics DG Vaishnav College Chennai-106

By understanding this presentation you can find o the no. of vibration that a molecule can execute o the type of vibration, whether it is stretching or bending or torsion or out -of plane bending o the no. of symmetric and asymmetric vibration the molecule executes and to which species it belongs to

o 3 N-6 degrees of freedom are required to describe vibration of a non-linear molecule o 3 N-5 degrees of freedom are required to describe vibration of a linear molecule

An exercise! Search Ur Allied Chemistry book!! For the following molecules, n Find the molecule is linear or non-linear n Calculate the number of normal modes of vibration CO 2, H 2 O, NH 3, BF 3, HCl, CH 4, CO, H 2 S, C 2 H 2, PCl 3, BCl 3, N 2 F 2, B 2 H 6, C 6 H 6, SF 6, C 3 H 4, COS, CH 3 Cl, H 2 O 2 This is usually a Part A question!

Internal Coordinates o Changes in the bond lengths and in the inter-bond angles. Types of internal coordinates and their notation n n bond stretching (Δr ) angle deformation (ΔΦ) torsion (Δτ ) out of plane deformation (ΔΦ')

How to calculate number of vibrations using internal coordinates? nr = b nΦ= 4 b-3 a+a 1 nτ = b-a 1 nΦ’= no. of linear subsections in a molecule where, b - no. of bonds (ignoring the type of bond) a - no. of atoms a 1 - no. of atoms with multiplicity one o o Multiplicity means the number of bonds meeting the atom , ignoring the bond type. In H 2 O, multiplicity of H is 1 and that of O is 2. In NH 3, multiplicity of N is 3, while H is 1. In C 2 H 2, it is 1 for H and 2 for C (not 4 b’coz you ignore bond type). In CH 4, it is 1 for H and 4 for C. In CO 2, it is 2 for C and 1 for O.

Types of vibrations o o nr - no. of stretching vibrations nΦ - deformations nτ - torsional vibrations nΦ’- out of plane bending

Exercise Again! o Now using the internal coordinates, calculate the normal modes of vibration again for the molecules CO 2, H 2 O, NH 3, BF 3, HCl, CH 4, CO, H 2 S, C 2 H 2, PCl 3, BCl 3, N 2 F 2, B 2 H 6, C 6 H 6, SF 6, C 3 H 4, COS, CH 3 Cl, H 2 O 2

How to calculate normal modes of vibration using character table of the molecule? Let us try to calculate normal modes of vibration using the formula ni'= (1/h)∑R ℓΨj (R)χj' (R) o o o ni' - number of normal modes of vibration h - order of the point group ℓi - dimension of the ith species Ψj(R) – from the character table χj' (R) =(NR-2)(1+2 cosθ) (for proper rotations) =NR(-1+2 cosθ) (for other symmetry operations) NR- number of atoms that undergo No change due to operation R θ - angle of rotation for R

For XY 2 bent molecule, calculate normal modes of vibration by all three methods

Using 3 N-6 formula For an XY 2 bent molecule o N=3 ; 3 N-6 = 3 modes of vibration

Using internal coordinates In XY 2 bent molecule, o b = 2 (no. of bonds) o a = 3 (no. of atoms) o a 1= 2 (no. of atoms with multiplicity one) Hence, n nr = 2 n nΦ = 4*2 -3*3+2 =1 n nτ = 2 -2 = 0 3 N-6 = nr+nΦ+nτ = 2+1= 3

o For a XY 2 bent molecule, n nr = 2 n nΦ = 1 implies, this molecule has § § two bond stretching vibrations and one angle deformation (bending)vibration
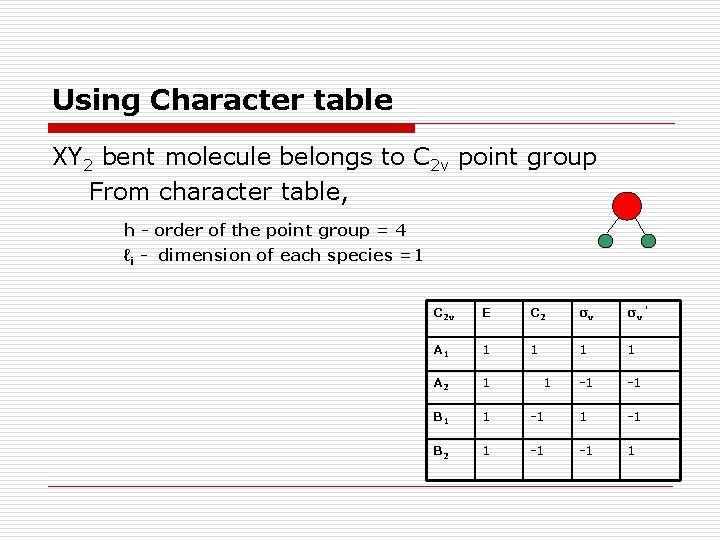
Using Character table XY 2 bent molecule belongs to C 2 v point group From character table, h - order of the point group = 4 ℓi - dimension of each species =1 C 2 v E C 2 σv σv ’ A 1 1 1 A 2 1 -1 -1 B 1 1 -1 B 2 1 -1 -1 1 1
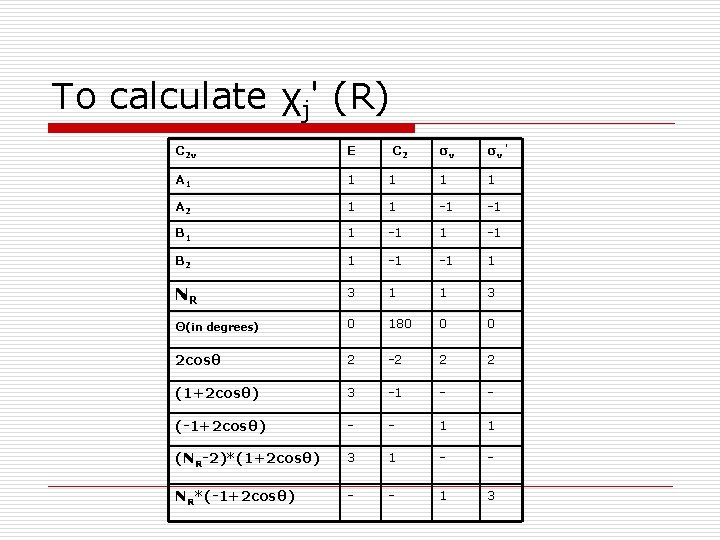
To calculate χj' (R) C 2 v E A 1 1 A 2 C 2 σv σv ’ 1 1 1 -1 -1 B 1 1 -1 B 2 1 -1 -1 1 NR 3 1 1 3 Θ(in degrees) 0 180 0 0 2 cosθ 2 -2 2 2 (1+2 cosθ) 3 -1 - - (-1+2 cosθ) - - 1 1 (NR-2)*(1+2 cosθ) 3 1 - - NR*(-1+2 cosθ) - - 1 3

ni'= (1/h)∑R ℓΨ (R)χ' (R) h - order of the C 2 v point group = 4 ℓi - dimension of each species in C 2 v point group=1 Ψj(R) – from the character table To find ni‘(A 1) h = 4 ; ℓ =1; Ψ (E) = 1; Ψ (C 2) = 1; Ψ (σv ’) = 1; χ' (E) = 3; χ' (C 2) = 1; χ' (σv ’) = 3. To find ni‘(A 2) h = 4 ; ℓ =1; Ψ (E) = 1; Ψ (C 2) = 1; Ψ (σv) = -1; Ψ (σv ’) = -1; χ' (E) = 3; χ' (C 2) = 1; χ' (σv ’) = 3. ni‘(A 1)= ni‘(A 2)= ni‘(B 1)= ni‘(B 2)= (1/4) [ 1*1*3 + + 1*1*1 + 1*1*3] =(1/4)*8=2 1*1*1 + 1*-1*3]=(1/4)*0=0 1*-1*1 + 1*1*3]=(1/4)*4=1

For an XY 2 bent molecule o There are 3 normal modes of vibration as 3 N-6 = 3 o Using internal coordinates, the nature of these 3 vibrations are found i. e. , § § two bond stretching vibrations and one angle deformation (bending) vibration o Further, using Character table, o o ni‘(A 1)=2 ni‘(B 2)=1 indicate, these 3 vibrations are distributed among A 1 and B 2 species. o From the knowledge of group theory, as the nature of A species is to have only symmetric vibrations and B is to have only asymmetric vibrations, o There are two symmetric vibrations and one asymmetric vibration Thus, knowledge of group theory systematically unfolds you the nature of vibration of any molecule!!
Tabulate and Tally! S. No. Molecule (formula) Linear/ Non-Linear Normal modes of vibration by using N 3 N-5/3 N-6 internal coordinates Character table Tally!

Congrats!! You have learnt to calculate normal modes of vibration. Try for many more molecules. Make it a fun game, a mental exercise, …

All the best and C u in the next presentation!! -uthra mam
- Slides: 20