Nontuberculous mycobacteria NTM Slide repository module 2 1

Non-tuberculous mycobacteria (NTM) Slide repository module 2 1

Development of the slide repository and financial support • The slide repository was developed by a multidisciplinary European scientific committee: – Stefano Aliberti, Respiratory Physician University of Milan, Italy – Claire Andrejak, Pulmonologist CHU Amiens-Picardie, Amiens, France – James Chalmers, Respiratory Physician Ninewells Hospital, Dundee, UK – Jakko van Ingen, Clinical Microbiologist Radboud University Medical Centre, Nijmegen, The Netherlands – Gianluca Milanese, Radiologist University of Parma, Italy – Eva Polverino, Pulmonologist, expert in respiratory infections Vall d’Hebron University Hospital, Barcelona, Spain – Dirk Wagner, Infectious disease expert University of Freiburg Medical Centre, Germany • The development of this slide repository by the multidisciplinary European scientific committee was facilitated by Physicians World Europe Gmb. H and funded by Insmed 2
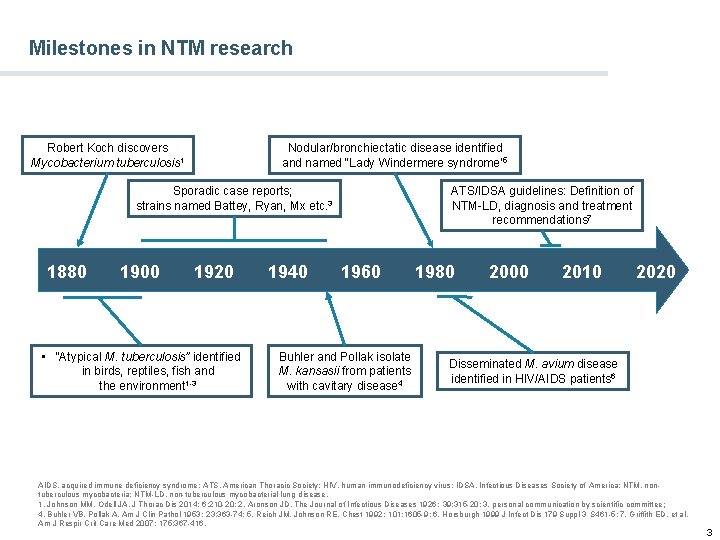
Milestones in NTM research Robert Koch discovers Mycobacterium tuberculosis 1 Nodular/bronchiectatic disease identified and named “Lady Windermere syndrome” 5 Sporadic case reports; strains named Battey, Ryan, Mx etc. 3 1880 1900 1920 • “Atypical M. tuberculosis” identified in birds, reptiles, fish and the environment 1 -3 1940 ATS/IDSA guidelines: Definition of NTM-LD, diagnosis and treatment recommendations 7 1960 Buhler and Pollak isolate M. kansasii from patients with cavitary disease 4 1980 2000 2010 2020 Disseminated M. avium disease identified in HIV/AIDS patients 6 AIDS, acquired immune deficiency syndrome; ATS, American Thoracic Society; HIV, human immunodeficiency virus; IDSA, Infectious Diseases Society of America; NTM, nontuberculous mycobacteria; NTM-LD, non-tuberculous mycobacterial lung disease. 1. Johnson MM, Odell JA. J Thorac Dis 2014; 6: 210 -20; 2. Aronson JD. The Journal of Infectious Diseases 1926; 39: 315 -20; 3. personal communication by scientific committee; 4. Buhler VB, Pollak A. Am J Clin Pathol 1953; 23: 363 -74; 5. Reich JM, Johnson RE. Chest 1992; 101: 1605 -9; 6. Horsburgh 1999 J Infect Dis 179 Suppl 3 S 461 -5; 7. Griffith ED, et al. Am J Respir Crit Care Med 2007; 175: 367 -416. 3

What are NTM? NOT assigned to either: 1, 2 Also known as: 2 • Mycobacterium tuberculosis complex • Mycobacterium leprae • • Environmental mycobacteria Opportunistic mycobacteria Atypical mycobacteria Mycobacteria other than tuberculosis (MOTT) NTM Recognition: • Identified soon after M. tuberculosis, not initially recognized as pathogenic 2 • Suspected as potential cause of human infections in the sanatorium era 4 • 1950 s: Direct evidence for causation of disease 4 • Opportunistic infections in HIV patients led to wider recognition & investigation 4 Characteristics: • Found in the environment 1, 2 • Opportunistic pathogens of humans & animals 3 • As of 2015: > 172 different species with distinct virulence features 3 HIV, human immunodeficiency virus; MOTT, mycobacteria other than tuberculosis; NTM, non-tuberculous mycobacteria. 1. Mc. Shane PJ, Glassroth J. Chest 2015; 148: 1517 -27; 2. Schönfeld N, et al. Pneumologie 2013; 67: 605 -33; 3. Faria S, et al. J Pathog 2015; 2015: 809014; 4. Orme IM, Ordway DJ. Infect Immun 2014; 82: 3516 -22. 4
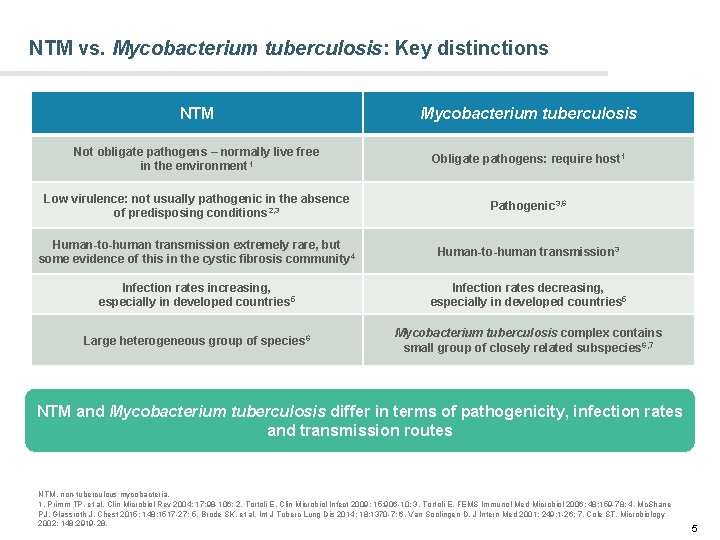
NTM vs. Mycobacterium tuberculosis: Key distinctions NTM Mycobacterium tuberculosis Not obligate pathogens – normally live free in the environment 1 Obligate pathogens: require host 1 Low virulence: not usually pathogenic in the absence of predisposing conditions 2, 3 Pathogenic 3, 6 Human-to-human transmission extremely rare, but some evidence of this in the cystic fibrosis community 4 Human-to-human transmission 3 Infection rates increasing, especially in developed countries 5 Infection rates decreasing, especially in developed countries 5 Large heterogeneous group of species 6 Mycobacterium tuberculosis complex contains small group of closely related subspecies 6, 7 NTM and Mycobacterium tuberculosis differ in terms of pathogenicity, infection rates and transmission routes NTM, non-tuberculous mycobacteria. 1. Primm TP, et al. Clin Microbiol Rev 2004; 17: 98 -106; 2. Tortoli E. Clin Microbiol Infect 2009; 15: 906 -10; 3. Tortoli E. FEMS Immunol Med Microbiol 2006; 48: 159 -78; 4. Mc. Shane PJ, Glassroth J. Chest 2015; 148: 1517 -27; 5. Brode SK, et al. Int J Tuberc Lung Dis 2014; 18: 1370 -7; 6. Van Soolingen D. J Intern Med 2001; 249: 1 -26; 7. Cole ST. Microbiology 2002; 148: 2919 -28. 5

NTM classification 1 rra tri od e es vial gil e. J vu S m 60 gil Sp vu m yr 1 PY ira nic R um vac ca van baa e lenii chub uen se sp. K MS sp. M CS rh 49 e Br TN ra ra e lep le p gastri kansasii kubicae te kyorinense celatum i shimolde e fraga IVM pi R xeno opi xen iae ern hib kii ae gb m. en hro nc rd. no a b go se lon nen se si en nu se Slow (usually 1 -3 weeks, sometimes up to 8 weeks 2) gordonae Rapid (usually 7 -10 days 2) mar. E urope mar inum M 8 lifla 2 ndii ulc era ns szu lga riy i ad he ns lac e us Growth rate 00 . 7 n ca 10 08 00 7 n. ca ca 8 00 60 n. rc R e 37 tub. H erc N Z tub. K erc tub um n a afric is oryg bovis ico Mex bovis o ky o T s bovi conspicuum sherrisii sp. JLS doricum thermores. triplex hassiacum florentinum phlei ca n sp palustre sp. IEC 1808 interj ectum para. IEC 26 par a. 0 715 6 par a. 1 004 pa 3 rm en bo se he mi sa cu sk m atc pa ra h. sc eu ro ro f. pa eu m e ns ke as br ne m 0 1 iu av m K 4 AP iu M av e ns ium av mbie lo co ra ae e im ch ens gon 0 yon 395 c. 1 intra c. 02 intra. 64 intrac ranii ind. p sm e ar g. M ia se C 2. 3 ns 6 co e nfl 0 M F ue nti T s f ab sc. allax G 00 ab 6 sc. 50 94 abs ces sus imm uno gen chelo nae A. su bflavu s tusciae B 3 s. NB rhode 623 g. JS e m s um grin pere m uitu. fort tion cep se n o c ten i r ge 41 ma. 1 sp 55. 1 yi sp sk lin wo NTM, non-tuberculous mycobacteria. 1. Fedrizzi T, et al. Sci Rep 2017; 7: 45258; 2. Thomson 2009 Respirology 14 (1), 12 -26. Figure adapted from Fedrizzi T, et al. Sci Rep 2017; 7: 45258 6

NTM are found throughout the environment Drinking water distribution systems (biofilm formation) Natural water sources Acidic, brownwater swamps Potting soils Boreal forest soils and peats Hot tubs and spas Metal removal fluid systems NTM habitats are intimately shared with those of humans NTM, non-tuberculous mycobacteria. Falkinham JO, 3 rd. J Appl Microbiol 2009; 107: 356 -67; pictures taken from https: //pixabay. com/. 7

Transmission of NTM Inhalation of NTM-laden aerosols or dust 1 Ingestion of soil or water 1 Likely routes of infection Contamination of hospital water supplies and medical equipment 2 Person-to-person transmission extremely rare, but some evidence of this in the CF community 3 Gastroesophageal reflux disease has been indicated as a mediator of NTM-LD 1 • Swallowing of NTM followed by gastric reflux leading to aspiration into the lung CF, cystic fibrosis; NTM, non-tuberculous mycobacteria; NTM-LD, non-tuberculous mycobacterial lung disease 1. Falkinham JO, 3 rd. J Appl Microbiol 2009; 107: 356 -67; 2. Johnson MM, Odell JA. J Thorac Dis 2014; 6: 210 -20; 3. Mc. Shane PJ, Glassroth J. Chest 2015; 148: 1517 -27. 8

The hydrophobic outer membrane supports NTM survival and distribution Lipopolysaccharide • Mycolic acid Cell wall • Promotes surface attachment and biofilm formation Prevents wash-out Peptidoglycan/ arabinan layer Plasma membrane • Protects against antimicrobial agents, including antibiotics and disinfectants • • Concentrates bacteria at air/water interface Aids aerosol distribution and transmission from water distribution systems by inhalation Protein Mycobacterium Biofilm formation and hydrophobic characteristics allow colonisation of unfavourable habitats and easy spread NTM, non-tuberculous mycobacteria. Falkinham JO, 3 rd. J Appl Microbiol 2009; 107: 356 -67. 9

Risk factors for NTM-LD NP-FR-00023 10
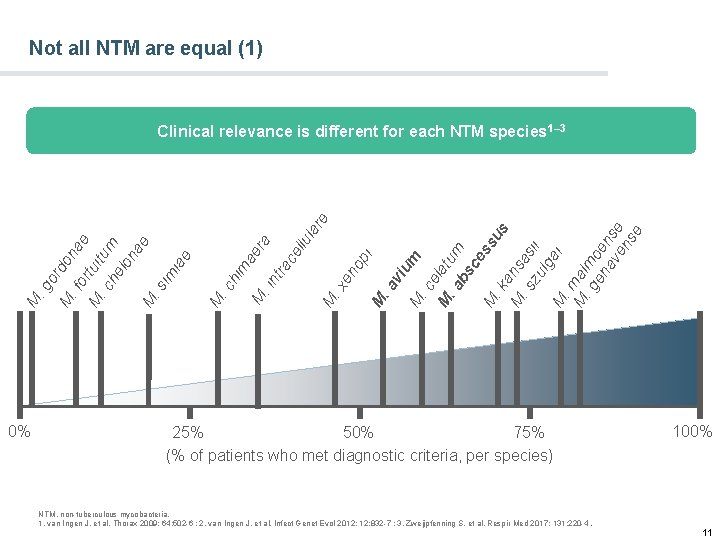
Not all NTM are equal (1) 0% ela. a tum bs ce ss M us. k an M. s sas zu ii lga M i. m M al. g m en oe av nse en se M . c vi. a M M i op M . x en tra um llu ce ra. in M . c M im. s M him iae ae ae lon m itu M . c he tu or . f M M . g or do na e lar e Clinical relevance is different for each NTM species 1– 3 25% 50% 75% (% of patients who met diagnostic criteria, per species) NTM, non-tuberculous mycobacteria. 1. van Ingen J, et al. Thorax 2009; 64: 502 -6 ; 2. van Ingen J, et al. Infect Genet Evol 2012; 12: 832 -7 ; 3. Zweijpfenning S, et al. Respir Med 2017; 131: 220 -4. 100% 11

Not all NTM are equal (2) NTM-LD frequency according to NTM species (N=104) Cases (n) 15 M. kansasii 15 4 11 M. xenopi 5 M. malmoense 3 15 8 = disease = no disease M. avium/ M. intracellulare 29 M. fortuitum/ M. chelonae 22 4 5 10 10 1 M. gordonae 0 20 40 51 % 60 80 100 NTM species isolated from pulmonary samples vary in virulence: Isolation of M. kansasii often correlates with disease NTM, non-tuberculous mycobacteria; NTM-LD, non-tuberculous mycobacterial lung disease. Adapted from Schönfeld N, et al. Pneumologie 2013; 67: 605 -33. 12

NTM disease: 4 main manifestations Pulmonary disease 1, 2 Predisposing lung conditions 1, 3 Predisposing genetic factors 4 Situational (hypersensitivity pneumonitis)2, 3 Lymphatic disease 1, 2 Typically an infantile disease affecting cervical lymph nodes 2 Also in adults with HIV infection 1 The lung is by far the most frequent disease site 1 Disseminated disease 1, 2 Most commonly seen in association with profound immunosuppression, e. g. HIV infection 2, 3 Skin/soft tissue disease 1, 2 The most common sources include: • contact with contaminated water or infected fish 2 • traumas and surgical wounds 2 Nosocomial infections have been described 3 Both host factors and organism characteristics influence the susceptibility and manifestations of NTM disease 3 HIV, human immunodeficiency virus; NTM, non-tuberculous mycobacteria. 1. Griffith DE, et al. Am J Respir Crit Care Med 2007; 175: 367 -416; 2. Tortoli E. Clin Microbiol Infect 2009; 15: 906 -10; 3. Johnson MM, Odell JA. J Thorac Dis 2014; 6: 210 -20; 4. Mc. Shane PJ, Glassroth J. Chest 2015; 148: 1517 -27. 13

Nodular/bronchiectatic and fibrocavitary disease – the two forms of NTM-LD Nodular/bronchiectatic “Lady Windermere syndrome” Fibrocavitary Epidemiology • Postmenopausal females 1 • Scoliosis, mitral valve prolapse, low BMI 1, 2 • No pre-existing lung disease 2 Clinical course • Prolonged cough, fatigue, weight loss 1 Microbiology (classical) • Low yield of Ziehl-Neelsen positive sputum culture - should repeat or use BAL 4 Radiology • Bronchiectasis with nodules “tree-in-bud” appearance 1 • Middle lobe and lingula worst affected 1, 2 Epidemiology • Mostly males 1 • Middle-aged (late 40 s-early 50 s)1 • Pre-existing COPD, silicosis, fibrosis 3 Clinical course • Tuberculosis-like, but slower 4 Microbiology (classical) • Often Ziehl-Neelsen positive sputum 4 • High yield of culture 4 Radiology • Fibro-cavitary lesions, upper lobes 3 BAL, bronchoalveolar lavage; COPD, chronic obstructive pulmonary disease; NTM-LD, non-tuberculous mycobacterial lung disease. 1. Griffith DE, et al. Am J Respir Crit Care Med 2007; 175: 367 -416; 2. Kim RD, et al. Am J Respir Crit Care Med 2008; 178: 1066 -74; 3. Johnson MM, Odell JA. J Thorac Dis 2014; 6: 210 -20; 4. personal communication by scientific committee. 14

Host risk factors – nodular/bronchiectatic NTM-LD Body morphotype of patients with active NTM infection compared with healthy controls 1 Matched control subjects* 161. 0 cm Female patients (n=60) 164. 7 cm* (Height) 28. 2 21. 1 (BMI) 95. 1 cm (Waist) 74. 2 cm * (Waist) • • 73. 1 kg 57. 2 kg* (Weight) Lean and tall stature 1 Low BMI 1, 2 Skeletal abnormalities 1, 2 Scoliosis Pectus excavatum Mitral valve prolapse 1 Among Caucasians, women have a higher prevalence than men 3 There is an association between nodular/bronchiectatic NTM-LD and a particular body habitus – predominantly in postmenopausal women 1, 4 *NHANES age- and ethnicity-matched female control subjects (2001 -2002 data). BMI, Body-Mass-Index; NTM, non-tuberculous mycobacteria; NTM-LD, non ‑tuberculous mycobacterial lung disease. 1. Adapted from Kim RD, et al. Am J Respir Crit Care Med 2008; 178: 1066 -74; 2. Dirac MA, et al. Am J Respir Crit Care Med 2012; 186: 684 -91; 3. Adjemian J, et al. Am J Respir Crit Care Med 2012; 185: 881 -6; 4. Griffith DE, et al. Am J Respir Crit Care Med 2007; 175: 367 -416. 15
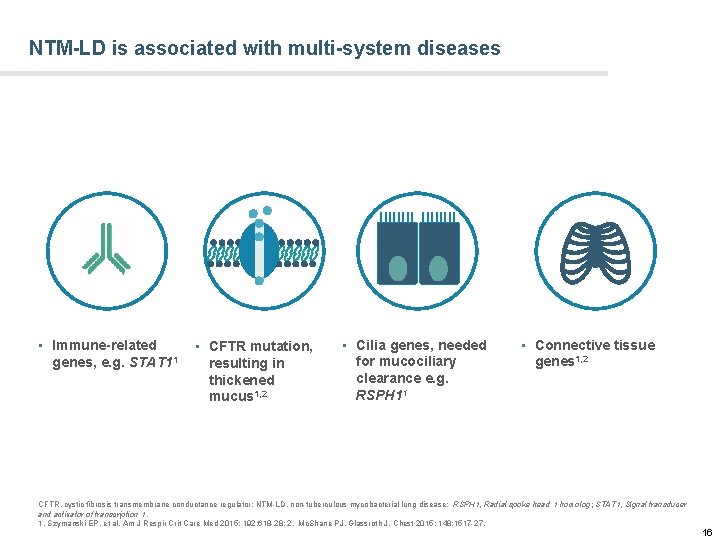
NTM-LD is associated with multi-system diseases • Immune-related genes, e. g. STAT 11 • CFTR mutation, resulting in thickened mucus 1, 2 • Cilia genes, needed for mucociliary clearance e. g. RSPH 11 • Connective tissue genes 1, 2 CFTR, cystic fibrosis transmembrane conductance regulator; NTM-LD, non-tuberculous mycobacterial lung disease; RSPH 1, Radial spoke head 1 homolog; STAT 1, Signal transducer and activator of transcription 1. 1. Szymanski EP, et al. Am J Respir Crit Care Med 2015; 192: 618 -28; 2. Mc. Shane PJ, Glassroth J. Chest 2015; 148: 1517 -27. 16
Host risk factors – fibrocavitary NTM-LD ♂ • Males in their late 40 s and early 50 s 1 • History of cigarette smoking • Often, excessive alcohol use 1 • Structural lung disease e. g. COPD, silicosis or prior tuberculosis 2 Fibrocavitary NTM-LD commonly occurs in older males, often smokers, with a history of underlying lung disease 1, 2 COPD, chronic obstructive pulmonary disease; NTM-LD, non-tuberculous mycobacterial lung disease. 1. Griffith DE, et al. Am J Respir Crit Care Med 2007; 175: 367 -416; 2. Johnson MM, Odell JA. J Thorac Dis 2014; 6: 210 -20. 17
Environmental risk factors for NTM-LD Areas in the United States* with a high risk for a NTM-LD infection Socioeconomic and environmental risk factors include: • • • Greater population density Higher education and income High evapotranspiration and percentages covered by surface water *US study chosen due to high number of unique cases: Nearly 2. 3 million individuals from all 50 states and the District of Columbia were included, from which 16, 508 NTM-LD claims representing 2, 548 unique cases were identified. Data are representative but not directly applicable to the European situation. NTM-LD, non-tuberculous mycobacterial lung disease. Adapted from Adjemian J, et al. Am J Respir Crit Care Med 2012; 186: 553 -8. 18

Genetic and iatrogenic factors associated with NTM-LD Genetic predisposition • Potentially a multigenic disease 1 Iatrogenic factors • • Mutation in CFTR gene 1, 2 Other mutations including immune factors, cilia genes and connective tissue genes 1, 2 Familial clustering of NTM-LD 3 - Majority of cases in non‑smoking siblings with high prevalence of scoliosis and CFTR mutations 3 • • Immunosuppressive treatments associated with NTM-LD include: - COPD patients treated with inhaled corticosteroids (adjusted OR 19. 6; 95% CI 9. 7– 39. 6)4 - DMARDs, anti-TNF-α therapy, oral corticosteroids 5, 6 Organ transplantation 7, 8 Haematological malignancy 9 CFTR, cystic fibrosis conductance regulator; CI, confidence interval; COPD, chronic obstructive pulmonary disease; DMARD, disease-modifying antirheumatic drug; NTM-LD, nontuberculous mycobacterial lung disease; OR, odds ratio; TNF, tumor necrosis factor. 1. Szymanski EP, et al. Am J Respir Crit Care Med 2015; 192: 618 -28; 2. Kim RD, et al. Am J Respir Crit Care Med 2008; 178: 1066 -74; 3. Colombo RE, et al. Chest 2010; 137: 62934; 4. Andréjak C, et al. Thorax 2013; 68: 256 -62; 5. Winthrop KL, et al. Emerg Infect Dis 2009; 15: 1556 -61 ; 6. Brode SK, et al. Thorax 2015; 70: 677 -82; 7. Knoll BM, et al. Transpl Infect Dis 2012; 14: 452 -60; 8. Daley CL. Curr Opin Organ Transplant 2009; 14: 619 -24 ; 9. Chen CY, et al. Eur J Clin Microbiol Infect Dis 2012; 31: 1059 -66. 19

The pattern of disease recognition has changed over time Changes in radiological appearance of NTM-LD cases in Queensland, Australia between 1999 and 2005 1999 2005 Incidence/100, 000 population P-value Cavitary disease 0. 9 0. 7 0. 25 Non-cavitary disease 0. 7 2. 1 <0. 0001 0. 7 0. 9 2. 1 Shift from cavitary disease in middle-aged men who smoke to the nodular/bronchiectatic form of disease in older women NTM-LD, non-tuberculous mycobacterial lung disease. Thomson RM, et al. Emerg Infect Dis 2010; 16: 1576 -83. 20
Bronchopulmonary factors associated with NTM-LD* Pneumococoniosis, Alveolar proteinosis COPD Silicosis Cystic fibrosis Bronchiectasis Cystic fibrosis Previous TB infection *Please note size of shapes is not proportional to predicted level of association. COPD, chronic obstructive pulmonary disease; NTM-LD, non-tuberculous mycobacterial lung disease; TB, tuberculosis. Sexton P, Harrison AC. Eur Respir J 2008; 31: 1322 -33. 21

Most common bronchopulmonary factors associated with NTM-LD Bronchiectasis COPD Meta-analysis of 8 studies showed that the overall prevalence of NTM was 9. 3% in patients with bronchiectasis 1 Most frequently NTM-LD‑associated condition in Germany 3 Cause and effect are hard to differentiate 2 COPD treated with ICS therapy is a strong risk factor for NTM-LD 4 No consistent evidence that NTM-LD and bronchiectasis severity are correlated 2 COPD and NTM-LD prevalence are both increasing 4 Many patients with NTM-LD have at least one additional lung disease: either bronchiectasis, COPD or both 5 COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; NTM, non-tuberculous mycobacteria; NTM-LD, non-tuberculous mycobacterial lung disease. 1. Chu H, et al. Arch Med Sci. 2014; 10: 661 -8; 2. Sexton P, Harrison AC. Eur Respir J 2008; 31: 1322 -33; 3. Ringshausen FC, et al. BMC Infect Dis 2013; 13: 231; 4. Andréjak C, et al. Thorax 2013; 68: 256 -62, 5. Griffith DE, Aksamit TR. Clin Chest Med 2012; 33: 283 -95. 22
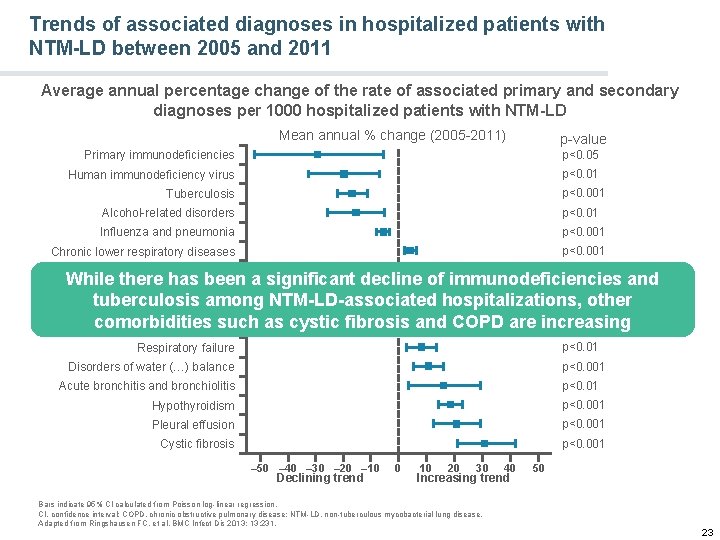
Trends of associated diagnoses in hospitalized patients with NTM-LD between 2005 and 2011 Average annual percentage change of the rate of associated primary and secondary diagnoses per 1000 hospitalized patients with NTM-LD Mean annual % change (2005 -2011) p-value Primary immunodeficiencies p<0. 05 Human immunodeficiency virus p<0. 01 p<0. 001 Tuberculosis Alcohol-related disorders p<0. 01 Influenza and pneumonia p<0. 001 Chronic lower respiratory diseases p<0. 001 COPD and emphysema p<0. 001 While there has been a significant decline of immunodeficiencies and p<0. 05 Haemoptysis tuberculosis among NTM-LD-associated hospitalizations, other p<0. 01 Bronchiectasis comorbidities such as cystic fibrosis and COPD are increasing p<0. 001 Primary hypertension p<0. 01 Respiratory failure p<0. 001 Disorders of water (…) balance p<0. 01 Acute bronchitis and bronchiolitis Hypothyroidism p<0. 001 Pleural effusion p<0. 001 Cystic fibrosis p<0. 001 – 50 – 40 – 30 – 20 – 10 Declining trend 0 10 20 30 40 Increasing trend Bars indicate 95% CI calculated from Poisson log-linear regression. CI, confidence interval; COPD, chronic obstructive pulmonary disease; NTM-LD, non-tuberculous mycobacterial lung disease. Adapted from Ringshausen FC, et al. BMC Infect Dis 2013; 13: 231. 50 23

NTM and disease NP-FR-00023 24

NTM organisms commonly isolated from the respiratory tract Growth rate classification Common NTM*1 Slow-growing mycobacteria (SGM) Mycobacterium avium complex (MAC) M. gordonae M. kansasii M. xenopi Rapid-growing mycobacteria (RGM) M. abscessus spp. M. fortuitium Uncommon NTM*1, 4 M. celatum M. malmoense† M. shimoidei M. simiae Sporadic isolation • ~1/3 of identified species considered clinically significant 2 • Growth rate classification (RGM vs. SGM) has clinically important repercussions: 3 § RGM and SGM differ in their antimicrobial susceptibility and treatment regimens; RGM are more likely to be resistant § SGM mostly responsible for pulmonary and lymphonodal diseases § RGM can also affect cutis, bones and joints *In alphabetical order; †In northern Europe. MAC, Mycobacterium avium complex; NTM, non-tuberculous mycobacteria; RGM, Rapid-growing mycobacteria; SGM, Slow-growing mycobacteria. 1. Hoefsloot W, et al. Eur Respir J 2013; 42: 1604 -13; 175: 367 -416; 2. Tortoli E. FEMS Immunol Med Microbiol 2006; 48: 159 -78; 3. Tortoli E. Clin Microbiol Infect 2009; 15: 906 -10; 4. personal communication by scientific committee. 25

Diagnosis and management of NTM-LD are challenging Late diagnosis Therapeutic challenge 1– 3 Non-specific symptoms Multi-drug regimens Symptoms may overlap with underlying lung disease 1 Treatment for 1– 2 years de lin e Risk of side effects G ui Refractory disease Poor adherence to evidence-based treatment guidelines 4 Little data on efficacy of other drugs or interventions 5 Promotes antibiotic resistance Cases should be referred to NTM-LD experts 6 A multidisciplinary approach is required to adequately diagnose and manage patients with NTM-LD 6, 7 NTM-LD, non-tuberculous mycobacterial lung disease. 1. Griffith DE, et al. Am J Respir Crit Care Med 2007; 175: 367 -416; 2. Thomson RM, Yew WW. Respirology 2009; 14: 12 -26; 3. van Ingen J, et al. Drug Resist Updat 2012; 15: 149 -61; 4. Adjemian J, et al. Ann Am Thorac Soc 2014; 11: 9 -16; 5. Griffith DE, Aksamit TR. Curr Opin Infect Dis 2012; 25: 218 -27; 6. Haworth CS, et al. Thorax 2017; 72: ii 1 -ii 64; 7. Ryu YJ, et al. Tuberc Respir Dis (Seoul) 2016; 79: 74 -84. 26

Epidemiology NP-FR-00023 27
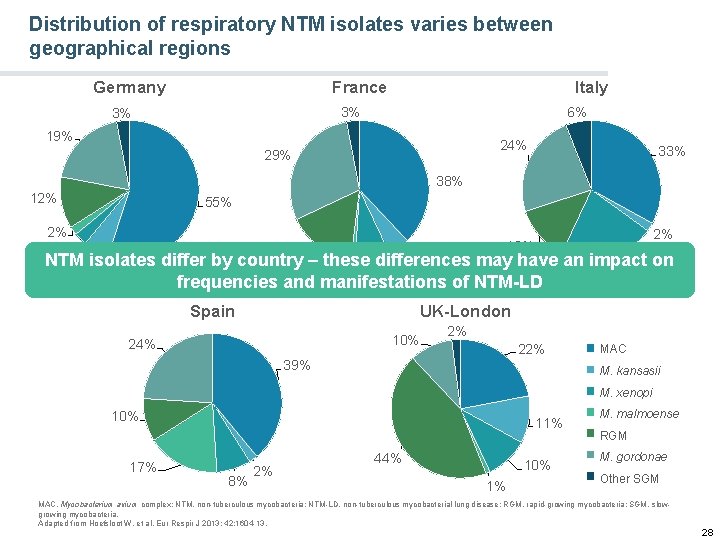
Distribution of respiratory NTM isolates varies between geographical regions Germany France Italy 3% 3% 6% 19% 24% 29% 33% 38% 12% 55% 2% 3% 2% 13% 16% 5% NTM isolates differ by country – these differences may have an impact on 6% 22% 1% 8% frequencies and manifestations of NTM-LD Spain UK-London 10% 24% 2% 22% 39% MAC M. kansasii M. xenopi 10% 17% 11% 8% 2% 44% 10% 1% M. malmoense RGM M. gordonae Other SGM MAC, Mycobacterium avium complex; NTM, non-tuberculous mycobacteria; NTM-LD, non-tuberculous mycobacterial lung disease; RGM, rapid-growing mycobacteria; SGM, slowgrowing mycobacteria. Adapted from Hoefsloot W, et al. Eur Respir J 2013; 42: 1604 -13. 28
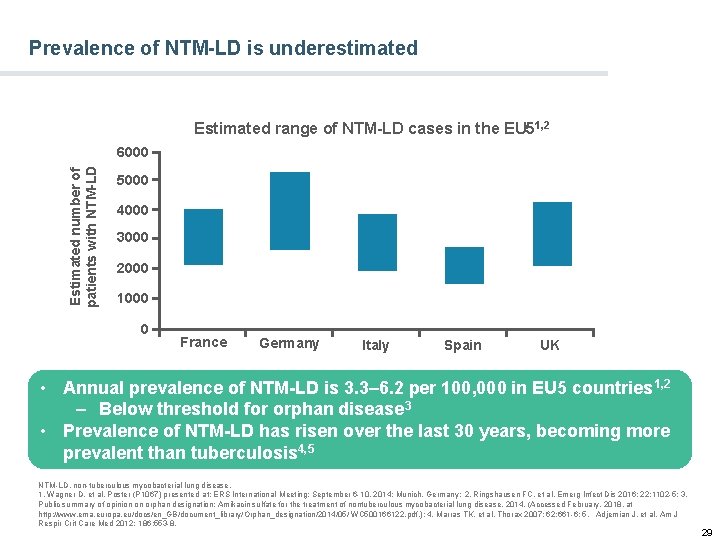
Prevalence of NTM-LD is underestimated Estimated range of NTM-LD cases in the EU 51, 2 Estimated number of patients with NTM-LD 6000 5000 4000 3000 2000 1000 0 France Germany Italy Spain UK • Annual prevalence of NTM-LD is 3. 3– 6. 2 per 100, 000 in EU 5 countries 1, 2 – Below threshold for orphan disease 3 • Prevalence of NTM-LD has risen over the last 30 years, becoming more prevalent than tuberculosis 4, 5 NTM-LD, non-tuberculous mycobacterial lung disease. 1. Wagner D, et al. Poster (P 1067) presented at: ERS International Meeting; September 6 -10, 2014; Munich, Germany; 2. Ringshausen FC, et al. Emerg Infect Dis 2016; 22: 1102 -5; 3. Public summary of opinion on orphan designation: Amikacin sulfate for the treatment of nontuberculous mycobacterial lung disease. 2014. (Accessed February, 2018, at http: //www. ema. europa. eu/docs/en_GB/document_library/Orphan_designation/2014/05/ WC 500166122. pdf. ); 4. Marras TK, et al. Thorax 2007; 62: 661 -6; 5. Adjemian J, et al. Am J Respir Crit Care Med 2012; 186: 553 -8. 29
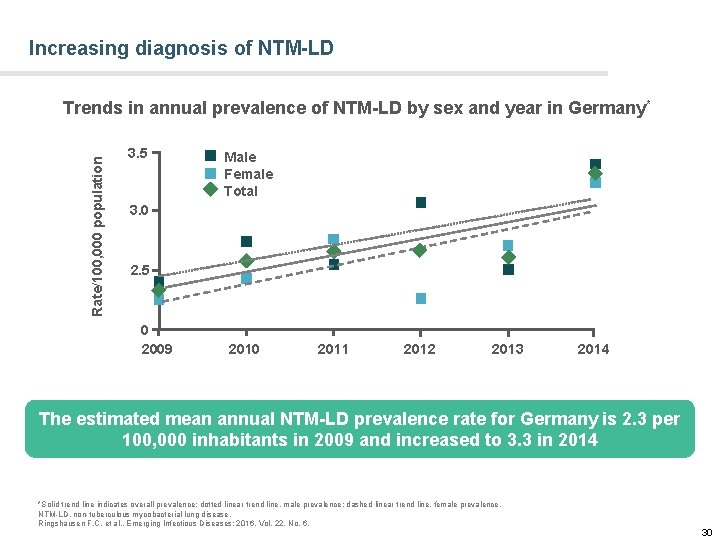
Increasing diagnosis of NTM-LD Rate/100, 000 population Trends in annual prevalence of NTM-LD by sex and year in Germany * 3. 5 Male Female Total 3. 0 2. 5 0 2009 2010 2011 2012 2013 2014 The estimated mean annual NTM-LD prevalence rate for Germany is 2. 3 per 100, 000 inhabitants in 2009 and increased to 3. 3 in 2014 *Solid trend line indicates overall prevalence; dotted linear trend line, male prevalence; dashed linear trend line, female prevalence. NTM-LD, non-tuberculous mycobacterial lung disease. Ringshausen F. C. et al. , Emerging Infectious Diseases: 2016, Vol. 22, No. 6. 30

Why are the rates of NTM infections increasing? Improved quality of diagnostic methods e. g. microbiological detection techniques 1 Ageing population 2, 3 with an increase in the prevalence of COPD 2 and other co-morbidities 1 Increased disease awareness 1 Tuberculosis infection can result in cross-protection against NTM 4 NTM-LD infection rates may be underestimated 5 Further research on the epidemiology of NTM infections by international research collaborations is required 6 COPD, chronic obstructive pulmonary disease; NTM, non-tuberculous mycobacteria; NTM-LD, non-tuberculous mycobacterial lung disease. 1. Mc. Shane PJ, Glassroth J. Chest 2015; DOI: 10. 1378/chest. 15 -0458; 2. van Ingen J, et al. Int J Tuberc Lung Dis 2010; 14: 1176 -80; 3. Al-Houqani M, et al. Chest 2012; 141: 190 -7; 4. Gopinath K, Singh S. PLo. S Negl Trop Dis 2010; 4: e 615; 5. van der Werf MJ, et al. BMC Infect Dis 2014; 14: 62; 6. Ringshausen FC, et al. BMC Infect Dis 2013; 13: 231. 31

Estimating NTM-LD numbers – an ongoing challenge x • NTM is not reportable to public health authorities 1 • NTM infections are reported in Queensland, Australia 2 • Demographic changes (e. g. increased life expectancy) 3 • Changing exposure conditions (e. g. popularity of showers) 4 • Varying prevalence of diseasepromoting factors (e. g. immunosuppression) 5 • More than 172 NTM species described 6 • Detection of NTM does not equate to a disease 7 • Awareness of NTM varies between countries 8 • NTM diagnostic methods used are variable between countries 8 The prevalence of NTM-LD is therefore likely to be underestimated 7 NTM, non-tuberculous mycobacteria; NTM-LD, non-tuberculous mycobacterial lung disease. 1. Mc. Shane PJ, Glassroth J. Chest 2015; DOI: 10. 1378/chest. 15 -0458; 2. Chou MP, et al. BMC Infect Dis 2014; 14: 279; 3. Stout JE, et al. Int J Infect Dis 2016; 45: 123 -3 4. Thomson 2013 J Clin Microbiol 51 (9), 3006 -11; 5. Henkle E, Winthrop K. Clin Chest Med 2015; 36: 91 -9; 6. Faria S, et al. J Pathog 2015; 2015: 809014; 7. van der Werf MJ, et al. BMC Infect Dis 2014; 14: 62; 8. personal communication by scientific committee. 32

Impact of disease NP-FR-00023 33

Quality of life is significantly reduced in patients with NTM-LD Mean SF-36 scores Health-related quality of life in 51 patients with NTM-LD from an ambulatory clinic in Canada (SF-36)† 100 90 80 70 60 50 40 30 20 10 0 * * * Normal population * *p<0. 001 Physical functioning Bodily pain General health perceptions Social functioning Most SF-36 scores were ≤ 10 points below the population-based norms, demonstrating that patients with NTM-LD have a reduction in their quality of life, associated with their lung function †Study period 08/2008 -07/2009. NTM-LD, non-tuberculous mycobacterial lung disease; SF-36, medical outcomes short-form – 36 version 2. Mehta M, Marras TK. Respir Med 2011; 105: 1718 -25. 34

NTM-LD can be a progressive, and ultimately lethal, lung disease If left untreated, MAC lung disease can result in extensive cavitary lung destruction and respiratory failure 2 NTM-LD can become a progressive lung disease 1, 2 Cystic fibrosis Decline in lung function among patients with NTM-LD (n=68)*3 • • Mean reduction in FEV 1 was 48 m. L/year Normal range = 28. 4 -35. 6 m. L/year Impaired health-related quality of life in patients with NTM-LD is significantly associated with decline in lung function 4 * Study from Taiwan (01/2000 – 04/2011). FEV 1, forced expiratory volume in 1 sec; MAC, Mycobacterium avium complex; NTM-LD, non-tuberculous mycobacterial lung disease. 1. Weiss CH, Glassroth J. Expert Rev Respir Med 2012; 6: 597 -612; 2. Griffith DE, et al. Am J Respir Crit Care Med 2007; 175: 367 -416; 3. Lee MR, et al. PLo. S One 2013; 8: e 58214; 4. Mehta M, Marras TK. Respir Med 2011; 105: 1718 -25. 35

Increasing hospitalization rate for NTM-LD Rate per 100, 000 population 1. 5 Annual age-adjusted hospitalization rate for NTM-LD in Germany 1 Female Male All 0. 5 0 2005 2006 2007 2008 Year 2009 2010 2011 From 2005 to 2011, the annual number of NTM-LD-associated hospitalizations in Germany increased by 4. 9% on average (p<0. 00001) NTM-LD, non-tuberculous mycobacterial lung disease. Adapted from Ringshausen FC, et al. BMC Infect Dis 2013; 13: 231. 36
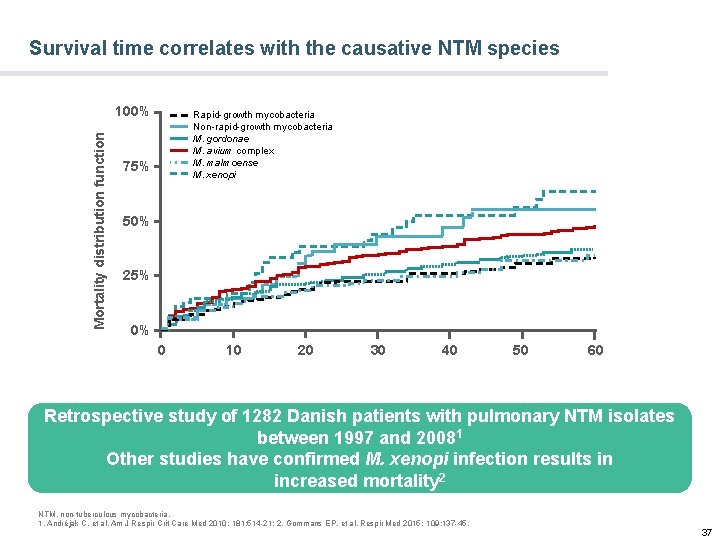
Survival time correlates with the causative NTM species Mortality distribution function 100% Rapid-growth mycobacteria Non-rapid-growth mycobacteria M. gordonae M. avium complex M. malmoense M. xenopi 75% 50% 25% 0% 0 10 20 30 40 50 60 Retrospective study of 1282 Danish patients with pulmonary NTM isolates between 1997 and 20081 Other studies have confirmed M. xenopi infection results in increased mortality 2 NTM, non-tuberculous mycobacteria. 1. Andréjak C, et al. Am J Respir Crit Care Med 2010; 181: 514 -21; 2. Gommans EP, et al. Respir Med 2015; 109: 137 -45. 37
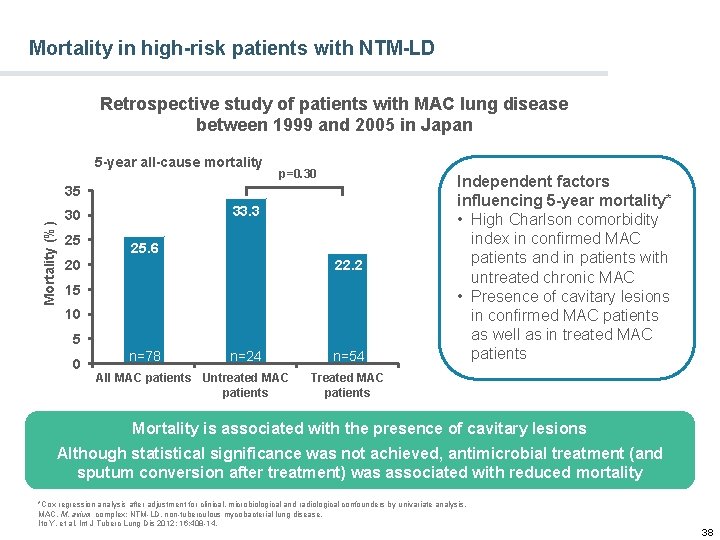
Mortality in high-risk patients with NTM-LD Retrospective study of patients with MAC lung disease between 1999 and 2005 in Japan 5 -year all-cause mortality p=0. 30 Mortality (%) 35 33. 3 30 25 25. 6 20 22. 2 15 10 5 0 n=78 n=24 All MAC patients Untreated MAC patients n=54 Independent factors influencing 5 -year mortality* • High Charlson comorbidity index in confirmed MAC patients and in patients with untreated chronic MAC • Presence of cavitary lesions in confirmed MAC patients as well as in treated MAC patients Treated MAC patients Mortality is associated with the presence of cavitary lesions Although statistical significance was not achieved, antimicrobial treatment (and sputum conversion after treatment) was associated with reduced mortality *Cox regression analysis after adjustment for clinical, microbiological and radiological confounders by univariate analysis. MAC, M. avium complex; NTM-LD, non-tuberculous mycobacterial lung disease. Ito Y, et al. Int J Tuberc Lung Dis 2012; 16: 408 -14. 38
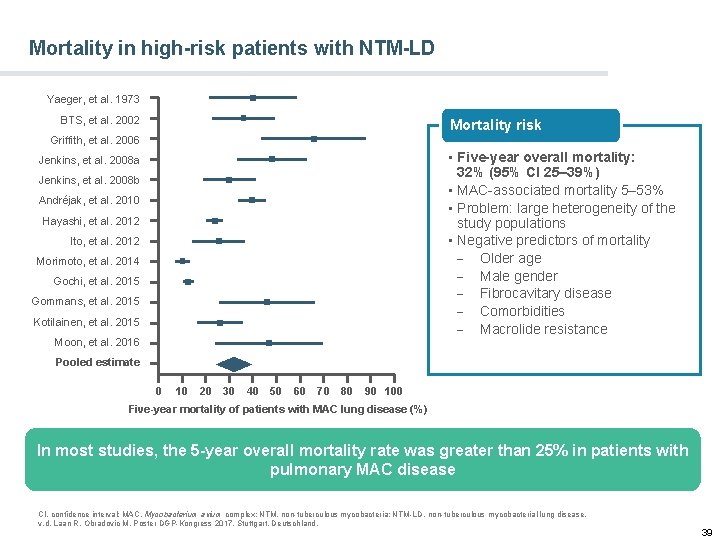
Mortality in high-risk patients with NTM-LD Yaeger, et al. 1973 BTS, et al. 2002 Mortality risk Griffith, et al. 2006 • Five-year overall mortality: 32% (95% CI 25– 39%) • MAC-associated mortality 5– 53% • Problem: large heterogeneity of the study populations • Negative predictors of mortality - Older age - Male gender - Fibrocavitary disease - Comorbidities - Macrolide resistance Jenkins, et al. 2008 a Jenkins, et al. 2008 b Andréjak, et al. 2010 Hayashi, et al. 2012 Ito, et al. 2012 Morimoto, et al. 2014 Gochi, et al. 2015 Gommans, et al. 2015 Kotilainen, et al. 2015 Moon, et al. 2016 Pooled estimate 0 10 20 30 40 50 60 70 80 90 100 Five-year mortality of patients with MAC lung disease (%) In most studies, the 5 -year overall mortality rate was greater than 25% in patients with pulmonary MAC disease CI, confidence interval; MAC, Mycobacterium avium complex; NTM, non-tuberculous mycobacteria; NTM-LD, non-tuberculous mycobacterial lung disease. v. d. Laan R, Obradovic M, Poster DGP-Kongress 2017, Stuttgart, Deutschland. 39
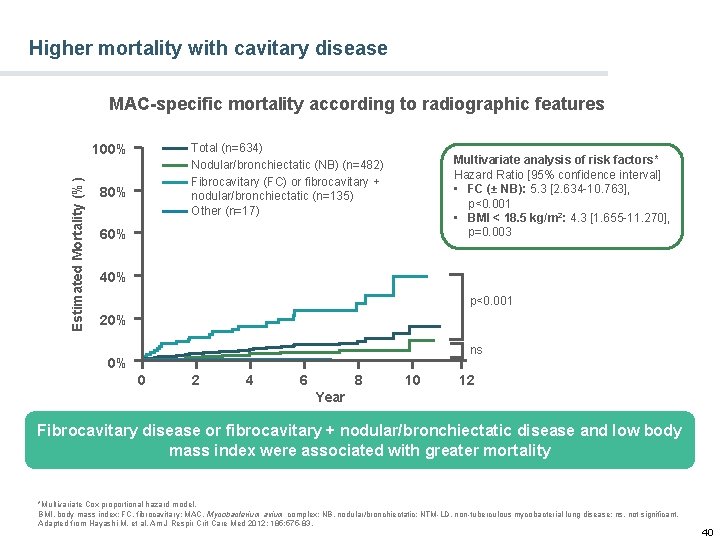
Higher mortality with cavitary disease MAC-specific mortality according to radiographic features Total (n=634) Nodular/bronchiectatic (NB) (n=482) Fibrocavitary (FC) or fibrocavitary + nodular/bronchiectatic (n=135) Other (n=17) Estimated Mortality (%) 100% 80% Multivariate analysis of risk factors* Hazard Ratio [95% confidence interval] • FC (± NB): 5. 3 [2. 634 -10. 763], p<0. 001 • BMI < 18. 5 kg/m 2: 4. 3 [1. 655 -11. 270], p=0. 003 60% 40% p<0. 001 20% ns 0% 0 2 4 6 8 10 12 Year Fibrocavitary disease or fibrocavitary + nodular/bronchiectatic disease and low body mass index were associated with greater mortality *Multivariate Cox proportional hazard model. BMI, body mass index; FC, fibrocavitary; MAC, Mycobacterium avium complex; NB, nodular/bronchiectatic; NTM-LD, non-tuberculous mycobacterial lung disease; ns, not significant. Adapted from Hayashi M, et al. Am J Respir Crit Care Med 2012; 185: 575 -83. 40

Summary • • NTM are related to M. tuberculosis, 1 however are not obligate pathogens and can survive in the environment, including water distribution systems 2 – NTM can be classified based on their growth rate 1 The mycobacterial outer membrane supports dissemination and adherence, helping to form biofilms 2 Different species of NTM have different clinical relevance 3, 4 Pulmonary disease is the most common form of NTM disease, with both nodular/bronchiectatic and fibrocavitary forms being observed 5 Risk factors for development of NTM include geographic location, 6 genetic factors 7– 9 and underlying lung conditions 1, 10 NTM diagnosis has become more frequent – this may be due to increased awareness 10, 11 – Disease prevalence between countries is also variable 12 NTM is a progressive lung disease 13, impacting patient quality of life 14 – Patients with fibrocavitary disease are at higher risk of mortality than those with nodular/bronchiectatic disease 15 NTM, non-tuberculous mycobacteria. 1. Fedrizzi T, et al. Sci Rep 2017; 7: 45258; 2. Falkinham JO, 3 rd. J Appl Microbiol 2009; 107: 356 -67; 3. van Ingen J, et al. Thorax 2009; 64: 502 -6; 4. Schönfeld N, et al. Pneumologie 2013; 67: 605 -33; 5. Griffith DE, et al. Am J Respir Crit Care Med 2007; 6. Adjemian J, et al. Am J Respir Crit Care Med 2012; 186: 553 -8; 7. Szymanski EP, et al. Am J Respir Crit Care Med 2015; 192: 618 -28; 8. Kim RD, et al. Am J Respir Crit Care Med 2008; 178: 1066 -74; 9. Colombo RE, et al. Chest 2010; 137: 629 -34; 10. Johnson MM, Odell JA. J Thorac Dis 2014; 6: 210 -2; 11. Thomson RM, et al. Emerg Infect Dis 2010; 16: 1576 -83; 12. Hoefsloot W, et al. Eur Respir J 2013; 42: 1604 -13; 13. Weiss CH, Glassroth J. Expert Rev Respir Med 2012; 6: 597 -612; 14. Mehta M, Marras TK. Respir Med 2011; 105: 1718 -25; 15. Hayashi M, et al. Am J Respir Crit Care Med 2012; 185: 575 -83. 41

Glossary • Alveolar proteinosis Rare lung disease characterised by abnormal accumulation of surfactant lipoprotein in the alveoli of the lungs • • Biofilm A group of microorganisms that are stuck together and adhere to a surface comprised of extracellular polymeric substances • Charlson comorbidity index System with 19 categories used to predict 10 -year mortality • EU 5 countries France, Germany, Italy, Spain, United Kingdom • Lady Windermere syndrome Nodular/bronchiectatic form of NTM‑LD, particularly associated with tall, thin postmenopausal women NTM-LD, non-tuberculous mycobacterial lung disease. • • Orphan disease Life-threatening or chronically debilitating disease affecting at a maximum of 5 in 10, 000 people in the EU, where marketing of medicines to treat the disease (potential orphan drugs) would be unlikely to justify investment needed for its development Pneumoconiosis Disease of the lungs caused by inhalation of dust Silicosis Disease of the lungs caused by inhalation of crystalline silica dust 42
- Slides: 42