NONSPORE FORMING GRAMPOSITIVE RODS Corynebacterium diphtheriae Listeria monocytogenes

NON–SPORE FORMING GRAMPOSITIVE RODS Corynebacterium diphtheriae Listeria monocytogenes Dr. Wasan Abdulilah Bakir

Bacterial shape Cocci Bacilli Gram +ve Gram -ve Enterobacteriaceae Gram –ve Neisseria spp. Gram +ve Staphylococcus Streptococcus Spore forming: 1. Bacillus spp. (aerobic) 2. Clostridium spp. (anaerobic) Non - spore forming: 1. Corynebacteria spp. 2. Listeria spp.
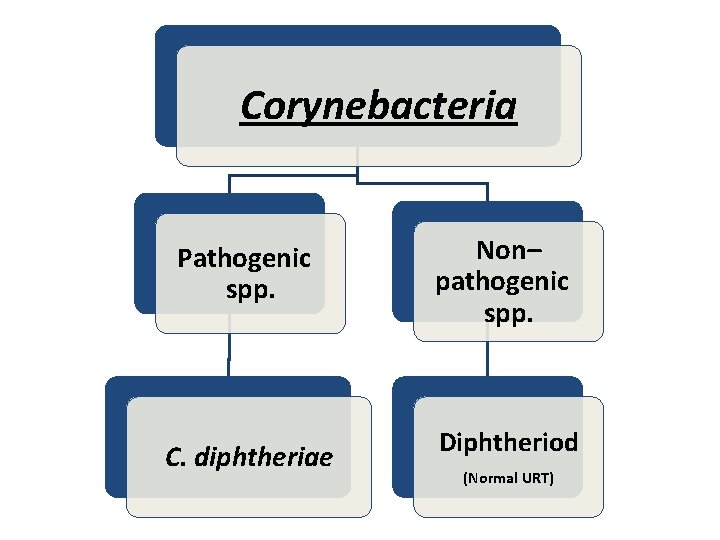
Corynebacteria Pathogenic spp. Non– pathogenic spp. C. diphtheriae Diphtheriod (Normal URT)

C. diphtheriae It is also known as the Klebs - Löffler bacilli

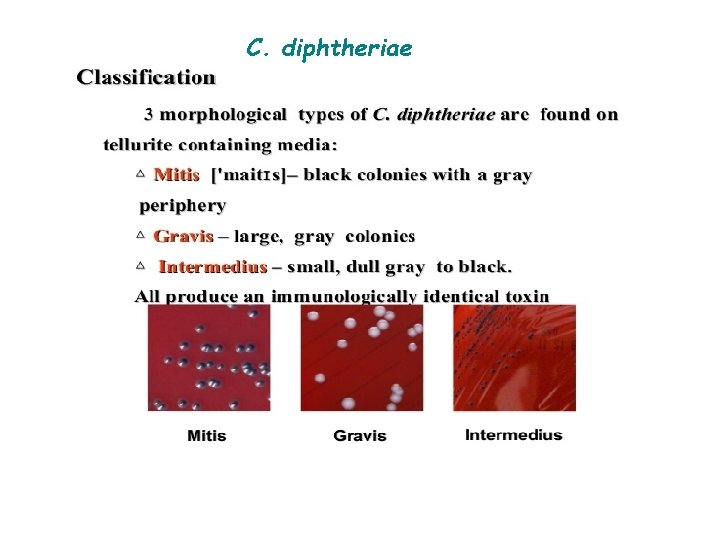
Corynebacterium diphtheriae C. diphtheriae: causes diphtheria. Other Corynebacterium spp. (> 100 species) and related genera may cause opportunistic infections. Important Properties o Gram-positive o irregularly-shaped rods. "Club shaped". (wider at one end), and are arranged in palisades or in V- or Lshaped formations. o Metachromatic granules (often near the poles) give the rod a beaded appearance.

C. diphtheriae o Grow aerobically on most media. o Corynebacteria grow on Löffler's serum medium more readily than other respiratory pathogens, and show typical morphology in smears. o Non-motile o Non capsulated.
C. diphtheriae

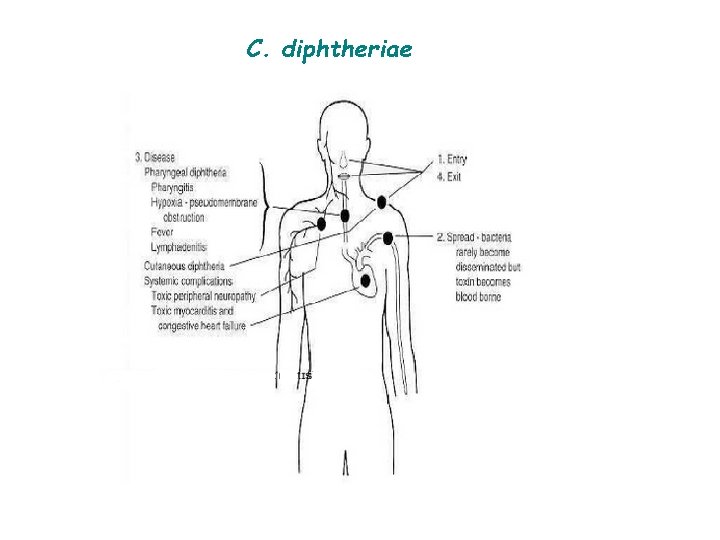
C. diphtheriae Transmission o Humans are the only natural host of C. diphtheriae. Both toxigenic and non toxigenic organisms reside in the upper respiratory tract. o It is spread by droplets or by contact to susceptible individuals. o The bacilli then grow on mucous membranes or in skin abrasions, and those that are toxigenic start producing toxin.

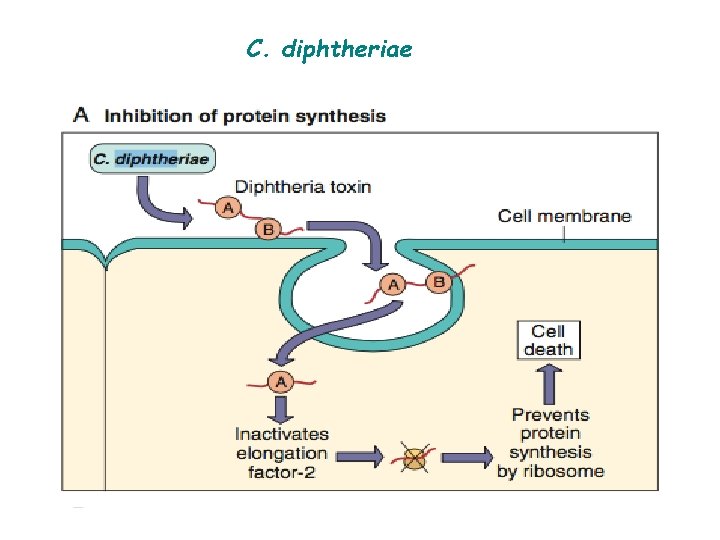
C. diphtheriae Pathogenesis Although exotoxin production is essential for pathogenesis, the organism must first establish and maintain itself in the throat. The principal human pathogen of the genus Corynebacterium is C diphtheriae, the causative agent of respiratory or cutaneous diphtheria. Diphtheria toxin The toxin is a single polypeptide with two functional fragment. • The binding (B) fragments mediates binding of the toxin to glycoprotein receptors on the cell membrane. • The active (A) possesses enzymatic activity that cleaves nicotinamide from nicotinamide adenine dinucleotide (NAD) and transfers the remaining ADP-ribose to elongation factor-2 (EF-2), thereby inactivating it.

C. diphtheriae o This toxin binds to a receptor (heparin-binding epidermal growth factor) on the surface of many eukaryotic cells, particularly heart and nerve cells, and results in inhibition of polypeptide chain elongation by ribosylation of the elongation factor EF-2. o triggers the entry of the toxin into the cell through receptormediated endocytosis. Acidification within a developing endosome leads to creation of a protein channel that facilitates movement of Fragment A into the host cell cytoplasm. o The DNA that codes for diphtheria toxin is part of the DNA of a temperate bacteriophage called beta phage. Bacteriophage: a virus that infects and replicates within bacteria. Bacteriophages are much used in genetic research.
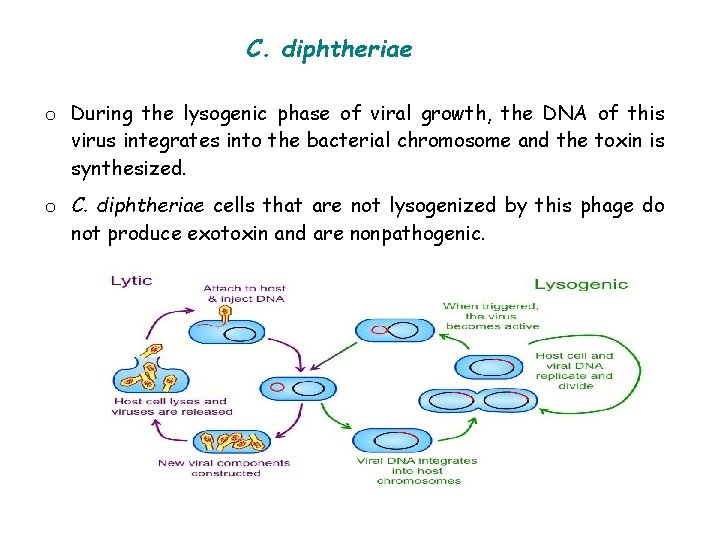
C. diphtheriae o During the lysogenic phase of viral growth, the DNA of this virus integrates into the bacterial chromosome and the toxin is synthesized. o C. diphtheriae cells that are not lysogenized by this phage do not produce exotoxin and are nonpathogenic.
C. diphtheriae
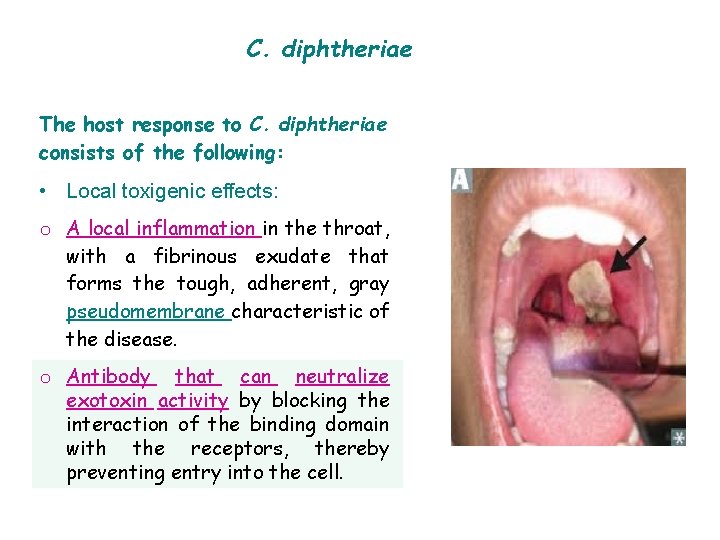
C. diphtheriae The host response to C. diphtheriae consists of the following: • Local toxigenic effects: o A local inflammation in the throat, with a fibrinous exudate that forms the tough, adherent, gray pseudomembrane characteristic of the disease. o Antibody that can neutralize exotoxin activity by blocking the interaction of the binding domain with the receptors, thereby preventing entry into the cell.
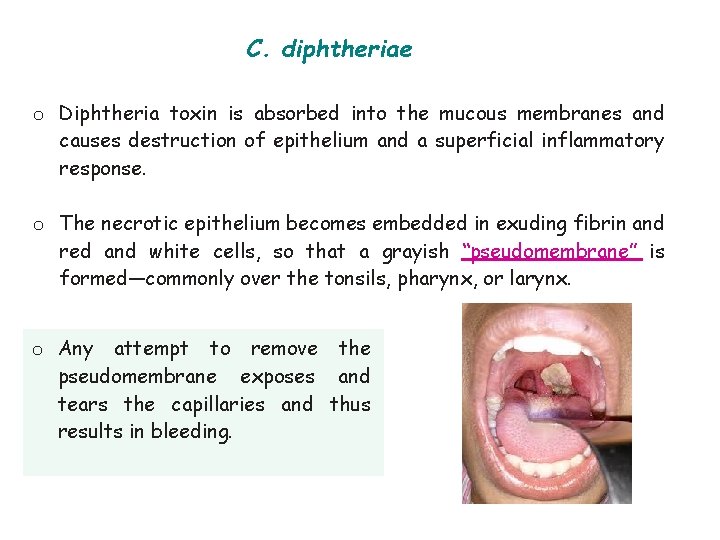
C. diphtheriae o Diphtheria toxin is absorbed into the mucous membranes and causes destruction of epithelium and a superficial inflammatory response. o The necrotic epithelium becomes embedded in exuding fibrin and red and white cells, so that a grayish “pseudomembrane” is formed—commonly over the tonsils, pharynx, or larynx. o Any attempt to remove the pseudomembrane exposes and tears the capillaries and thus results in bleeding.
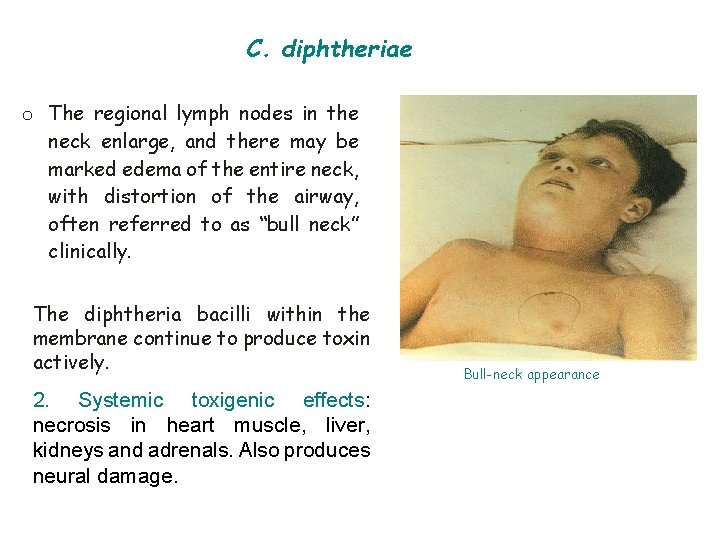
C. diphtheriae o The regional lymph nodes in the neck enlarge, and there may be marked edema of the entire neck, with distortion of the airway, often referred to as “bull neck” clinically. The diphtheria bacilli within the membrane continue to produce toxin actively. 2. Systemic toxigenic effects: necrosis in heart muscle, liver, kidneys and adrenals. Also produces neural damage. Bull-neck appearance
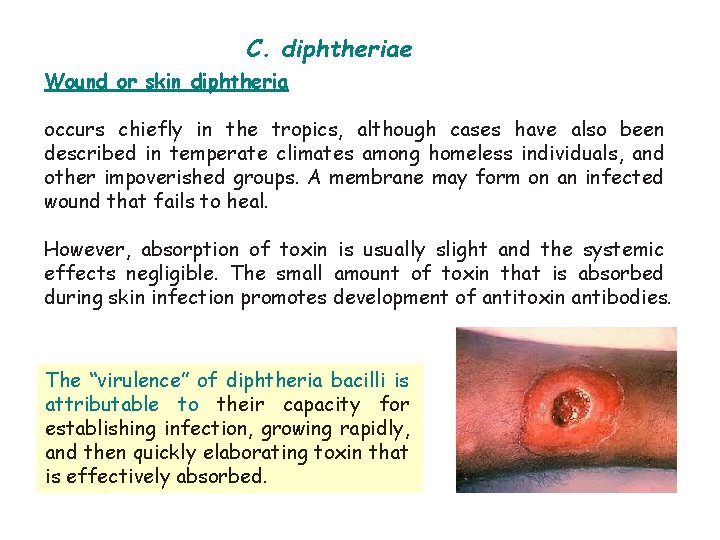
C. diphtheriae Wound or skin diphtheria occurs chiefly in the tropics, although cases have also been described in temperate climates among homeless individuals, and other impoverished groups. A membrane may form on an infected wound that fails to heal. However, absorption of toxin is usually slight and the systemic effects negligible. The small amount of toxin that is absorbed during skin infection promotes development of antitoxin antibodies. The “virulence” of diphtheria bacilli is attributable to their capacity for establishing infection, growing rapidly, and then quickly elaborating toxin that is effectively absorbed.

C. diphtheriae Clinical Findings When diphtheritic inflammation begins in the respiratory tract: sore throat and low-grade fever usually develop. Prostration and dyspnea soon follow because of the obstruction caused by the membrane. This obstruction may even cause suffocation if not promptly relieved by intubation or tracheostomy. Irregularities of cardiac rhythm indicate damage to the heart. Later, there may be difficulties with vision, speech, swallowing, or movement of the arms or legs.
C. diphtheriae

C. diphtheriae Diagnosis o Swab from the nose, throat, or other suspected lesions must be obtained before antimicrobial drugs are administered. Swabs should be collected from beneath any visible membrane. The swab should then be placed in semisolid transport media. o Smears stained with alkaline methylene blue or Gram stain show beaded rods in typical arrangement.
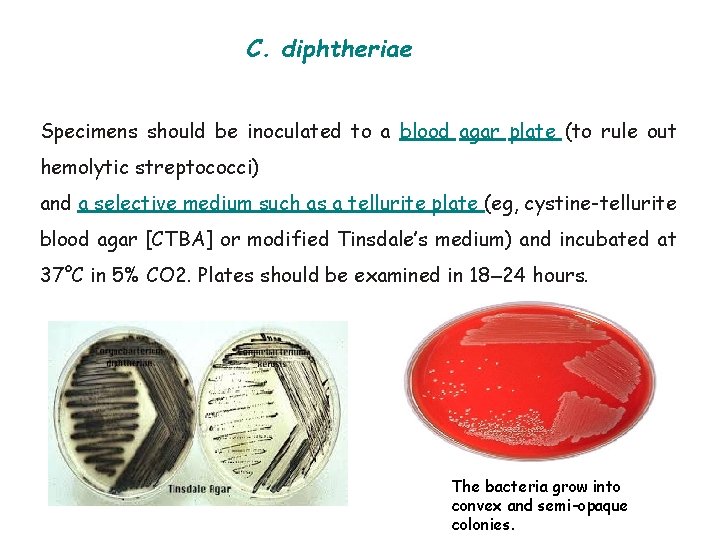
C. diphtheriae Specimens should be inoculated to a blood agar plate (to rule out hemolytic streptococci) and a selective medium such as a tellurite plate (eg, cystine-tellurite blood agar [CTBA] or modified Tinsdale’s medium) and incubated at 37°C in 5% CO 2. Plates should be examined in 18– 24 hours. The bacteria grow into convex and semi-opaque colonies.

C. diphtheriae In 36– 48 hours, the colonies on tellurite medium are sufficiently definite for recognition of C diphtheriae. On cystine tellurite agar, the colonies are black with a brown halo. Note: Specific treatment must never be delayed for laboratory reports if the clinical pictures strongly suggestive of diphtheria.

C. diphtheriae C diphtheriae isolate should be subjected to testing for toxigenicity. Such tests are performed only in reference public health laboratories. There are several methods, as follows: 1. Elek immunoprecipitation method A filter paper strip containing antitoxin is placed on an agar plate. The cultures to be tested (at least 10 colonies should be chosen) for toxigenicity. After 48 hours of incubation, the antitoxin diffusing from the paper strip has precipitated the toxin diffusing from toxigenic cultures and has resulted in precipitin bands between the strip and the bacterial growth. Toxigenicity test

C. diphtheriae

C. diphtheriae 2. Polymerase chain reaction (PCR)–based methods have been described for detection of the diphtheria toxin gene (tox). PCR assays for tox can also be used directly on patient specimens before culture results are available. 3. Enzyme-linked immunosorbent assays can be used to detect diphtheria toxin from clinical C diphtheria isolates. 4. An immunochromatographic strip assay allows detection of diphtheria toxin in a matter of hours. This assay is highly sensitive. The latter two assays are not widely available.

C. diphtheriae Treatment o The treatment of choice is antitoxin. The antitoxin should be given intravenously on the day the clinical diagnosis of diphtheria is made and need not be repeated. Intramuscular injection may be used in mild cases. Diphtheria antitoxin will only neutralize circulating toxin that is not bound to tissue. o Penicillin G or erythromycin is also recommended. Antibiotics inhibit growth of the organism, reduce toxin production, and decrease the incidence of chronic carriers.

C. diphtheriae Prevention o Diphtheria is rare because children are immunized with diphtheria toxoid (usually given as a combination of diphtheria toxoid, tetanus toxoid, and a cellular pertussis vaccine, often abbreviated as DTa. P). o Diphtheria toxoid is prepared by treating the exotoxin with formaldehyde. This treatment inactivates the toxic effect but leaves the antigenicity intact. o Immunization does not prevent nasopharyngeal carriage of the organism.

C. diphtheriae Skin test = Schick test To determine whether the patient is susceptible to diphtheria infection or not by detecting the presence or absence of antibodies. Procedures: 1. 0. 1 ml of toxin is injected intradermally in one arm. 2. 0. 1 ml of heated (inactivated) toxin as a control is injected in the other arm. 3. Read after 24 -48 hours. Positive: (Susceptible) Redness and swelling that increases for several days and then fades, leaving brownish pigmented area. The control site shows no reaction. Negative: (Not susceptible) Neither injection site shows any reaction).


LISTERIA MONOCYTOGENES There are several species in the genus Listeria. Of these, L monocytogenes is important as a cause of a wide spectrum of disease in animals and humans. L monocytogenes is capable of growing and surviving over a wide range of environmental conditions. It can survive at refrigerator temperatures (4°C), under conditions of low p. H and high salt conditions. Therefore, it is able to overcome food preservation and safety barriers, making it an important foodborne pathogen.
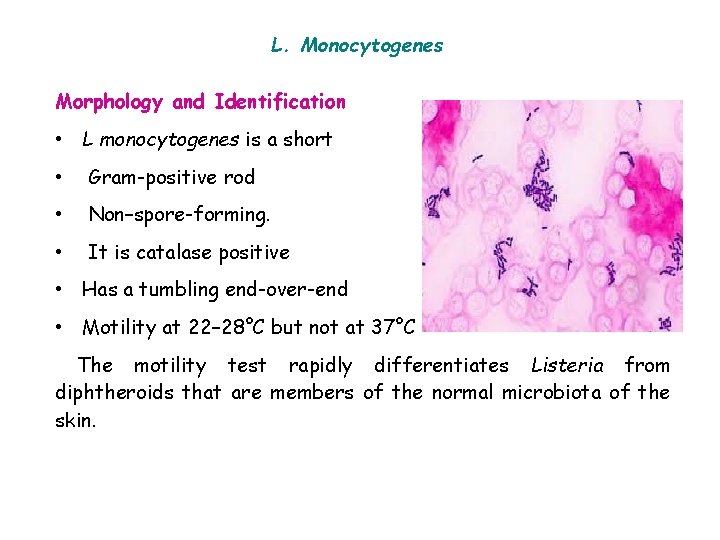
L. Monocytogenes Morphology and Identification • L monocytogenes is a short • Gram-positive rod • Non–spore-forming. • It is catalase positive • Has a tumbling end-over-end • Motility at 22– 28°C but not at 37°C The motility test rapidly differentiates Listeria from diphtheroids that are members of the normal microbiota of the skin.

Pathogenesis L. Monocytogenes Listeria infections occur primarily in two clinical settings: (1) in the fetus or in a newborn as a result of transmission across the placenta or during delivery (2) in pregnant women and immunosuppressed adults, especially renal transplant patients. (Note that pregnant women have reduced cell-mediated immunity during the third trimester. )

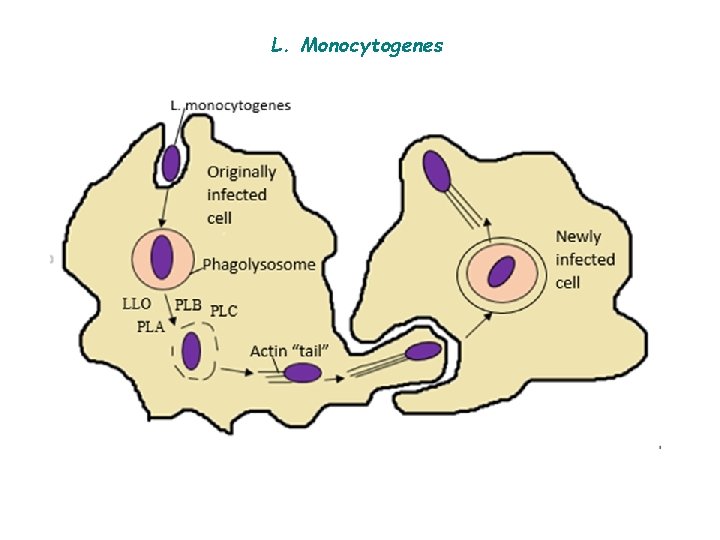
L. Monocytogenes L monocytogenes enters the body through the gastrointestinal tract after ingestion of contaminated foods such as cheese, fruit, or vegetables. The organism has several adhesin proteins (Ami, Fbp A, and flagellin proteins) that facilitate bacterial binding to the host cells and that contribute to virulence. It has cell wall surface proteins called internalins A and B that interact with E-cadherin, a receptor on epithelial cells, promoting phagocytosis into the epithelial cells. After phagocytosis, the bacterium is enclosed in a phagolysosome, where the low p. H activates the bacterium to produce listeriolysin O.
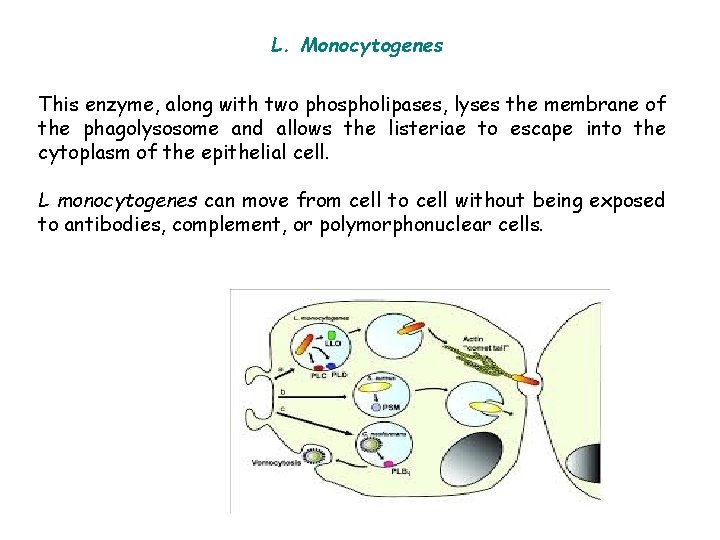
L. Monocytogenes This enzyme, along with two phospholipases, lyses the membrane of the phagolysosome and allows the listeriae to escape into the cytoplasm of the epithelial cell. L monocytogenes can move from cell to cell without being exposed to antibodies, complement, or polymorphonuclear cells.
L. Monocytogenes

L. Monocytogenes Virulence factors 1. Growth at low temperature A property of L. Monocytogenes that affects its food – borne transmission is the ability to multiply at low temp. (4°C), So, listeriosis is associated with ingestion of milk, meat or vegetable products. 2. Motility Like V. cholerae, Listeria which is also acquired by ingestion are attach to the intestinal mucosa. Although Listeria actively motile by means of peritrichous flagella at room temp. (20 – 25°C), but the organisms do not synthesize flagella at body temp. (37°C).

L. Monocytogenes 3. Adhesion 4. Listeriolysin O Hemolysin, pore forming toxin, escape from phagocytic endosome to cytoplasm. Required for intracellular growth. 5. Phospholipase Also involve in the escape of microorganism from phagocytic endosome to cytoplasm. 6. Internalins protein A and B

L. Monocytogenes Clinical Findings Infection during pregnancy can cause abortion, premature delivery, or sepsis during the peripartum period. Newborns infected at the time of delivery can have acute meningitis. The bacteria reach the meninges via the bloodstream (bacteremia). The infected mother either is asymptomatic or has an influenza like illness. L. monocytogenes infections in immunocompromised adults can be either sepsis or meningitis.

L. Monocytogenes Gastroenteritis caused by L. monocytogenes is characterized by watery diarrhea, fever, headache, myalgias, and abdominal cramps but little vomiting. Outbreaks are usually caused by contaminated dairy products, but undercooked meats such as chicken and hot dogs and readyto-eat foods such as coleslaw have also been involved. Laboratory Diagnosis
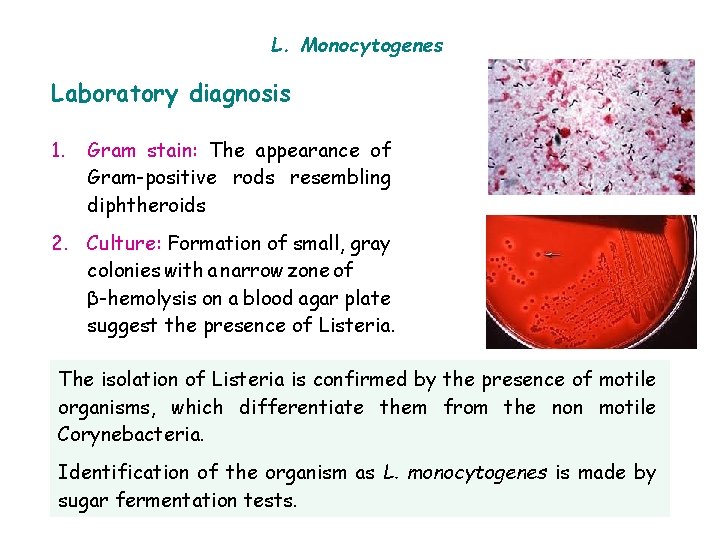
L. Monocytogenes Laboratory diagnosis 1. Gram stain: The appearance of Gram-positive rods resembling diphtheroids 2. Culture: Formation of small, gray colonies with a narrow zone of β-hemolysis on a blood agar plate suggest the presence of Listeria. The isolation of Listeria is confirmed by the presence of motile organisms, which differentiate them from the non motile Corynebacteria. Identification of the organism as L. monocytogenes is made by sugar fermentation tests.

L. Monocytogenes Treatment of invasive disease, such as meningitis and sepsis, consists of trimethoprim-sulfamethoxazole. o Combinations, such as ampicillin and gentamicin or ampicillin and trimethoprim-sulfamethoxazole, can also be used. o Resistant strains are rare. Listeria gastroenteritis typically does not require treatment.

L. Monocytogenes Prevention o Prevention is difficult because there is no immunization. o Limiting the exposure of pregnant women and immunosuppressed patients to potential sources such as farm animals, unpasteurized milk products, and raw vegetables is recommended. o Trimethoprim-sulfamethoxazole given to immunocompromised patients to prevent Pneumocystis pneumonia can also prevent listeriosis.

Thank you
- Slides: 42