Nonmotor symptom complex and comorbidities Parkinsons disease 1

Non-motor symptom complex and comorbidities Parkinson’s disease 1

Non-motor symptom complex of Parkinson’s disease • Non-motor symptoms are reported by nearly all patients with Parkinson’s disease (PD)1 -3 • The PRIAMO study interviewed 1, 076 individuals being treated for PD at several centres across Italy: 1 • Almost all patients with PD reported non-motor symptoms (98. 6%) • A mean number of 7. 8 non-motor symptoms per patient were reported at the time of the interview • Psychiatric symptoms were the most frequent non-motor symptoms (66. 8%) • Non-motor symptoms have a significant negative impact on the quality of life of patients with PD, in some cases exceeding the impact of motor symptoms 4 • However, the range of non-motor symptoms can be present in 68– 88% of comparably aged individuals, often making it difficult to attribute the non-motor symptoms explicitly to PD (or its treatment)3, 5 PRIAMO=Parkinson and non-motor symptoms study 1. Barone et al. Mov Disord 2009; 24(11): 1641– 1649; 2. Goldman & Holden. Curr Treat Options Neurol 2014; 16(3): 281; 3. Pfeiffer. Parkinsonism Relat Disord 2016; 22: S 119–S 122; 4. Martinez-Martin et al. Mov Disord 2011; 26(3): 399– 406; 5. Lim & Lang. Mov Disord 2010; 25(Suppl 1): S 123– 130 2
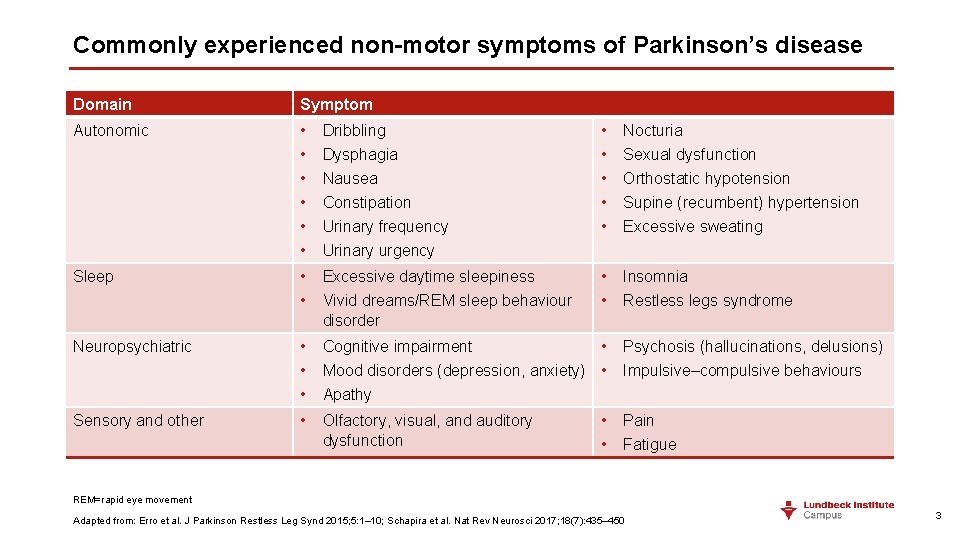
Commonly experienced non-motor symptoms of Parkinson’s disease Domain Symptom Autonomic • • • Dribbling Dysphagia Nausea • • • Nocturia Sexual dysfunction Orthostatic hypotension • Constipation • Supine (recumbent) hypertension • Urinary frequency • Excessive sweating • Urinary urgency Sleep • • Excessive daytime sleepiness Vivid dreams/REM sleep behaviour disorder • • Insomnia Restless legs syndrome Neuropsychiatric • • • Cognitive impairment • Mood disorders (depression, anxiety) • Apathy Psychosis (hallucinations, delusions) Impulsive–compulsive behaviours Sensory and other • Olfactory, visual, and auditory dysfunction Pain Fatigue • • REM=rapid eye movement Adapted from: Erro et al. J Parkinson Restless Leg Synd 2015; 5: 1– 10; Schapira et al. Nat Rev Neurosci 2017; 18(7): 435– 450 3

The impact of non-motor symptoms on quality of life • A Singaporean study assessed the impact of PD symptoms on the quality of life of approximately 200 patients with PD 1 • A questionnaire study in the UK assessed the effect of non-motor symptoms on quality of life in 158 patients with PD 2 • Motor symptoms, as measured by the UPDRS scale, were not significantly correlated with quality of life, as measured by the PDQ scale, in the multivariate analysis 1 • Many non-motor symptoms correlated with quality of life scores: 2 • Depression (p<0. 001) • Anxiety (p<0. 001) • Impaired concentration (p<0. 001) • Memory complaints (p<0. 001) • Sleep disturbance (p=0. 002) • The quality of life of the patients, as measured by the PDQ scale, correlated with the total number of non-motor symptoms observed (p<0. 001)1 The non-motor symptom complex has a significant negative impact on the quality of life of patients with PD 1, 2 PDQ=Parkinson’s Disease Questionnaire; UPDRS=Unified Parkinson’s Disease Rating Scale 1. Prakash et al. Eur J Neurol 2016; 23(5): 854– 860; 2. Duncan et al. Mov Disord 2014; 29(2): 195– 202 4

Cognitive symptoms of Parkinson’s disease 5

Cognition and cognitive impairment in Parkinson’s disease • In PD, there is a spectrum of cognitive dysfunction, ranging from MCI to PD dementia 1 -3 • Ideally, the different forms of cognitive impairment should be defined using a standardised approach and, to this end, the MDS task force has developed diagnostic criteria for MCI 1 • MCI is common in non-demented patients with PD, and its presence is associated with increasing age, disease duration, and • These clinical, cognitive, and functional disease severity 1 criteria were designed to be consistent with the MDS proposed criteria for • MCI also tends to predict the onset of 2 thereby allowing the PD-related dementia, dementia; with time, dementia develops in transition over time between normal the majority of patients with PD and MCI 1, 3 cognition, MCI, and dementia 1 • The diagnostic guidelines should enable clinicians to identify patients at greater risk of developing dementia, who may benefit from early interventions 1 MCI=mild cognitive impairment; MDS=Movement Disorder Society 1. Litvan et al. Mov Disord 2012; 27(3): 349– 356; 2. Emre et al. Mov Disord 2007; 22(12): 1689– 1707; 3. Weil et al. Curr Neurol Neurosci Rep 2018; 18(4): 17 6

MDS diagnostic criteria for MCI in Parkinson’s disease I. Inclusion criteria 1 • • Diagnosis of PD based on the UK PD Brain Bank Criteria • Cognitive deficits on either formal neuropsychological testing or a scale of global cognitive abilities (detailed in Section III) • Cognitive deficits not sufficient to interfere significantly with functional independence, although subtle difficulties on complex functional tasks may be present Gradual decline, in the context of established PD, in cognitive ability reported by either the patient or informant, or observed by the clinician II. Exclusion criteria 1 • Diagnosis of PD dementia based on MDS Task Force proposed criteria 2 • Other primary explanations for cognitive impairment (e. g. , delirium, stroke, major depression, metabolic abnormalities, adverse effects of medication, or head trauma) • Other PD-associated comorbid conditions (e. g. , motor impairment or severe anxiety, depression, excessive daytime sleepiness, or psychosis) that, in the opinion of the clinician, significantly influence cognitive testing III. Specific guidelines for PD–MCI Level I and Level II categories 1 A. Level I (abbreviated assessment) • Impairment on a scale of global cognitive abilities validated for use in PD or • Impairment on at least two tests, when a limited battery of neuropsychological tests is performed (i. e. , the battery includes less than two tests within each of the five cognitive domains or less than five cognitive domains are assessed) B. Level II (comprehensive assessment) • Neuropsychological testing that includes two tests within each of the five cognitive domains (i. e. , attention and working memory, executive, language, memory, and visuospatial) • Impairment on at least two neuropsychological tests, represented by either two impaired tests in one cognitive domain or one impaired test in two different cognitive domains • Impairment on neuropsychological tests may be demonstrated by: • Performance approximately 1 to 2 standard deviations below appropriate norms • Significant decline demonstrated on serial cognitive testing • Significant decline from estimated premorbid levels MCI=mild cognitive impairment; MDS=Movement Disorder Society 1. Litvan et al. Mov Disord 2012; 27(3): 349– 356; 2. Emre et al. Mov Disord 2007; 22(12): 1689– 1707 7
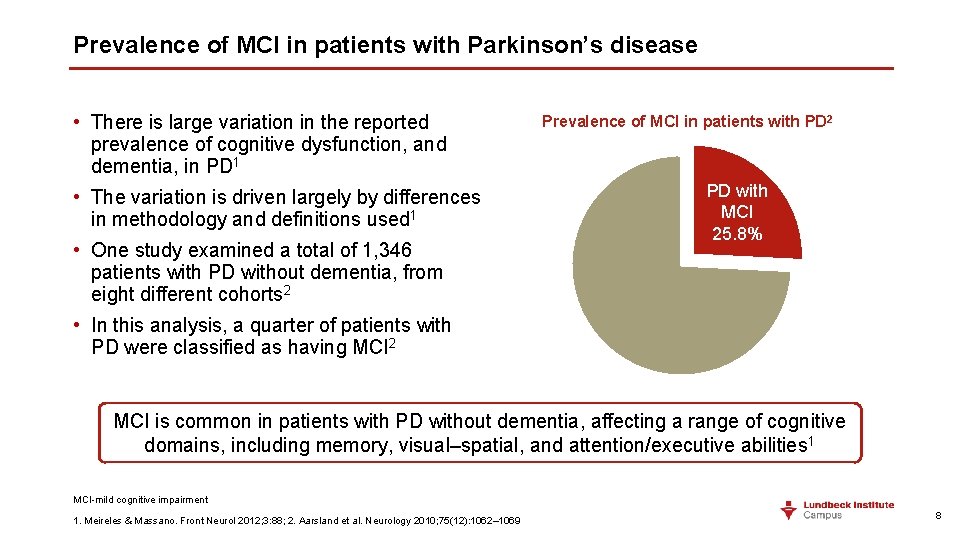
Prevalence of MCI in patients with Parkinson’s disease • There is large variation in the reported prevalence of cognitive dysfunction, and dementia, in PD 1 • The variation is driven largely by differences in methodology and definitions used 1 • One study examined a total of 1, 346 patients with PD without dementia, from eight different cohorts 2 Prevalence of MCI in patients with PD 2 PD with MCI 25. 8% • In this analysis, a quarter of patients with PD were classified as having MCI 2 MCI is common in patients with PD without dementia, affecting a range of cognitive domains, including memory, visual–spatial, and attention/executive abilities 1 MCI-mild cognitive impairment 1. Meireles & Massano. Front Neurol 2012; 3: 88; 2. Aarsland et al. Neurology 2010; 75(12): 1062– 1069 8

Diagnostic criteria for dementia in Parkinson’s disease MDS-proposed criteria for dementia in PD 1 -3 1. Core features: Diagnosis of PD & dementia syndrome 2. Associated clinical features: Impairment of at least 2 of 4 cognitive domains (may be supported by behavioural symptoms) Features which make diagnosis uncertain • Coexistence of any abnormality that could itself cause cognitive impairment, but not cause dementia • Unknown time interval between onset of motor and cognitive symptoms Features which make diagnosis impossible Cognitive and behavioural symptoms presenting as a result of other conditions, for example: • Acute confusion due to systemic diseases/abnormalities or drug intoxication • Major depression according to DSM-IV • Features of ‘probable vascular dementia’ according to NINDS–AIREN • The risk of developing dementia for patients with PD, at any time, is approximately 4– 6 times that for people of a similar age without PD 2 • Dementia also seems to be more prevalent in patients with motor symptoms dominated by postural instability–gait difficulty symptoms, rather than in those for whom tremor is dominant 2 • Drawing from these findings, the MDS task force proposed a simple set of diagnostic criteria that could be used by clinicians without specialist training 1 -3 • The criteria particularly focus on the timing of dementia symptoms; they should follow the onset of motor symptoms by ≥ 1 year 3 • This distinguishes PD-related dementia from dementia with Lewy bodies (DLB), which has a different disease course 1 AIREN=Association Internationale pour la Recherche et l’Enseignement en Neurosciences; DLB=dementia with Lewy bodies; DSM-IV=Diagnostic and Statistical Manual of Mental Disorders, fourth edition; MDS=Movement Disorder Society; NINDS=National Institute of Neurological Disorders and Stroke 1. Poewe et al. Int J Clin Pract 2008; 62(10): 1581– 1587; 2. Emre et al. Mov Disord 2007; 22(12): 1689– 1707; 3. Dubois et al. Mov Disord 2007; 22(16): 2314– 2324 9
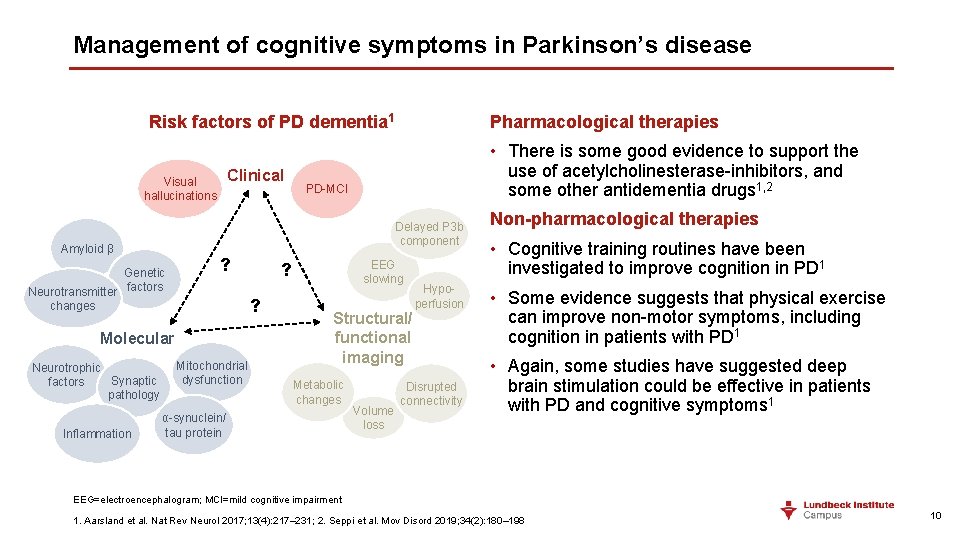
Management of cognitive symptoms in Parkinson’s disease Risk factors of PD dementia 1 Clinical Visual hallucinations Genetic Neurotransmitter factors changes ? PD-MCI Mitochondrial dysfunction EEG slowing ? ? Molecular Inflammation • There is some good evidence to support the use of acetylcholinesterase-inhibitors, and some other antidementia drugs 1, 2 Delayed P 3 b component Amyloid β Neurotrophic Synaptic factors pathology Pharmacological therapies Structural/ functional imaging Metabolic changes α-synuclein/ tau protein Volume loss Hypoperfusion Disrupted connectivity Non-pharmacological therapies • Cognitive training routines have been investigated to improve cognition in PD 1 • Some evidence suggests that physical exercise can improve non-motor symptoms, including cognition in patients with PD 1 • Again, some studies have suggested deep brain stimulation could be effective in patients with PD and cognitive symptoms 1 EEG=electroencephalogram; MCI=mild cognitive impairment 1. Aarsland et al. Nat Rev Neurol 2017; 13(4): 217– 231; 2. Seppi et al. Mov Disord 2019; 34(2): 180– 198 10

“ Cognitive impairment specifically contributes to poorer quality of life, even in early Parkinson’s disease Lawson et al. Parkinsonism Relat Disord 2014; 20(10): 1071– 1075 11

Mood disorders 12

The spectrum of mood disorders in Parkinson’s disease • The most common mood disorders in PD are: 1 • Depression • Anxiety • Many studies have used neuroimaging techniques in an attempt to identify brain regions linked to the above symptoms, including limbic and cortical areas of the brain, and the basal ganglia 2, 3 • These studies have identified regions of the brain in connection with mood disorders – some of which are implicated also in the pathology of PD 2, 4 • However, much more research is needed to translate experimental observations into viable treatments 2 1. Tan. Parkinsonism Relat Disord 2012; 18(Suppl 1): S 74–S 76; 2. Wen et al. Eur J Neurol 2016; 23(6): 1001– 1019; 3. Schapira et al. Nat Rev Neurosci 2017; 18(7): 435– 450; 4. Aarsland et al. Nat Rev Neurol 2011; 8(1): 35– 47 13

Depression in Parkinson’s disease • Depression in PD may be due to multiple factors; it could be a psychological reaction to having a chronic and progressive disease, and it could also share the same biological or causal roots in the brain as other symptoms of PD 1 • One meta-analysis found that the prevalence of depression in patients with PD was roughly 17%2 • Depression in patients with PD is strongly linked to reduced quality of life, as well as sleep problems and impairments in ADLs 1, 3 Treatment of depression in PD 1 • If the depression is linked to medication ‘OFF’ periods, then optimisation of the PD treatments should be the first priority • Mild depression can be treated with non-pharmacological options, such as counselling, patient support groups or cognitive behavioural therapy • Controlled studies suggest that pharmacological treatment with DAs, TCAs, SSRIs, and SNRIs may be effective for depression in PD 1, 4 ADL=activity of daily living; DA=dopamine agonist; TCA=tricyclic antidepressant; SNRI=serotonin–noradrenaline reuptake inhibitor; SSRI=selective serotonin reuptake inhibitor 1. Tan. Parkinsonism Relat Disord 2012; 18(Suppl 1): S 74–S 76; 2. Reijnders et al. Mov Disord 2008; 23(2): 138– 189; 3. Schrag. J Neurol Sci 2006; 248(1– 2): 151– 157; 4. Aarsland et al. Nat Rev Neurol 2011; 8(1): 35– 47 14

Anxiety in Parkinson’s disease • Anxiety disorder spectrum in PD: 1 • • Panic attacks Generalised anxiety disorders Social phobias Phobic disorders Agoraphobia Obsessive–compulsive disorder Anxiety disorders not otherwise specified Treatment of anxiety in PD 1 • Anxiety symptoms may occasionally be caused by dopaminergic therapy; therefore, dose reduction or substitution may be beneficial • If the anxiety symptoms are related to medication ‘OFF’ periods, then the PD therapy should be optimised • One study found the lifetime prevalence of • Psychological interventions, such as an anxiety disorder among patients with relaxation techniques, behavioural training, PD to be 49%, with the most common counselling, and massage therapy may diagnosis being ‘anxiety disorder not also be considered otherwise specified’ 2 • Pharmacological treatments include low dose TCAs, certain anxiolytics, and SSRIs SSRI=selective serotonin reuptake inhibitor; TCA=tricyclic antidepressant 1. Tan. Parkinsonism Relat Disord 2012; 18(Suppl 1): S 74–S 76; 2. Pontone et al. Mov Disord 2009; 24(9): 1333– 1338 15

Apathy in Parkinson’s disease • Apathy refers to reduced interest and motivation in goal-directed behaviours, indifference, and flattened affect 1, 2 Treatment of apathy in PD • Diagnosis is difficult, as apathy is also included as a criterion for diagnosis of other conditions, e. g. , depression 3 • Non-pharmacological strategies include providing an individual daily schedule and structure with varied activities to keep patients engaged and active 1 • Reported prevalence rates for apathy in PD range from 17– 70%1, 2 • Apathy has been linked to more severe cognitive impairment, and the presence of apathy predicts cognitive decline in patients with PD 1, 4 • If the apathy is secondary to depression, then the depression should be treated 1 • Pharmacological strategies are not well studied in PD, but include acetylcholinesterase inhibitors, dopamine agonists, psychostimulants, and certain wakefulness-promoting agents 1, 5, 6 1. Tan. Parkinsonism Relat Disord 2012; 18(Suppl 1): S 74–S 76; 2. Pagonabarraga et al. Lancet Neurol 2015; 14(5): 518– 531; 3. APA. DSM-5. 2013; 4. Dujardin et al. Mov Disord 2009; 24(16): 2391– 2397; 5. Aarsland et al. Mov Disord 2009; 24(15): 2175– 2186; 6. Seppi et al. Mov Disord 2019; 34(2): 180– 198 16

Psychosis and Parkinson’s disease 17

Psychosis and Parkinson’s disease • The pathophysiology underlying psychosis in PD is not fully understood, but psychosis and hallucinations can be an adverse event of some dopaminergic and anticholinergic agents 1, 2 • Delusions tend to manifest as paranoia, and occur in roughly 5% of patients with PD 3, 4 • Visual hallucinations occur in approximately 30% of patients with PD 4 • Psychotic symptoms are generally associated with increased morbidity and mortality risk in patients with PD 3 • So-called ‘minor’ hallucinations sometimes seen in PD include: 3 • Transient peripheral field hallucination – patients describe seeing a shadow or an animal, like a dog, in either peripheral field and often turn to see what it is, only to have it disappear • Presence hallucination – these are feelings that there is a person or animal behind, or nearby, but not seen • Illusion – real objects are seen transformed into other shapes, for example a fire hydrant at a distance becomes a man walking a dog Psychosis in PD reduces quality of life for patients, and increases caregiver distress, and is an independent risk factor for nursing home placement and development of dementia 4 1. Samudra et al. Drugs Aging 2016; 33(12): 855– 863; 2. Taddei et al. Parkinsons Dis 2017; 2017: 3256542; 3. Friedman. Behav Neurol 2013; 27(4): 469– 477; 4. Fénelon & Alves. J Neurol Sci 2010; 289(1– 2): 12– 17 18

The prevalence of psychosis in patients with Parkinson’s disease • The reported prevalence of psychosis among patients with PD varies widely, because of: 1 • Differences in methodology • Lack of agreed definitions • Different effects of medications • In one US study, roughly one quarter of the non-demented patients with PD had psychosis – of whom 52% had major hallucinations or delusions, the rest had ‘minor’ symptoms 2 • The development of psychosis among patients with PD appears to be related to: 1 • Longer disease duration • Greater disease severity • The genetic subtype of PD • Presence of non-motor symptoms including dementia and RBD • Other medical conditions • Medication-related factors Psychosis in PD is more common in advanced disease and is associated with other comorbidities, including cognitive impairment, sleep disorders, and depression 1 RBD=REM (rapid eye movement) sleep behaviour disorder 1. Chang & Fox. Drugs 2016; 76(11): 1093– 1118; 2. Mack et al. Am J Geriatr Psychiatry 2012; 20(2): 123– 132 19
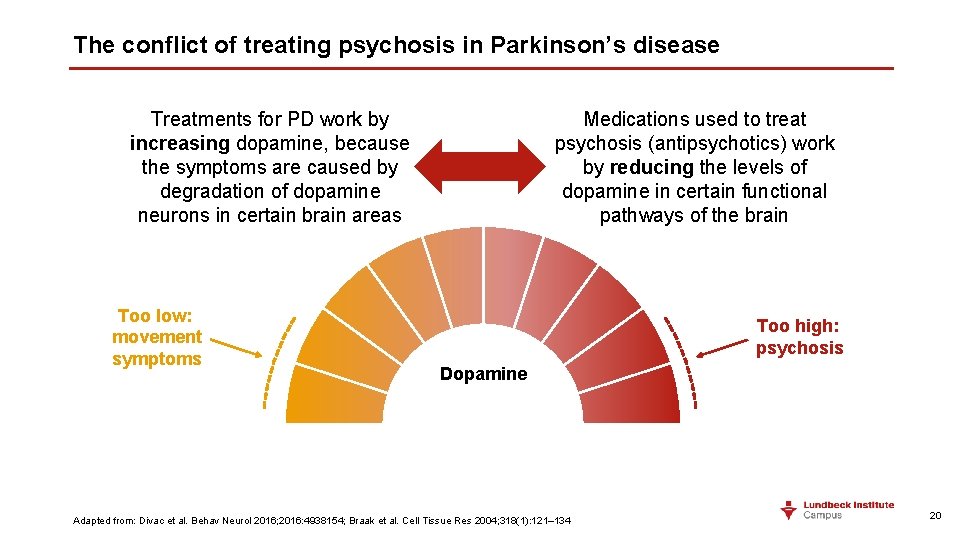
The conflict of treating psychosis in Parkinson’s disease Treatments for PD work by increasing dopamine, because the symptoms are caused by degradation of dopamine neurons in certain brain areas Too low: movement symptoms Medications used to treat psychosis (antipsychotics) work by reducing the levels of dopamine in certain functional pathways of the brain Too high: psychosis Dopamine Adapted from: Divac et al. Behav Neurol 2016; 2016: 4938154; Braak et al. Cell Tissue Res 2004; 318(1): 121– 134 20

Other important non-motor symptoms 21

Gastrointestinal symptoms • The α-synuclein accumulation that is characteristic of PD is not restricted to the brain, and is found in the gastrointestinal tract (and elsewhere) of patients with PD 1 • There is widespread gastrointestinal dysfunction seen in patients with PD, the severity of which varies between patients 1 -3 • The importance of the gut flora is gaining prominence in PD research 2, 4, 5 Constipation 1 • Constipation is the most common gastrointestinal symptom of PD, and is reported in 80– 90% of cases • Constipation is present during the long prodromal phase of the disease, making it a potential biomarker for PD • The mechanisms underlying constipation are unknown, but it can also be a side effect of some of the drugs used to treat PD • The gastrointestinal dysfunction seen in PD can include: • Constipation in PD can be treated with a variety • Oral problems, including swallowing, drooling, and of laxatives, a high-fibre diet, and possibly dental problems 1 -3 7, 8 probiotics 1 -3 • Constipation • Malnutrition 1 -3 • Gastroparesis 6 1. Fasano et al. Lancet Neurol 2015; 14(6): 625– 639; 2. Pfeiffer. Curr Treat Options Neurol 2018; 20(12): 54; 3. Todorova et al. Pract Neurol 2014; 14(5): 310– 322; 4. Lubomski et al. J Neurol 2019 [Epub]; 5. Scheperjans. Curr Opin Neurol 2016; 29(6): 773– 780; 6. Barboza et al. Expert Opin Pharmacother 2015; 16(16): 2449– 2464; 7. Palma & Kaufmann. Mov Disord 2018; 33(3): 372– 390; 8. Barichella et al. Neurology 2016; 87(12): 1274– 1280 22

Speech problems • Hypophonia – soft and weak voice due to lack of coordination of the vocal muscles 1 Treatments for speech problems in PD • There are many therapies that have been • Dysarthria – slurred or slow speech that can studied to try to help patients with PD be difficult to understand, caused by communicate more effectively: disturbances of muscular control because of • One study of singing-based therapy in damage to the central or peripheral nervous patients with PD demonstrated significant 1, 2 system improvements in vocal intensity 4 • The Lee Silverman Voice Treatment, a set of daily exercises, has been shown to • Speech problems are relatively common in improve speech in patients with PD 5 patients with PD, with estimates as high as • Some approaches focus on communication, 90% of patients developing dysarthria or rather than speech, e. g. , identification of the speech problems at some point in the course of topic of conversation, and the use of the disease 2, 3 gestures 2 RCT=randomised controlled trial 1. Dorland’s Illustrated Medical Dictionary. 2009; 2. Tjaden. Top Geriatr Rehabil 2008; 24(2): 115– 126; 3. Schalling et al. Folia Phoniatr Logop 2017; 69(3): 131– 141; 4. Tamplin et al. Neurorehabil Neural Repair 2019; 33(6): 453– 463; 5. Ramig et al. Mov Disord 2018; 33(11): 1777– 1791 23

Swallowing problems (dysphagia) • Dysphagia describes a situation where swallowing becomes difficult 1 Treatments for dysphagia 6 • It is common in patients with PD, affecting >80% at some time during the disease 2 • Patients with PD and dysphagia should be monitored for malnutrition, dehydration, and pulmonary problems • The swallowing problems in PD are often attributed to bradykinesia and muscular rigidity 3 • Levodopa doses should be timed so that meals can be taken during ON time • Although dysphagia typically emerges in the later stages of PD, it can be an early symptom 3 • Active range of motion exercises can be used to strengthen the muscles involved • Dysphagia is an often-overlooked condition, with an impact on the physical, psychological, social, and financial wellbeing of patients with PD and their carers 4 • Other interventions include sipping water, sucking throat lozenges, and synthetic saliva • Tests such as VFSS or FEES can be used to analyse how well a patient is swallowing 5 FEES=fiberoptic endoscopic evaluation of swallowing; VFSS=videofluoroscopic swallowing study Swallowing problems reduce quality of life, complicate medication intake, and can lead to malnutrition and aspiration pneumonia, which is a major cause of death in PD 2 1. Martin (ed). Oxford. Concise Colour Medical Dictionary. 2007; 2. Suttrup & Warnecke. Dysphagia 2016; 31(1): 24– 32; 3. Fasano et al. Lancet Neurol 2015; 14(6): 625– 639; 4. Dziewas et al. Dysphagia 2017; 32(1): 78– 82; 5. Kwon et al. J Mov Disord 2019; 12(3): 1522– 160; 6. Tjaden. Top Geriatr Rehabil 2008; 24(2): 115– 126 24

Sleep disturbances • Many patients with PD (60– 90%) suffer from sleep disturbances, usually emerging early in the course of the disease 1, 2 • ‘Sleep disturbances’ is an umbrella term that includes: 1, 2 • Insomnia – affects ~80% of patients with PD • Excessive daytime sleepiness – affects up to 50% of patients with PD • Periodic limb movements, and restless legs syndrome – are slightly more common among patients with PD than the general population • RBD – present in 25– 50% of patients with PD • Sleep disturbances in PD are thought to be linked to the dopamine system, particularly involving the substantia nigra pars compacta and the ventral tegmental area 1 • Treatments include 3 • Sleep hygiene (e. g. , regular daytime exercise, avoiding stimulants at bedtime, regular hours of sleep at night) • Various pharmacological therapies, including wakefulness-promoting agents, anxiolytics, hypnotics, TCAs, and melatonin Sleep disturbances in PD negatively impact patients’ quality of life, and they are contributing factors for earlier institutionalisation and higher caregiver burden 1 RBD=REM (rapid eye movement) sleep behaviour disorder; TCA=tricyclic antidepressant 1. Lima. Sleep Med Rev 2013; 17(5): 367– 375; 2. Maass & Reichmann. J Neural Transm (Vienna) 2013; 120(4): 565– 569; 3. Todorova et al. Pract Neurol 2014; 14(5): 310– 322 25

Pain • Pain is a symptom of PD in ~60% of cases, 2– 3 times more common than in a non-PD population of the same age 1 • The location of the pain is variable, but is often in the lower back, the legs or the shoulders 1 Classifications of pain in PD Dystonic – associated with abnormal postures and can respond to levodopa therapy 1 Musculoskeletal – muscle/joint pain, inflammation, bone deformity, reduced joint mobility, and abnormal posture. 1 Associated with muscle rigidity and can respond to levodopa 1 • The parts of the brain damaged by PD are involved in pain processing, which has led to the suggestion that this hypofunction could Neuropathic/radicular – peripheral neuropathic result in amplification of sensory stimuli leading pain: restricted to the affected nerve or nerve to spontaneous pain sensation when dopamine root 1 1 levels are low Central or primary – neuropathic pain that is not • In a study of 1, 957 patients with early/moderate restricted to the affected nerve or nerve root. 1 PD, 42% of patients reported moderate to May vary with the medication cycle as a nonsevere pain on the SFMPQ 2 motor fluctuation 1 Pain in patients with PD is common and often disabling, but not well understood 1 SFMPQ=Short Form Mc. Gill Pain Questionnaire 1. Young Blood et al. Parkinsons Dis 2016; 2016: 6067132; 2. Silverdale et al. Parkinsonism Relat Disord 2018; 56: 27– 32 26

Neurogenic orthostatic hypotension 27

Postural/orthostatic hypotension • Orthostatic hypotension (OH) is a condition in Neurogenic orthostatic hypotension which the blood pressure is abnormally low • The autonomic nervous system is responsible upon rising from a sitting or lying position; it can for regulating blood pressure, including lead to ‘syncope’ (fainting) and falls 1, 2 correcting for changes that occur when moving • It is a common symptom, thought to affect from sitting to a standing posture 5, 6 30– 47% of patients with PD 3 • However, the neurodegenerative process • The symptoms are lightheadedness or underlying PD is thought to lead to damage to 3 dizziness on standing, and fatigue the autonomic nervous system 3, 6 • Treatment options for OH include increasing salt and water intake, avoiding volumedepleting drugs, some physical measures, and certain pharmacological therapies 2, 4 OH is a common and challenging symptom affecting PD patients 3 • This can lead to orthostatic hypotension of neurogenic cause (n. OH)3, 6 • Unfortunately, some anti-parkinsonian treatments can worsen the problems of postural hypotension for patients with PD, making treatment decisions complicated 3, 7 n. OH=neurogeneic orthostatic hypotension; OH=orthostatic hypotension 1. Martin (ed). Oxford. Concise Colour Medical Dictionary. 2007; 2. Gibbons et al. J Neurol 2017; 264(8): 1567– 1582; 3. Sánchez-Ferro et al. Front Neurol 2013; 4: 64; 4. Todorova et al. Pract Neurol 2014; 14(5): 310– 322; 5. Freeman. N Engl J Med 2008; 358(6): 615– 624; 6. Kaufmann et al. Expert Rev Cardiovasc Ther 2015; 13(8): 875– 891; 7. Espay et al. Lancet Neurol 2016; 15(9): 954– 966 28
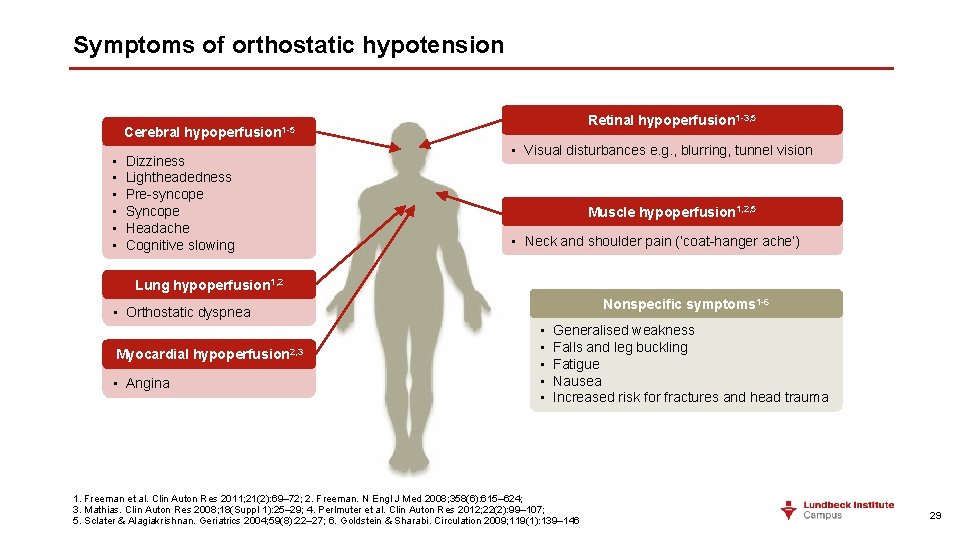
Symptoms of orthostatic hypotension Cerebral • • • Retinal hypoperfusion 1 -3, 5 hypoperfusion 1 -5 Dizziness Lightheadedness Pre-syncope Syncope Headache Cognitive slowing • Visual disturbances e. g. , blurring, tunnel vision Muscle hypoperfusion 1, 2, 5 • Neck and shoulder pain (‘coat-hanger ache’) Lung hypoperfusion 1, 2 Nonspecific symptoms 1 -6 • Orthostatic dyspnea Myocardial hypoperfusion 2, 3 • Angina • • • Generalised weakness Falls and leg buckling Fatigue Nausea Increased risk for fractures and head trauma 1. Freeman et al. Clin Auton Res 2011; 21(2): 69– 72; 2. Freeman. N Engl J Med 2008; 358(6): 615– 624; 3. Mathias. Clin Auton Res 2008; 18(Suppl 1): 25– 29; 4. Perlmuter et al. Clin Auton Res 2012; 22(2): 99– 107; 5. Sclater & Alagiakrishnan. Geriatrics 2004; 59(8): 22– 27; 6. Goldstein & Sharabi. Circulation 2009; 119(1): 139– 146 29

Patient impact of neurogenic orthostatic hypotension • OH has an impact on patients’ lives: • Falls and fear of falling 1, 2 • Depression 3 • Impaired cognitive functioning 3 -5 • Reduced physical fitness 6 • Decreased functioning and Qo. L 2, 3 • Social isolation 6 • Loss of independence 2 • OH has been linked to an increased risk of all-cause mortality 7 OH has considerable impact on the lives of patients with PD 1 -6 • n. OH can be an overlooked problem: • In one survey, 26% of patients, and 33% of caregivers said that they had to mention n. OH symptoms repeatedly to healthcare providers 8 • Patients themselves can be unaware of OH 9, 10 • The following percentage of patients with PD reported the following limitations due to OH: 8 Physical activity/exercise – 53% Housework – 47% Travelling – 47% Running errands – 46% Socialising – 45% n. OH=neurogenic orthostatic hypotension; OH=orthostatic hypotension; Qo. L=quality of life 1. Goldstein & Sharabi. Circulation 2009; 119(1): 139– 146; 2. Sclater & Alagiakrishnan. Geriatrics 2004; 59(8): 22– 27; 3. Perlmuter et al. Clin Auton Res 2012; 22(2): 99– 107; 4. Centi et al. Neurology 2017; 88(1): 17– 24; 5. Boylan & Messinis. Neurology 2017; 88(1): 11– 12; 6. Mak & Pang. J Neurol 2009; 256(10): 1689– 1695; 7. Maule et al. Funct Neurol 2012; 27(2): 101– 106; 8. Claassen et al. BMC Neurol 2018; 18(1): 125; 9. Merola et al. Parkinsonism Relat Disord 2016; 33: 65– 71; 10. Palma & Kaufmann. Mov Disord 2018; 33(3): 372– 390 30

Falls and fear of falling in patients with Parkinson’s disease • In a study of 844 nursing-home residents, OH was an • One study found that falls were the most common reason for hospitalisation in patients with PD 4 independent risk factor falls 1 • Another study found that patients with severe OH had • Falls put patients at risk for fractures, and head trauma 5, 6 a higher prevalence of falls than moderate OH (57. 9% versus 38. 9%, p<0. 001)2 • After experiencing a fall, approximately a third of patients develop a fear of falling, some of whom • A systematic review of falls in 22 studies of patients 3 experience a self-imposed reduction in activity 7 with PD found: Patients (%, range) 100% 80% 61% Range: 35– 90% 39% 40% Range: 18– 65% 20% 0% Experienced at least 1 fall Recurrent falls • Interviews of patients with PD with a fear of falling revealed several themes: 8 • Fear of falling varies from patient to patient • Fear of falling creates disturbances in everyday life, including making daily activities hazardous, and inducing a feeling of vulnerability • Patients handle a fear of falling by adopting a number of different strategies, including cautiousness, and avoiding certain activities OH=orthostatic hypotension 1. Ooi et al. Am J Med 2000; 108(2): 106– 111; 2. Merola et al. Parkinsonism Relat Disord 2016; 33: 65– 71; 3. Allen et al. Parkinsons Dis 2013; 2013: 906274; 4. Lubomski et al. J Neurol Neurosurg Psychiatry 2015; 86(3): 324– 330; 5. Goldstein & Sharabi. Circulation 2009; 119(1): 139– 146; 6. Pathak et al. Mov Disord 2005; 20(9): 1213– 1219; 7. Vellas et al. Ageing 1997; 26(3): 189– 193; 8. Jonasson et al. BMC Geriatr 2018; 18(1): 44 31

Diagnosis of neurogenic orthostatic hypotension Diagnosis and evaluation of n. OH 1 -3 Confirmed, persistent OH Yes Discontinue, or change dose Yes Treat underlying cause of OH No Investigate other causes Hypotensive drugs? e. g. , vasodilators No Identifiable cause? e. g. , hypovolaemia, dehydration, adrenal insufficiency No Neurogenic? e. g. , presence of noradrenergic denervationa Yes Management of n. OH • Differential diagnosis is vital when identifying n. OH in patients – there are many disorders of the ANS that are associated with orthostatic intolerance: 3 • Reflex syncope, including neurocardiogenic syncope, and carotid sinus hypersensitivity • Postural orthostatic tachycardia syncope, primary and secondary • Autonomic failure, both acute and chronic, including multiple system atrophy, pure autonomic failure, and PD • Non-neurogenic causes of OH include volume depletion, polypharmacy, anemia, and infection 4, 5 ae. g. , indicated by an inadequate/blunted increase in heart rate (<15 bpm, or ratio of <0. 5 in change in heart rate over change in systolic blood pressure) in an individual developing hypotension upon standing; 4, 5 ANS=autonomic nervous system; n. OH=neurogenic orthostatic hypotension; OH=orthostatic hypotension 1. Goldstein & Sharabi. Circulation 2009; 119(1): 139– 146; 2. Maule et al. Cardiovasc Hematol Disord Drug Targets 2007; 7(1): 63– 70; 3. Grubb. Circulation 2005; 111(22): 2997– 3006; 4. Gibbons et al. J Neurol 2017; 264(8): 1567– 1582; 5. Norcliffe-Kaufmann et al. Ann Neurol 2018; 83(3): 522– 531 32

Management of neurogenic orthostatic hypotension • The goal of therapy in patients with OH is to reduce postural symptoms, rather than achieve normotension 1 • Non-pharmacological interventions should always be the first-line response to OH and n. OH; such interventions include: 1 -4 • • Avoiding quick positional changes Increasing fluid intake Adjusting PD medication Increasing salt intake Wearing compression stockings and/or abdominal binders Elevation of the head of the bed Avoiding over-heating Physical measures, including standing up and crossing legs, standing up and squeezing hands tightly • Pharmacological interventions include a variety of potential therapies that act on blood vessels, blood volume, or pressor mechanisms: 1, 4 • • Mineralocorticoids α 1 -adrenergic agonists Replenishing plasma noradrenaline levels/ noradrenaline precursor 5 Vasopressin receptor agonists Venous α-adrenergic agonists Erythropoiesis stimulating agents Prostaglandin synthetase inhibitors Acetylcholinesterase inhibitors Healthcare providers must often make use of non-pharmacological treatments and pharmacotherapy to improve the functioning and Qo. L of patients with n. OH 3 n. OH=neurogenic orthostatic hypotension; OH=orthostatic hypotension; Qo. L=quality of life 1. Maule et al. Cardiovasc Hematol Disord Drug Targets 2007; 7(1): 63– 70; 2. Claassen et al. BMC Neurol 2018; 18(1): 125; 3. Isaacson. Am J Manag Care 2015; 21(13 Suppl): S 258–S 268; 4. Freeman. N Engl J Med. 2008; 358(6): 615– 624; 5. Kaufmann et al. Expert Rev Cardiovasc Ther 2015; 13(8): 875– 891 33

Comorbidities of PD 34
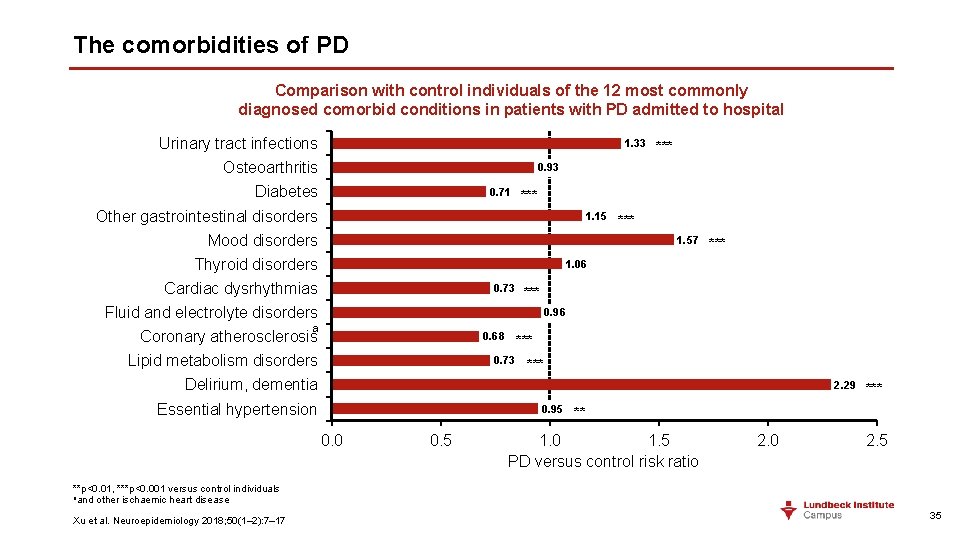
The comorbidities of PD Comparison with control individuals of the 12 most commonly diagnosed comorbid conditions in patients with PD admitted to hospital Urinary tract infections 1. 33 Osteoarthritis *** 0. 93 Diabetes 0. 71 *** Other gastrointestinal disorders 1. 15 Mood disorders *** 1. 57 Thyroid disorders *** 1. 06 Cardiac dysrhythmias 0. 73 *** Fluid and electrolyte disorders 0. 96 a Coronary atherosclerosis 0. 68 Lipid metabolism disorders *** 0. 73 *** Delirium, dementia 2. 29 Essential hypertension 0. 95 0. 0 0. 5 *** ** 1. 0 1. 5 PD versus control risk ratio 2. 0 2. 5 **p<0. 01, ***p<0. 001 versus control individuals aand other ischaemic heart disease Xu et al. Neuroepidemiology 2018; 50(1– 2): 7– 17 35

“ Improved awareness and understanding of the relationship between non-motor symptoms, disability, and quality of life will encourage a multidisciplinary approach to patient care Duncan et al. Mov Disord 2014; 29(2): 195– 202 36
- Slides: 36