Nonmetals Metals Describe Metals Properties shiny ductile malleable
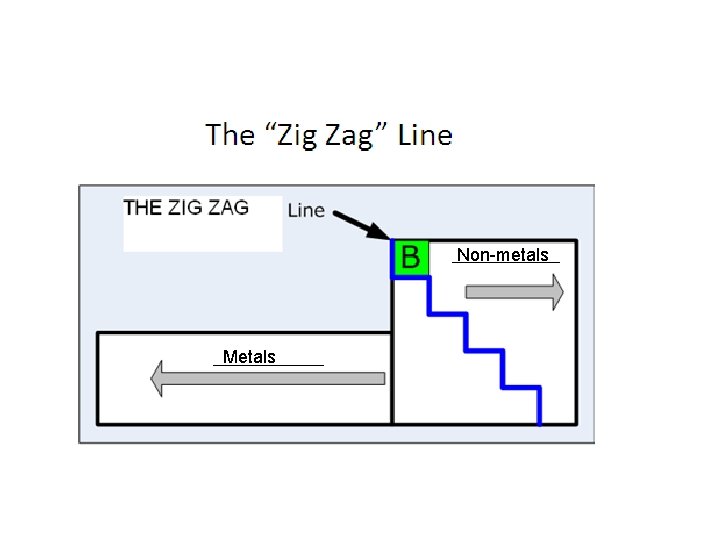
Non-metals Metals

Describe Metals: Properties: shiny, ductile, malleable, conduct thermal and electrical energy Where are they located on the periodic table? To the left of the “zig zag” line

Describe Non-Metals Properties: dull, brittle, non-conductive Where are they located on the periodic table? To the right of the “zig zag” line Describe Metalloids Properties: have some properties of both metals & non-metals, semi conductors Where are they located on the periodic table? – on the “zig zag” line
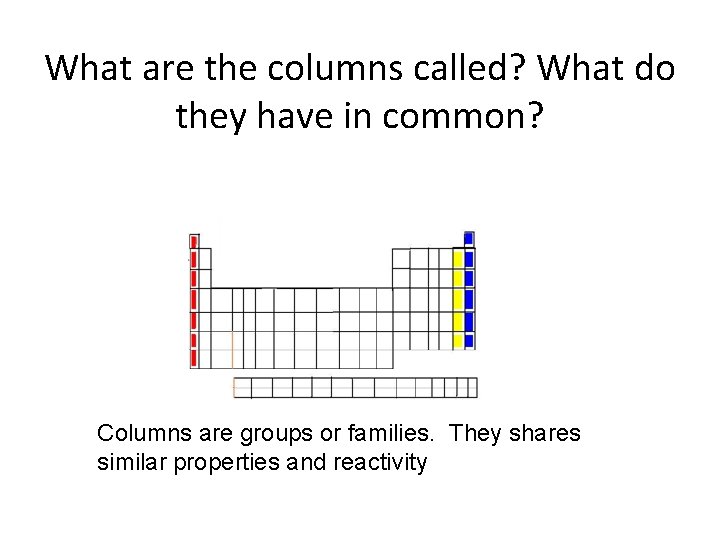
What are the columns called? What do they have in common? Columns are groups or families. They shares similar properties and reactivity
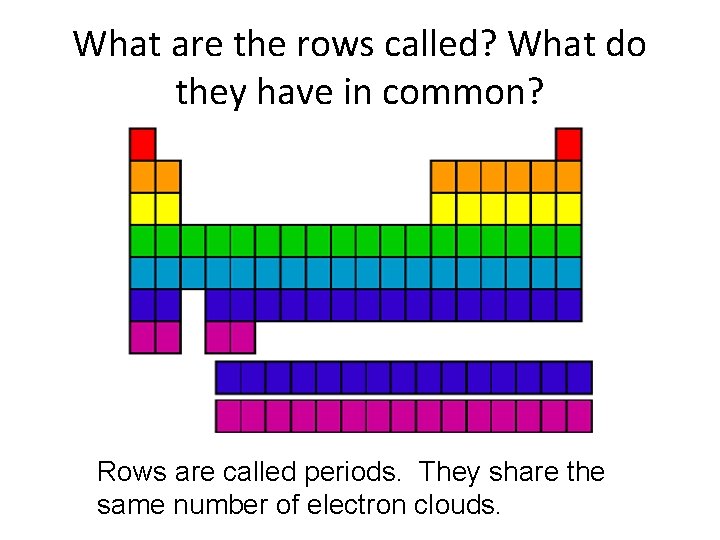
What are the rows called? What do they have in common? Rows are called periods. They share the same number of electron clouds.

Questions 1. Most of the elements are what state of matter at room temperature? solid 2. How many elements are liquids at room temperature? 2 3. Which elements all tend to be solids at room temperature? metals 4. Which elements all tend to be gasses at room temperature. Non-metals

Atoms of an Element 1. How many protons are in this element? 9 2. How many neutrons are in this element? 10 3. What is the atomic number? 9 4. What is the atomic mass? 19 5. How many electrons are in this element? 9

Elements, Compounds & Mixtures 1. If it is on the periodic table, it is an element. 2. If it is not on the periodic table, it is a compound or a mixture. 3. If it is two or more substances, chemically combined then it is a compound. 4. If it is not a pure substance then it is a mixture. 5. The smallest part that in which an element can be divided and still have the properties of that element is an atom.
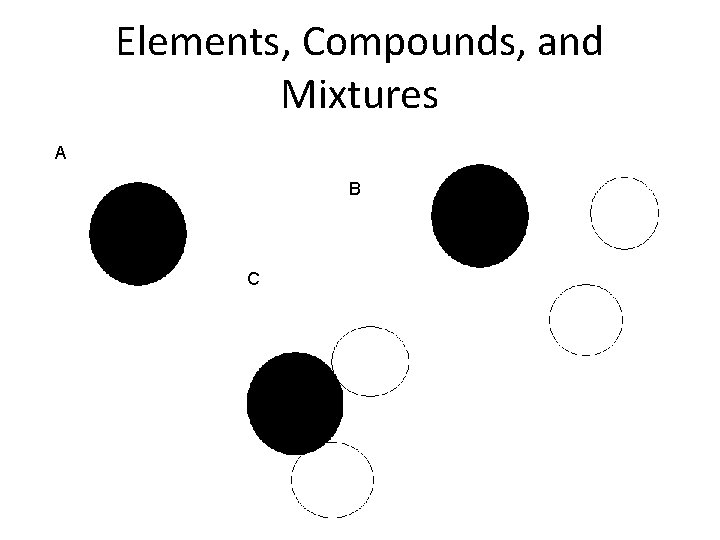
Elements, Compounds, and Mixtures A B C
- Slides: 9