NONMETALLIC STRUCTURAL MATERIALS Polymers plastics Ceramics Glass amorphous













- Slides: 18

NONMETALLIC STRUCTURAL MATERIALS • Polymers (plastics) • Ceramics • Glass (amorphous materials)

Composition of Ceramics • Crystal phase – basic part of ceramics structure • Amorphous (glassy) phase – binder • Gas phase in ceramics pores CERAMICS Oxide ceramics based on Al 2 O 3 Zr. O 2 and other oxides Oxigen-free ceramics based on Si. C Si 3 N 4 Metal ceramics, or cermets

Advantages of Ceramics • • • High hardness and wear resistance High service temperature ( up to 3500 °С) Corrosion resistance in various environments Low electric conductivity (dielectrics) Heat insulators Low labour input of processing (both product and material are created simultaneously; strengthening and finishing operations are not required) • Low density

Basic Lacks of Ceramics • Britlness (impact strength of ceramics is 40 times less than metals have) • Oxide ceramics does not withstand the sharp changes of temperature • Oxigen-free ceramics can be obtained by hot pressing only (technological problems)

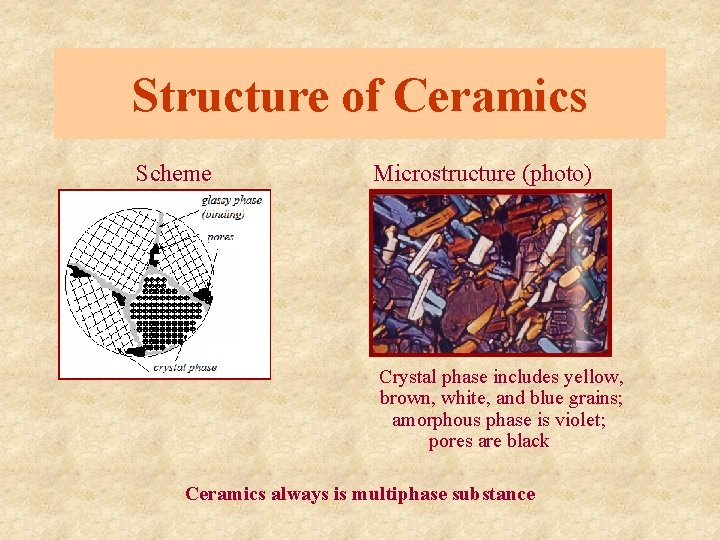
Structure of Ceramics Scheme Microstructure (photo) Crystal phase includes yellow, brown, white, and blue grains; amorphous phase is violet; pores are black Ceramics always is multiphase substance

Ceramic Shrinkage in Sintering A ready porcelain figure and a cup have the sizes approximately 13 % less than green (unburnt) work-peaces

Application of Ceramics Building ceramics: bricks and tile These buildings in Amsterdam are more than 400 years old

Application of Ceramics Pyramids are the most ancient building structures Khafr’s Pyramid and Great Sphinx Great pyramids in Giza: XXVI century B. C.

Application of Ceramics: technical alumina ceramics Thread feeders, spools (parts for weaving industry) Nozzles of torches Dies for wire drawing Parts of machines Sanitaryware items

“Ductile” ceramics based on Zr. O 2: the stress near the growing crack tip causes a phase transformation; a new phase with bigger specific volume hinders crack growth

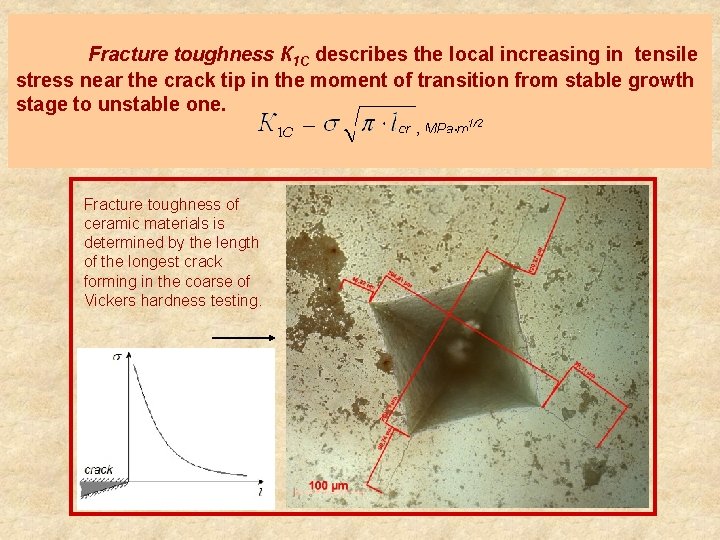
Fracture toughness К 1 С describes the local increasing in tensile stress near the crack tip in the moment of transition from stable growth stage to unstable one. Fracture toughness of ceramic materials is determined by the length of the longest crack forming in the coarse of Vickers hardness testing.

Building Ceramics • Bricks and ceramic tiles are formed and baked clay; their composition: hydrated aluminosilicates Al 2(Si 2 O 5)(OH)4 and impurities. • Cement is a mix of limestone Ca. O, silicon dioxide Si. O 2 and aluminium oxide Al 2 O 3. It hardens being mixed with water. • Concret is a composite material including sand stones, binded together with hardened cement. Ratio sand to gravel is 60: 40.

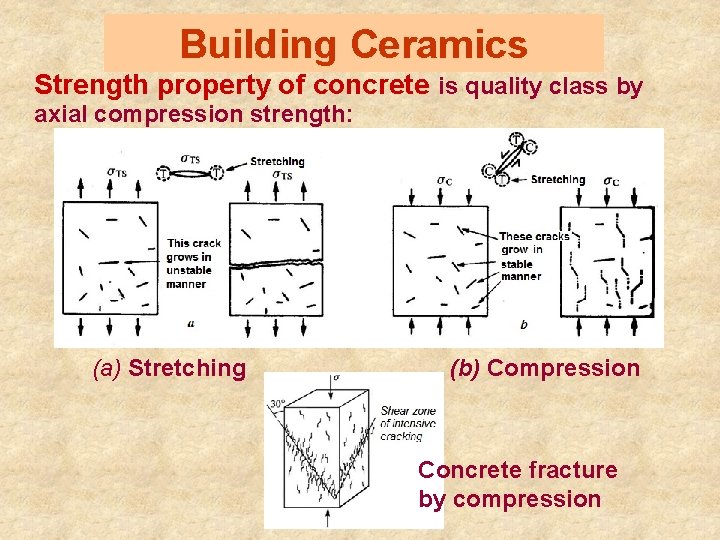
Building Ceramics Strength property of concrete is quality class by axial compression strength: (a) Stretching (b) Compression Concrete fracture by compression

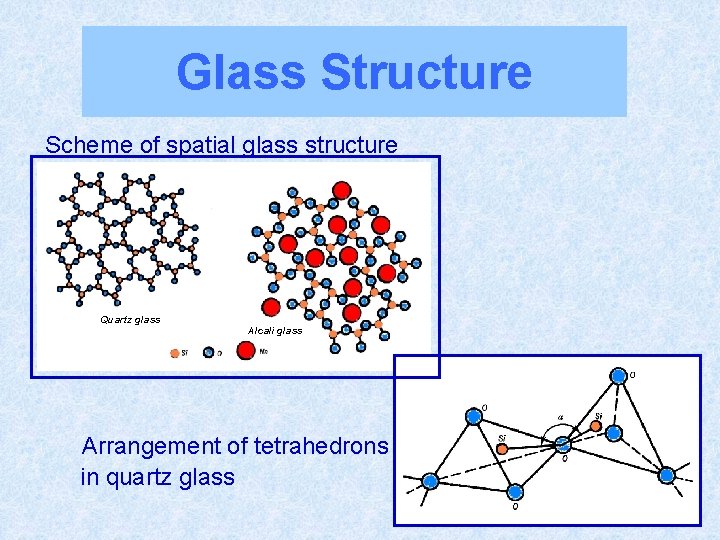
Glass Structure Scheme of spatial glass structure Quartz glass Alcali glass Arrangement of tetrahedrons in quartz glass

Application of Glass Constructional glass: window glass pane Art glass works

Application of Glass Technical glass: insulators of power line

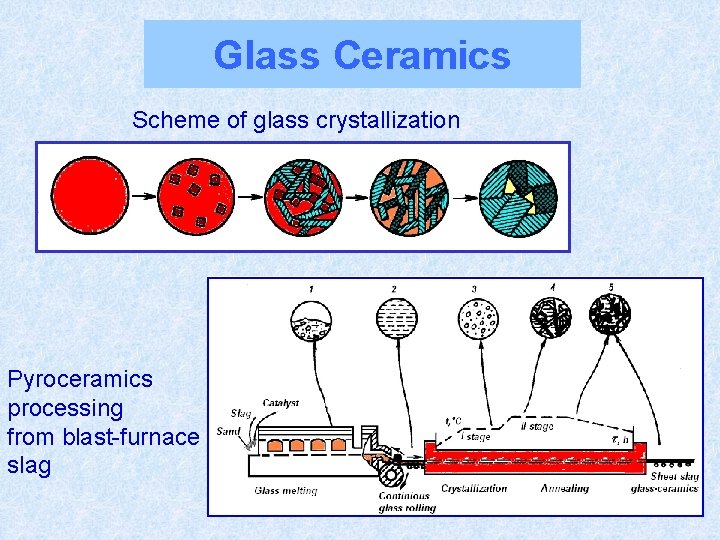
Glass Ceramics Scheme of glass crystallization Pyroceramics processing from blast-furnace slag

Glass Ceramics a b Process of glass crystallization × 7500 (grain size 1 -2 μm) c d