NonIsothermal Reactor Design Problems Problems The following reaction

Non-Isothermal Reactor Design Problems

Problems The following reaction CH 3 C 0 CH 3 CH 2 CO + CH 4 is I order w. r. t. acetone and the specific reaction rate can be expressed by: ln k = 34. 34 – 34222/T, where k is in sec-1 and T is in K. It is desired to feed 8000 kg of acetone/hr to an adiabatic PFR. If the feed is pure acetone, the inlet temperature is 1035 K and the pressue 162 k. Pa, calculate the volume of PFR required to obtain 20% conversion of acetone. DATA: Species Hf 0 at 298 K Acetone -216. 67 26. 63 0. 183 -45. 86 x 10 -6 Ketene -61. 09 20. 04 0. 0945 -30. 95 x 10 -6 Methane -74. 81 13. 39 0. 077 -18. 71 x 10 -6 (k. J/mol) CP = + T 2, where CP is J/mol K and T is in K

Solution: HR 0(TR) = -61. 09 -74. 81 + 216. 67 = 80. 77 k. J/kmol
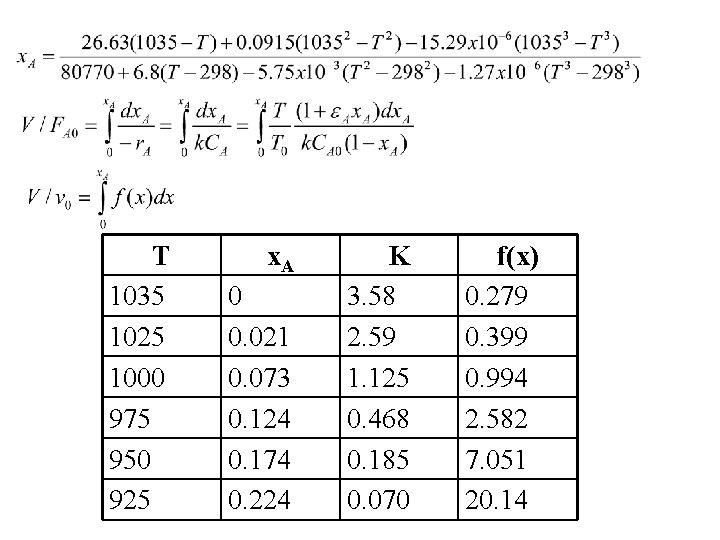
T 1035 1025 1000 975 950 925 x. A 0 0. 021 0. 073 0. 124 0. 174 0. 224 K 3. 58 2. 59 1. 125 0. 468 0. 185 0. 070 f(x) 0. 279 0. 399 0. 994 2. 582 7. 051 20. 14
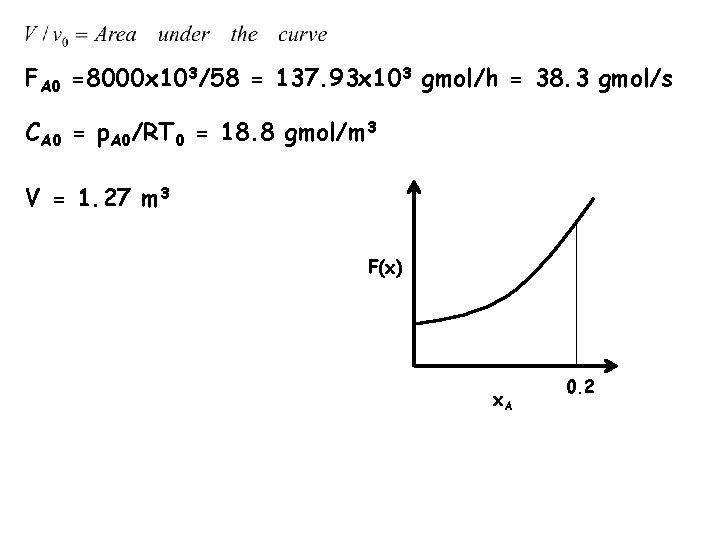
FA 0 =8000 x 103/58 = 137. 93 x 103 gmol/h = 38. 3 gmol/s CA 0 = p. A 0/RT 0 = 18. 8 gmol/m 3 V = 1. 27 m 3 F(x) x. A 0. 2

Problem: The gas phase reaction 2 A R is carried out in an adiabatic PFR. The feed consisting of pure A enters the reactor at 800 K, 2 atm and at a flowrate of 75 kmol/hr. Find the volume of the reactor necessary to obtain 40% conversion of A. DATA: CPA = 12 k. Cal/kmol K; CPB = 9 k. Cal / kmol K k. P = 0. 01 T – 7, where k. P is in kmol/(m 3 hr atm 2) and T is in K HR 0 (298) = -100 k. Cal/kmol Solution:

Why T/T 0 is not coming into picture ? ? A = -0. 5; p. A 0 = PT 0 = 2 atm; FA 0 = 75 kmol/hr Integrate and find the volume ? ?

Problem: The gas phase reaction A + 2 B 2 R with –r. A = k. P p. A 2 is carried out in an adiabatic PFR. The feed to the reactor consists of equimolar quantities of A & B and enters at 600 K and 2 atm. The feed flowrate is 60 kmol/hr. Find the volume of the reactor necessary to obtain 80% conversion of A. DATA: CPA = 20; CPB = 8; CPR = 18 k. Cal / kmol K k. P = 0. 01 T – 6, where k. P is in kmol/(m 3 hr atm 2) and T is in K HR 0 (298) = 1500 k. Cal/kmol

Problem: For the elementary liquid phase reaction A B, determine the adiabatic equilibrium temperature and adiabatic equilibrium conversion, when pure A is fed at 300 K. DATA: CPA = CPB = 50 Cal / mol K k. P = 0. 01 T – 6, where k. P is in kmol/(m 3 hr atm 2) and T is in K HA, f 0 (298) = -40, 000 Cal/mol; HB 0, f (298) = -60, 000 Cal/mol The equilibrium constant is 100000 at 298 K Solution: At equilibrium: k 1 CAe = k 2 CBe CAe = CBe/Ke CA 0 (1 -x. Ae) = CA 0 x. Ae/Ke x. Ae = Ke/(1+Ke)

dln. K/d. T = HR 0/RT 2 HR 0 is independent of temperature Energy Balance:
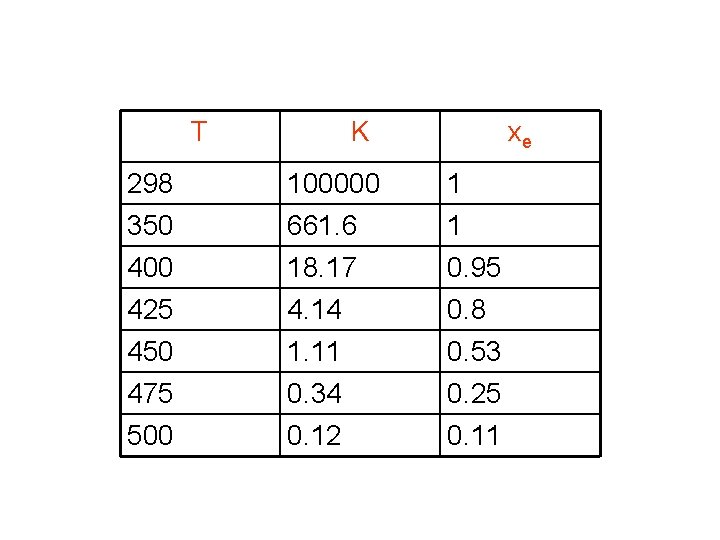
T K xe 298 350 100000 661. 6 1 1 400 18. 17 0. 95 425 450 475 500 4. 14 1. 11 0. 34 0. 12 0. 8 0. 53 0. 25 0. 11
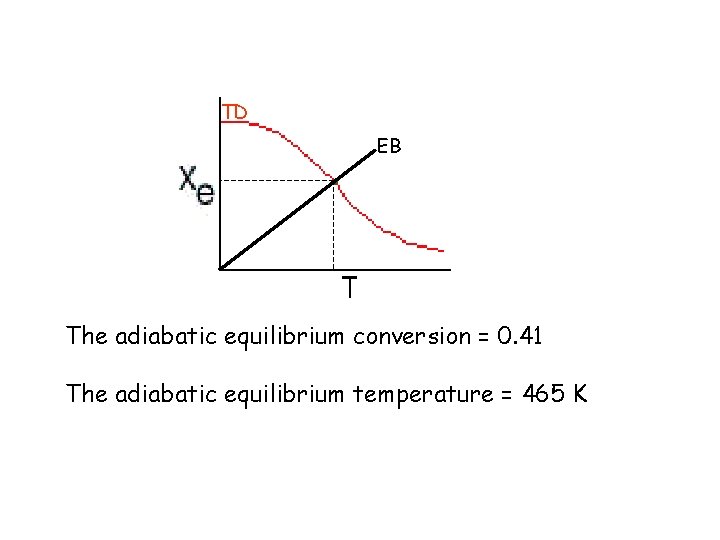
TD TD EB The adiabatic equilibrium conversion = 0. 41 The adiabatic equilibrium temperature = 465 K
- Slides: 12