Nonalcoholic Fatty Liver Disease NAFLD Masi Khaja M
![Nonalcoholic Fatty Liver Disease [NAFLD] Masi Khaja, M. D Nonalcoholic Fatty Liver Disease [NAFLD] Masi Khaja, M. D](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-1.jpg)


![Nonalcoholic Fatty Liver Disease [NAFLD] • Histology based STEATOSIS • No significant alcohol intake Nonalcoholic Fatty Liver Disease [NAFLD] • Histology based STEATOSIS • No significant alcohol intake](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-4.jpg)
![Grading for Steatosis [Adapted from Brunt et al. Am J Gastroenterol 1999] n GRADE Grading for Steatosis [Adapted from Brunt et al. Am J Gastroenterol 1999] n GRADE](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-6.jpg)
![Nonalcoholic Steatohepatitis [NASH] n n n Ludwig et al. [1980 – Mayo Clinic] – Nonalcoholic Steatohepatitis [NASH] n n n Ludwig et al. [1980 – Mayo Clinic] –](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-7.jpg)
![Grading for Steatohepatitis [Adapted from Brunt et al. Am J Gastroenterol 1999] n GRADE Grading for Steatohepatitis [Adapted from Brunt et al. Am J Gastroenterol 1999] n GRADE](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-9.jpg)
![Staging for Fibrosis [Adapted from Brunt et al. Am J Gastroenterol 1999] v STAGE Staging for Fibrosis [Adapted from Brunt et al. Am J Gastroenterol 1999] v STAGE](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-10.jpg)






![Keys to the Diagnosis of NASH [2] Laboratory v v AST & ALT 2 Keys to the Diagnosis of NASH [2] Laboratory v v AST & ALT 2](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-19.jpg)

![NAFLD/NASH Prevalence [%] Population Screening Autopsy Morbid Obesity [BMI >35] NAFLD 15 -29 15 NAFLD/NASH Prevalence [%] Population Screening Autopsy Morbid Obesity [BMI >35] NAFLD 15 -29 15](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-22.jpg)

![Conditions Associated with NAFLD/NASH n Chronic Metabolic Syndrome [“ 10 NASH”] n n n Conditions Associated with NAFLD/NASH n Chronic Metabolic Syndrome [“ 10 NASH”] n n n](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-26.jpg)

![Pathogenesis of NASH [2] Environment Socioeconomics Public Problems Nutritional Practices Obesity/DM Hepatic Steatosis NASH Pathogenesis of NASH [2] Environment Socioeconomics Public Problems Nutritional Practices Obesity/DM Hepatic Steatosis NASH](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-28.jpg)
![Pathogenesis of NASH [3] Genetic Determinants n n n STEATOSIS Obesity/Lipodystrophy Insulin signaling n Pathogenesis of NASH [3] Genetic Determinants n n n STEATOSIS Obesity/Lipodystrophy Insulin signaling n](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-29.jpg)
![Pathogenesis of NASH [4] The “Two Hit” Hypothesis Normal Steatosis VULNERABLE Inflammation & Fibrosis Pathogenesis of NASH [4] The “Two Hit” Hypothesis Normal Steatosis VULNERABLE Inflammation & Fibrosis](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-30.jpg)
![Pathogenesis of NASH [5] Fat Accumulation in the Liver Increased Fatty Acid Influx • Pathogenesis of NASH [5] Fat Accumulation in the Liver Increased Fatty Acid Influx •](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-31.jpg)
![Pathogenesis of NASH [6] Insulin Resistance Obesity Hyperlipidemia Disrupted Insulin Signaling Insulin Resistance Hyperinsulinemia Pathogenesis of NASH [6] Insulin Resistance Obesity Hyperlipidemia Disrupted Insulin Signaling Insulin Resistance Hyperinsulinemia](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-32.jpg)
![Pathogenesis of NASH [7] Molecular Mechanisms for NAFLD-related Insulin Resistance IKK [inhibitor kappa beta Pathogenesis of NASH [7] Molecular Mechanisms for NAFLD-related Insulin Resistance IKK [inhibitor kappa beta](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-33.jpg)


![Treatment [2] Treat Co-morbidities n DM n n No data Lipids n Clofibrate ineffective Treatment [2] Treat Co-morbidities n DM n n No data Lipids n Clofibrate ineffective](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-37.jpg)
![Treatment [3] Wt loss n Diet [4 studies] n n n Histologic exacerbation if Treatment [3] Wt loss n Diet [4 studies] n n n Histologic exacerbation if](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-38.jpg)
![Treatment [4] Wt loss Guidelines n 10% wt loss n n n Gradual [31/2 Treatment [4] Wt loss Guidelines n 10% wt loss n n n Gradual [31/2](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-40.jpg)
![Treatment [5] Wt loss v Bariatric Surgery: v Jejunoileal bypass – causes NASH v Treatment [5] Wt loss v Bariatric Surgery: v Jejunoileal bypass – causes NASH v](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-41.jpg)
![Treatment [6] Potential Pharmacologic Treatment n Insulin-sensitizing agents: n n n Lipid-lowering agents: n Treatment [6] Potential Pharmacologic Treatment n Insulin-sensitizing agents: n n n Lipid-lowering agents: n](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-42.jpg)
![Treatment [7] NASH & Insulin-Sensitizing Agents PI Treatment Caldwell Troglitazone Am J Gastro 2001 Treatment [7] NASH & Insulin-Sensitizing Agents PI Treatment Caldwell Troglitazone Am J Gastro 2001](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-43.jpg)
![Treatment [8] NASH & Vitamin E PI Treatment Lavine Vit E J Pediatr 2000 Treatment [8] NASH & Vitamin E PI Treatment Lavine Vit E J Pediatr 2000](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-44.jpg)
![Treatment [9] Cytoprotective Agents n Ursodeoxycholic acid [UCDA] Ref N Study Type Duration AST/ALT Treatment [9] Cytoprotective Agents n Ursodeoxycholic acid [UCDA] Ref N Study Type Duration AST/ALT](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-47.jpg)
![Treatment [9] NASH & Ursodeoxycholic Acid n Lindor et al Hepatology 2004 n Placebo-controlled Treatment [9] NASH & Ursodeoxycholic Acid n Lindor et al Hepatology 2004 n Placebo-controlled](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-48.jpg)
![Treatment [10] NASH & Ursodeoxycholic Acid n Safe & well-tolerated n 40% steatosis improved Treatment [10] NASH & Ursodeoxycholic Acid n Safe & well-tolerated n 40% steatosis improved](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-49.jpg)
![Treatment [11] Summary n Reduce insulin resistance n n n Fibrates Statins Lipotropic Agents Treatment [11] Summary n Reduce insulin resistance n n n Fibrates Statins Lipotropic Agents](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-50.jpg)


![NAFLD NAFL NASH Cirrhosis Steatosis Fibrosis [macrovesicular] Inflammation Ballooning Steatosis Mallory’s Hyaline Hepatocellular Cancer NAFLD NAFL NASH Cirrhosis Steatosis Fibrosis [macrovesicular] Inflammation Ballooning Steatosis Mallory’s Hyaline Hepatocellular Cancer](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-53.jpg)
![Current Treatment Algorithm Treat Co-morbidity Confirm Histology [Prognosis] No Proven Treatment [Vitamin E ? Current Treatment Algorithm Treat Co-morbidity Confirm Histology [Prognosis] No Proven Treatment [Vitamin E ?](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-54.jpg)



- Slides: 58
![Nonalcoholic Fatty Liver Disease NAFLD Masi Khaja M D Nonalcoholic Fatty Liver Disease [NAFLD] Masi Khaja, M. D](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-1.jpg)
Nonalcoholic Fatty Liver Disease [NAFLD] Masi Khaja, M. D

Objectives Epidemiology n Natural History n Clinical Presentation n Pathogenesis n Approach to Diagnosis n Treatment n

What is NAFLD?
![Nonalcoholic Fatty Liver Disease NAFLD Histology based STEATOSIS No significant alcohol intake Nonalcoholic Fatty Liver Disease [NAFLD] • Histology based STEATOSIS • No significant alcohol intake](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-4.jpg)
Nonalcoholic Fatty Liver Disease [NAFLD] • Histology based STEATOSIS • No significant alcohol intake [<20 g/d male; <10 g/d female] • Excluding 2 nd causes STEATOHEPATITIS BRIDGING FIBROSIS CIRRHOSIS • Histology based – steatosis with necrosis, inflammation & perisinusoidal fibrosis [zone 3 around central vein] [ETOH & HCV = fibrosis initially portal distribution]

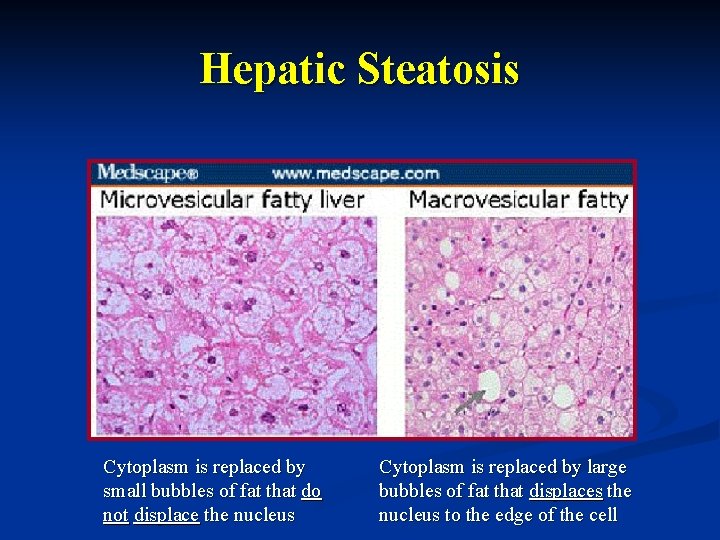
Hepatic Steatosis Cytoplasm is replaced by small bubbles of fat that do not displace the nucleus Cytoplasm is replaced by large bubbles of fat that displaces the nucleus to the edge of the cell
![Grading for Steatosis Adapted from Brunt et al Am J Gastroenterol 1999 n GRADE Grading for Steatosis [Adapted from Brunt et al. Am J Gastroenterol 1999] n GRADE](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-6.jpg)
Grading for Steatosis [Adapted from Brunt et al. Am J Gastroenterol 1999] n GRADE 1: <33% hepatocytes affected n GRADE 2: 33% to 66% of hepatocytes affected n GRADE 3: > 66% of hepatocytes are affected
![Nonalcoholic Steatohepatitis NASH n n n Ludwig et al 1980 Mayo Clinic Nonalcoholic Steatohepatitis [NASH] n n n Ludwig et al. [1980 – Mayo Clinic] –](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-7.jpg)
Nonalcoholic Steatohepatitis [NASH] n n n Ludwig et al. [1980 – Mayo Clinic] – coined the term NASH 20 patients evaluated over 10 -year period Histologic evidence of alcoholic hepatitis but no history of alcohol abuse or other causes of liver disease: n n n n Hallmark: moderate to severe macrovesicular steatosis with lobular inflammation 70% Mallory bodies 70% perisinusoidal, centrilobular, and/or septal fibrosis 15% cirrhosis 60% F [54 yrs]; 90% obese [>110% of ideal body weight]; 25% dyslipidemia +/- DM Asymptomatic 90% abnormal LFT; 75% hepatomegaly; 25% splenomegaly; 5% ascites

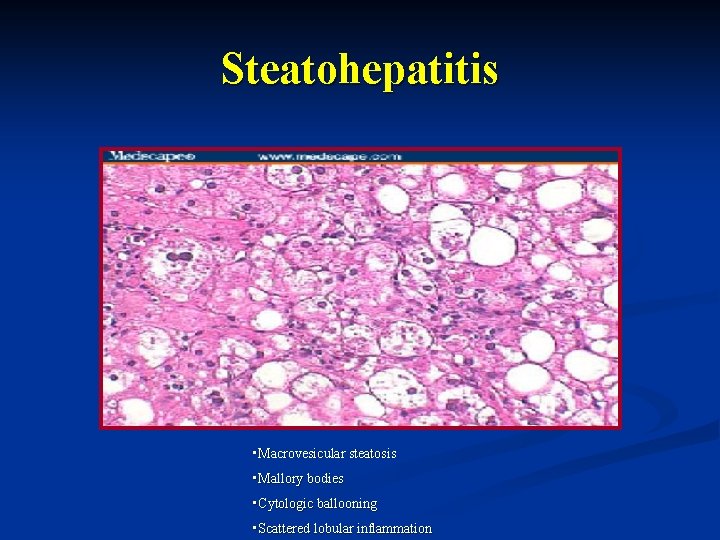
Steatohepatitis • Macrovesicular steatosis • Mallory bodies • Cytologic ballooning • Scattered lobular inflammation
![Grading for Steatohepatitis Adapted from Brunt et al Am J Gastroenterol 1999 n GRADE Grading for Steatohepatitis [Adapted from Brunt et al. Am J Gastroenterol 1999] n GRADE](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-9.jpg)
Grading for Steatohepatitis [Adapted from Brunt et al. Am J Gastroenterol 1999] n GRADE 1 [mild]: n n GRADE 2 [moderate]: n n n Steatosis – predominantly macrovesicular, involves up to 66% of lobules Ballooning – occasionally observed; zone 3 hepatocytes Lobular inflammation – scattered & mild acute [polymorphs] & chronic inflammation [mononuclear cells] Steatosis – any degree; usually mixed macro & microvesicular Ballooning – obvious & present in zone 3 Lobular inflammation – polymorphs may be noted associated with ballooned hepatocytes, +/- pericellular fibrosis mild chronic inflammation Portal inflammation – none, mild to moderate GRADE 3 [severe]: n n Steatosis – usually >66% [zone 3 or panacinar]; commonly mixed steatosis Ballooning – predominantly zone 3; marked Lobular inflammation – scattered acute & chronic inflammation] Portal inflammation – mild to moderate
![Staging for Fibrosis Adapted from Brunt et al Am J Gastroenterol 1999 v STAGE Staging for Fibrosis [Adapted from Brunt et al. Am J Gastroenterol 1999] v STAGE](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-10.jpg)
Staging for Fibrosis [Adapted from Brunt et al. Am J Gastroenterol 1999] v STAGE 1: zone 3 perivenular, perisinusoidal, or pericellular fibrosis; focal or extensive v STAGE 2: as above, with focal or extensive periportal fibrosis v STAGE 3: bridging fibrosis, focal or extensive v STAGE 4: cirrhosis

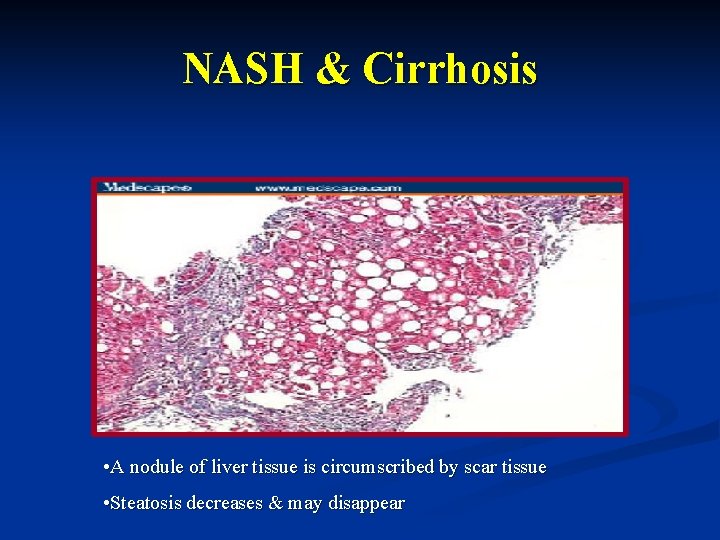
NASH & Cirrhosis • A nodule of liver tissue is circumscribed by scar tissue • Steatosis decreases & may disappear

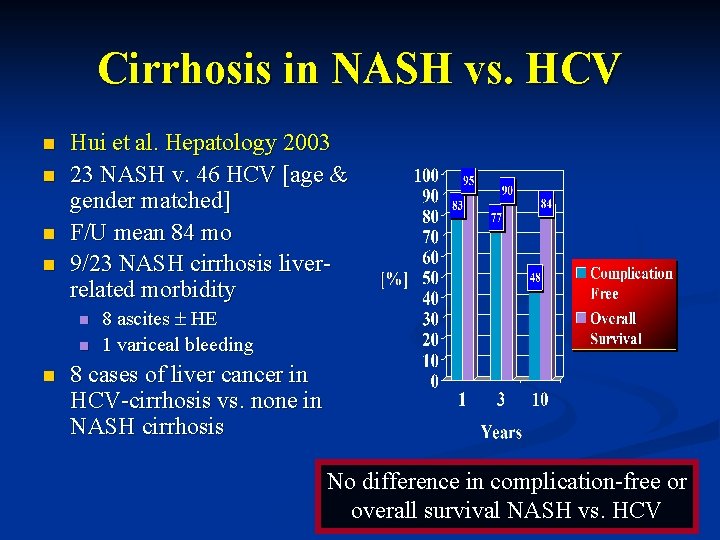
Cirrhosis in NASH vs. HCV n n Hui et al. Hepatology 2003 23 NASH v. 46 HCV [age & gender matched] F/U mean 84 mo 9/23 NASH cirrhosis liverrelated morbidity n n n 8 ascites HE 1 variceal bleeding 8 cases of liver cancer in HCV-cirrhosis vs. none in NASH cirrhosis No difference in complication-free or overall survival NASH vs. HCV

Risk Factors for Fibrosis AST/ALT <1 DM Obesity + - >1 <1 >1 4 0 47 66 0 0 12 13 <45 Age [years] • Mayo Clinic • Data are [%] of Patients with Fibrosis >45

Role of Liver Biopsy n AGAINST Prognosis usually good n No effective treatment n Risk n Cost n n IN FAVOR Excludes other diseases n Identifies type of fatty liver n Estimates prognosis n

Clinical Predictors of Fibrosis in NAFLD n n Ratziu et al. Gastroenterology, 2000 93 overweight [BMI >25] patients with NAFLD Septal fibrosis 30% Cirrhosis 11% Variable OR 95% CI P Age 50 BMI 28 Serum triglycerides 1. 7 ALT 2 N 14. 1 5. 7 5 4. 6 3. 7 - 54 1. 6 - 20 1. 4 - 17 1. 3 - 16 <0. 001 0. 006 0. 01 0. 02

Non-Invasive Markers of Fibrosis in NASH n The BARG [Harrison et al. Hepatology 2003 abstract] n Scoring system for advanced fibrosis [stage 3 -4] n BMI 28 n AST/ALT 0. 8 n Hgb. A 1 c > 5. 2 n Sensitivity 71% & specificity 76% n PPV 64% & NPV 81%

Diagnosis

Keys to the Diagnosis of NASH Clinical n n History of chronic mild elevation of AST/ALT Exclude xs ETOH use by corroborated hx n n n W: >20 g daily [17 oz beer, 7 oz wine, 2 oz distilled spirits] M: >60 g daily [50 oz beer, 20 oz wine, 5 oz distilled spirits] Asymptomatic or nonspecific constitutional symptoms – Fatigue most common sx No stigmata of chronic liver disease [except NASH cirrhosis] – 15% of pt may present for the first time with features of cirrhosis Exclude medications & dietary abnormalities by history
![Keys to the Diagnosis of NASH 2 Laboratory v v AST ALT 2 Keys to the Diagnosis of NASH [2] Laboratory v v AST & ALT 2](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-19.jpg)
Keys to the Diagnosis of NASH [2] Laboratory v v AST & ALT 2 -4 x ULN [AST: ALT <1] Remainder of LFTs nl/near nl v HBs. Ag, anti-HCV, ASMA & AMA [-] ANA 1/320 v NL ceruloplasmin, 1 -antitrypsin & transferrin saturation v Radiology [not required for Dx] v v v U/S – bright [hyperechoic/ echogenicity] liver CT-scan – low-density liver compared with spleen [darker] MRI- focal fat appears bright on T 1 -weighted imaging Liver Bx

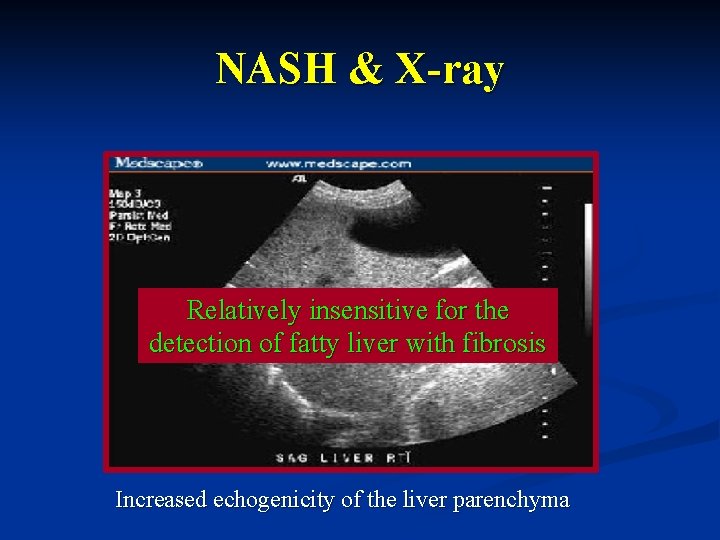
NASH & X-ray Relatively insensitive for the detection of fatty liver with fibrosis Increased echogenicity of the liver parenchyma

Natural History
![NAFLDNASH Prevalence Population Screening Autopsy Morbid Obesity BMI 35 NAFLD 15 29 15 NAFLD/NASH Prevalence [%] Population Screening Autopsy Morbid Obesity [BMI >35] NAFLD 15 -29 15](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-22.jpg)
NAFLD/NASH Prevalence [%] Population Screening Autopsy Morbid Obesity [BMI >35] NAFLD 15 -29 15 -25 71 -100 NASH 1. 2 2. 1 -2. 4 25 -50 • Highest prevalence 40 -49 yrs age • M=F • DM: NAFLD 50% • Morbidly obese & DM: NAFL 100%; NASH 50%; Cirrhosis 19%

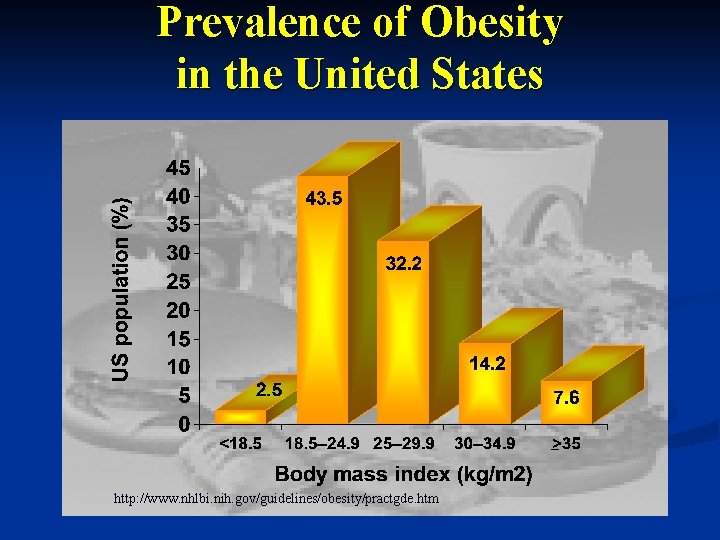
Prevalence of Obesity in the United States _ http: //www. nhlbi. nih. gov/guidelines/obesity/practgde. htm

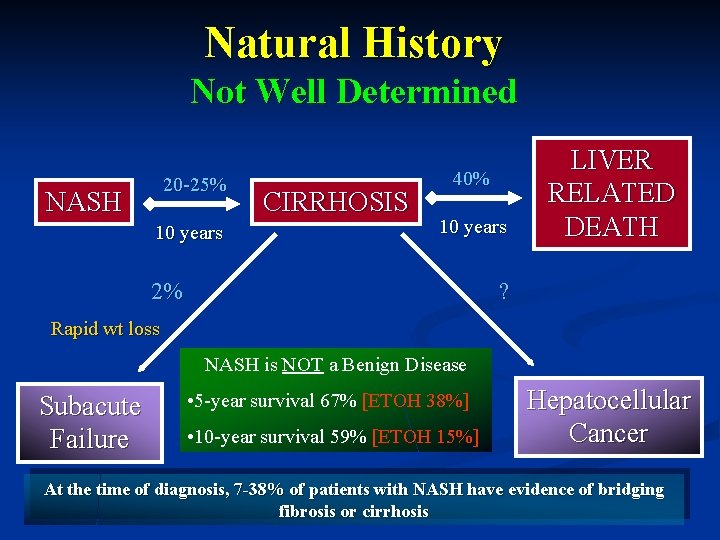
Natural History Not Well Determined 20 -25% NASH 10 years CIRRHOSIS 40% 10 years 2% LIVER RELATED DEATH ? Rapid wt loss NASH is NOT a Benign Disease Subacute Failure • 5 -year survival 67% [ETOH 38%] • 10 -year survival 59% [ETOH 15%] Hepatocellular Cancer At the time of diagnosis, 7 -38% of patients with NASH have evidence of bridging fibrosis or cirrhosis

Pathophysiology
![Conditions Associated with NAFLDNASH n Chronic Metabolic Syndrome 10 NASH n n n Conditions Associated with NAFLD/NASH n Chronic Metabolic Syndrome [“ 10 NASH”] n n n](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-26.jpg)
Conditions Associated with NAFLD/NASH n Chronic Metabolic Syndrome [“ 10 NASH”] n n n JI bypass TPN Rapid wt loss Drugs & Toxins n Over weight DM Dyslipidemia Subacute Metabolic Syndromes n n n Corticosteroids Tamoxifen Amiodarone Industrial Solvents Inherited Metabolic Diseases n n n Lipodystrophy [insulin resistance] Abetalipoproteinemia Wilson’s disease

Pathogenesis of NASH Environment & Genetics Nutrition NASH Genome
![Pathogenesis of NASH 2 Environment Socioeconomics Public Problems Nutritional Practices ObesityDM Hepatic Steatosis NASH Pathogenesis of NASH [2] Environment Socioeconomics Public Problems Nutritional Practices Obesity/DM Hepatic Steatosis NASH](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-28.jpg)
Pathogenesis of NASH [2] Environment Socioeconomics Public Problems Nutritional Practices Obesity/DM Hepatic Steatosis NASH
![Pathogenesis of NASH 3 Genetic Determinants n n n STEATOSIS ObesityLipodystrophy Insulin signaling n Pathogenesis of NASH [3] Genetic Determinants n n n STEATOSIS Obesity/Lipodystrophy Insulin signaling n](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-29.jpg)
Pathogenesis of NASH [3] Genetic Determinants n n n STEATOSIS Obesity/Lipodystrophy Insulin signaling n IKK [inhibitor kappa beta kinase enzyme that induces TNF & other cytokines insulin resistance] n n n Fatty acid metabolism n n PPAR [peroxisomes] MTP AOX n OXIDATIVE STRESS Iron Homeostasis n Cytochrome P 450 n n n HFE CYP 2 E 1 Inflammation Cytokines n n TNF Fibrogenesis
![Pathogenesis of NASH 4 The Two Hit Hypothesis Normal Steatosis VULNERABLE Inflammation Fibrosis Pathogenesis of NASH [4] The “Two Hit” Hypothesis Normal Steatosis VULNERABLE Inflammation & Fibrosis](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-30.jpg)
Pathogenesis of NASH [4] The “Two Hit” Hypothesis Normal Steatosis VULNERABLE Inflammation & Fibrosis 1 st “HIT” 2 nd “HIT” [fat accumulation in the liver] [cell injury {oxidative stress} & fibrosis] Insulin resistance oxidative stress [CYP 2 E 1, iron] [superoxide, hydroxyl & hydroxyethyl radicals] Hyperinsulinemia Hepatic leptin resistance Lipid peroxidation [hydrogen peroxide] TNF [endotoxin from the gut] FFA flux into the liver TGF FFA oxidant Leptin Triglyceride export
![Pathogenesis of NASH 5 Fat Accumulation in the Liver Increased Fatty Acid Influx Pathogenesis of NASH [5] Fat Accumulation in the Liver Increased Fatty Acid Influx •](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-31.jpg)
Pathogenesis of NASH [5] Fat Accumulation in the Liver Increased Fatty Acid Influx • Obesity • Hyperinsulinemia • Insulin resistance • Diet Decreased Fatty Acid Oxidation Triglyceride • Genetic Deficiencies • Leptin deficiency/resistance • Drugs Increased Fatty Acid Synthesis • Hyperinsulinemia • XS CHO Feeding • Leptin deficiency/resistance VLDL • Genetic deficiencies • Drugs • Insulin resistance
![Pathogenesis of NASH 6 Insulin Resistance Obesity Hyperlipidemia Disrupted Insulin Signaling Insulin Resistance Hyperinsulinemia Pathogenesis of NASH [6] Insulin Resistance Obesity Hyperlipidemia Disrupted Insulin Signaling Insulin Resistance Hyperinsulinemia](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-32.jpg)
Pathogenesis of NASH [6] Insulin Resistance Obesity Hyperlipidemia Disrupted Insulin Signaling Insulin Resistance Hyperinsulinemia Diabetes Lipolysis in peripheral adipose tissue Hepatic FFA production Suppression of hepatic FFA oxidation FFA
![Pathogenesis of NASH 7 Molecular Mechanisms for NAFLDrelated Insulin Resistance IKK inhibitor kappa beta Pathogenesis of NASH [7] Molecular Mechanisms for NAFLD-related Insulin Resistance IKK [inhibitor kappa beta](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-33.jpg)
Pathogenesis of NASH [7] Molecular Mechanisms for NAFLD-related Insulin Resistance IKK [inhibitor kappa beta kinase] Insulin Resistance NF- B [nuclear factor kappa beta] TNF activates IKK which initiates a self-reinforcing positive feedback loop that perpetuates insulin resistance & the production of TNF NAFLD

Treatment

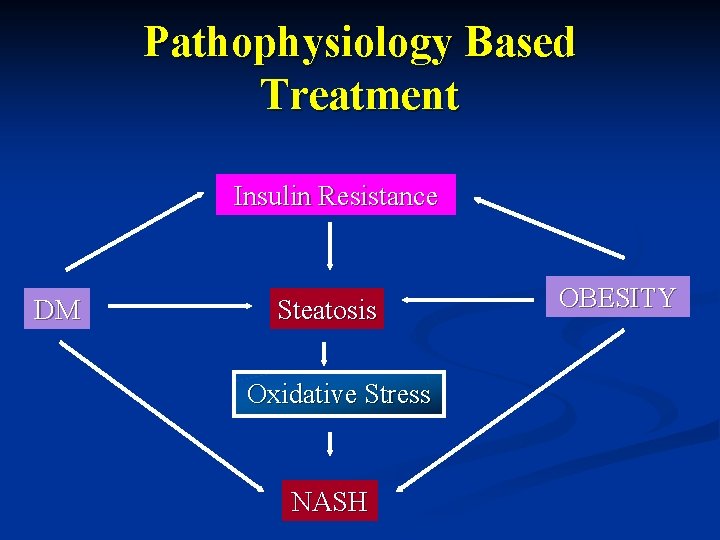
Pathophysiology Based Treatment Insulin Resistance DM Steatosis Oxidative Stress NASH OBESITY

Treatment General Consideration n Hepatotoxic agents should be avoided n Etoh n Drugs n Hydrocarbon solvents n Appetite suppressive agents are not recommended n Need for liver biopsy n Treat co-morbidities
![Treatment 2 Treat Comorbidities n DM n n No data Lipids n Clofibrate ineffective Treatment [2] Treat Co-morbidities n DM n n No data Lipids n Clofibrate ineffective](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-37.jpg)
Treatment [2] Treat Co-morbidities n DM n n No data Lipids n Clofibrate ineffective n Gemfibrozil may work n HMGCo. A reductase inhibitors efficacy unknown n Pt with LFT do NOT susceptibility to hepatotoxicity from Statins n n Chalasani et al Gastroenterology 2004 Wt loss
![Treatment 3 Wt loss n Diet 4 studies n n n Histologic exacerbation if Treatment [3] Wt loss n Diet [4 studies] n n n Histologic exacerbation if](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-38.jpg)
Treatment [3] Wt loss n Diet [4 studies] n n n Histologic exacerbation if wt loss >1. 6 g/wk n n n Starvation-induced flux of fat from peripheral stores to the liver Luyckx et al. Diabetes Metab 1998 Diet & Exercise [5 studies] n n n ALT improves Histologic data are inconsistent Both ALT & steatosis improve More effective than wt loss alone Orlistat [lipase inhibitor]

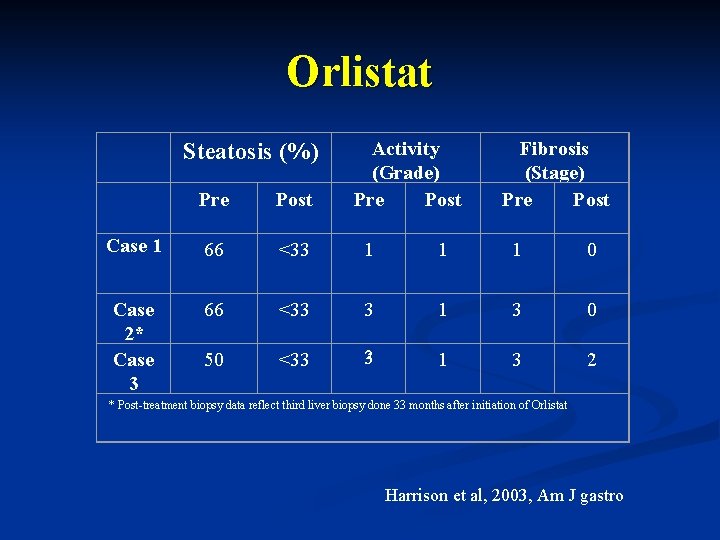
Orlistat Steatosis (%) Activity (Grade) Pre Post Fibrosis (Stage) Pre Post Case 1 66 <33 1 1 1 0 Case 2* Case 3 66 <33 3 1 3 0 50 <33 33 1 3 2 * Post-treatment biopsy data reflect third liver biopsy done 33 months after initiation of Orlistat Harrison et al, 2003, Am J gastro
![Treatment 4 Wt loss Guidelines n 10 wt loss n n n Gradual 312 Treatment [4] Wt loss Guidelines n 10% wt loss n n n Gradual [31/2](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-40.jpg)
Treatment [4] Wt loss Guidelines n 10% wt loss n n n Gradual [31/2 lbs per week] Diet [individualized] n Reduced caloric + balanced n AHA n Low glycemic diet Graduated exercise program
![Treatment 5 Wt loss v Bariatric Surgery v Jejunoileal bypass causes NASH v Treatment [5] Wt loss v Bariatric Surgery: v Jejunoileal bypass – causes NASH v](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-41.jpg)
Treatment [5] Wt loss v Bariatric Surgery: v Jejunoileal bypass – causes NASH v Laparoscopic adjustable gastric banding [LAGB] v Dixon et al. Hepatology 2004 v 36 pt: baseline liver biopsy: NASH in 23 & steatosis in 12 v Mean wt loss 34± 17 kg v F/U bx 25. 6± 10 mo: v Significant improvement in lobular steatosis, necroinflammatory changes and fibrosis [p<0. 001 for all] v Only 4 of the repeat bx had NASH
![Treatment 6 Potential Pharmacologic Treatment n Insulinsensitizing agents n n n Lipidlowering agents n Treatment [6] Potential Pharmacologic Treatment n Insulin-sensitizing agents: n n n Lipid-lowering agents: n](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-42.jpg)
Treatment [6] Potential Pharmacologic Treatment n Insulin-sensitizing agents: n n n Lipid-lowering agents: n n n Thiazolidinediones Metformin HMG-Co. A RIs Fibrates Cytoprotective agents n Ursodeoxycholic acid n Anti-oxidants: n n n Vitamin E Hepatic iron reduction Betaine S-adenosyl-methionine N-acetyl cysteine Probucol
![Treatment 7 NASH InsulinSensitizing Agents PI Treatment Caldwell Troglitazone Am J Gastro 2001 Treatment [7] NASH & Insulin-Sensitizing Agents PI Treatment Caldwell Troglitazone Am J Gastro 2001](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-43.jpg)
Treatment [7] NASH & Insulin-Sensitizing Agents PI Treatment Caldwell Troglitazone Am J Gastro 2001 [Rezulin] Nair Metformin N Duration 10 4 -6 mo ↓inflammation 15 12 mo ↓ALT/AST & IR [3 mo] No improvement after 3 mo Aliment Pharmacol Ther 2004 Sanyal Hepatology 2002 Tetri Hepatology 2003 Pioglitazone 21 6 mo ±Vit E Rosiglitazone 22 48 weeks [Avandia] 4 mg bid Promrat Pioglitazone Hepatology 2004 [Actos] 30 mg qd Histology/Outcome ↓steatosis, ballooning ↓steatosis, inflammation, ballooning & fibrosis [zone 3] Within 6 months of stopping treatment: ALT Wt gain in 67% [peripheral fat] 18 48 weeks 66% ↓steatosis, inflammation, ballooning & fibrosis Nl ALT in 72%
![Treatment 8 NASH Vitamin E PI Treatment Lavine Vit E J Pediatr 2000 Treatment [8] NASH & Vitamin E PI Treatment Lavine Vit E J Pediatr 2000](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-44.jpg)
Treatment [8] NASH & Vitamin E PI Treatment Lavine Vit E J Pediatr 2000 400 -1200 IU Hasegawa Sanyal Clin Gastro Hepatol 2004 Kugelmas Harrison N Duration Histology/Outcome 11 4 -10 mo N/A, ↓ALT α tocopherol 12 12 mo ↓ 5/9 steatosis, inflammation & fibrosis Pioglitazone ±Vit E 21 6 mo Vit E alone improved steatosis Combination improved steatosis, ballooning & pericellular fibrosis Exercise 16 12 weeks N/A, ↓ALT ±Vit E No difference Similar ↓ fibrosis in both groups Vit E + C vs. 23 6 mo placebo

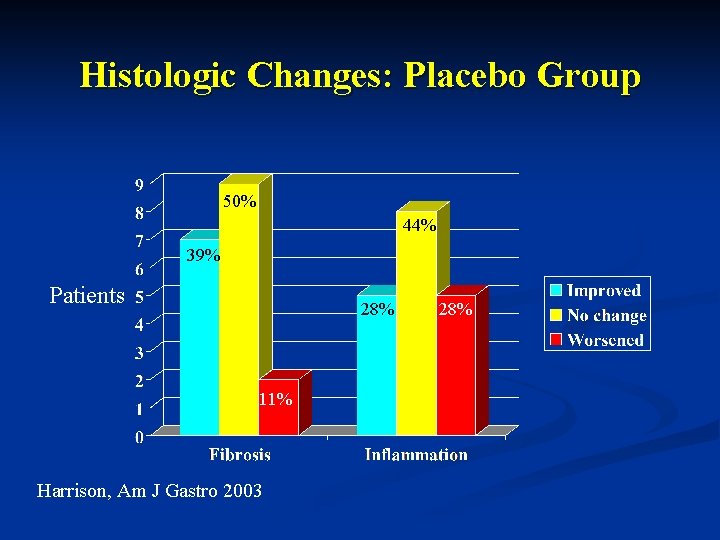
Histologic Changes: Placebo Group 50% 44% 39% Patients 28% 11% Harrison, Am J Gastro 2003 28%

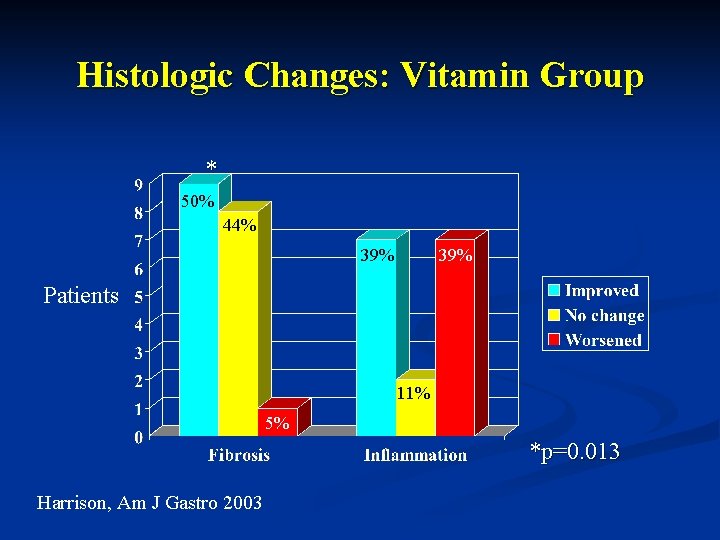
Histologic Changes: Vitamin Group * 50% 44% 39% Patients 11% 5% *p=0. 013 Harrison, Am J Gastro 2003
![Treatment 9 Cytoprotective Agents n Ursodeoxycholic acid UCDA Ref N Study Type Duration ASTALT Treatment [9] Cytoprotective Agents n Ursodeoxycholic acid [UCDA] Ref N Study Type Duration AST/ALT](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-47.jpg)
Treatment [9] Cytoprotective Agents n Ursodeoxycholic acid [UCDA] Ref N Study Type Duration AST/ALT Histology Laurin, 1996 24 Open label 12 mo Improved Steatosis Improved Guma, 1997 24 Randomized, open 6 mo Improved Not performed Ceriani, 1998 31 Open label 6 mo Improved Not performed
![Treatment 9 NASH Ursodeoxycholic Acid n Lindor et al Hepatology 2004 n Placebocontrolled Treatment [9] NASH & Ursodeoxycholic Acid n Lindor et al Hepatology 2004 n Placebo-controlled](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-48.jpg)
Treatment [9] NASH & Ursodeoxycholic Acid n Lindor et al Hepatology 2004 n Placebo-controlled trial n n 174 liver Bx-proven NASH [166 analyzed] n 2 year study n LFT and histology end points n UDCA 13 -15 mg/kg/d Follow-up pathology in 95 persons
![Treatment 10 NASH Ursodeoxycholic Acid n Safe welltolerated n 40 steatosis improved Treatment [10] NASH & Ursodeoxycholic Acid n Safe & well-tolerated n 40% steatosis improved](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-49.jpg)
Treatment [10] NASH & Ursodeoxycholic Acid n Safe & well-tolerated n 40% steatosis improved spontaneously n 20% fibrosis improved spontaneously [? Sampling variability] n No difference in biochemistries & liver histology n UDCA at a dose of 13 -15 mg/kg/d was NOT effective in pt with NASH
![Treatment 11 Summary n Reduce insulin resistance n n n Fibrates Statins Lipotropic Agents Treatment [11] Summary n Reduce insulin resistance n n n Fibrates Statins Lipotropic Agents](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-50.jpg)
Treatment [11] Summary n Reduce insulin resistance n n n Fibrates Statins Lipotropic Agents n n Betaine Choline Reduce Oxidant Stress n n n Lipid Lowering Agents n n Wt loss Thiazolidinediones Metformin n Reduce TNF n n n Hepatic iron reduction Vit E Antibiotics Probiotics Cytoprotection n Ursodiol

NASH & OLTx 1 -2% of OLTx n Post-OLTx n NASH recurs in 30% n Causes multifactorial: n Persistent hypertriglyceridemia n Obesity n DM n Corticosteroid – promote fatty liver & DM n Cyclosporine – mitochondrial toxicity n

Conclusions
![NAFLD NAFL NASH Cirrhosis Steatosis Fibrosis macrovesicular Inflammation Ballooning Steatosis Mallorys Hyaline Hepatocellular Cancer NAFLD NAFL NASH Cirrhosis Steatosis Fibrosis [macrovesicular] Inflammation Ballooning Steatosis Mallory’s Hyaline Hepatocellular Cancer](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-53.jpg)
NAFLD NAFL NASH Cirrhosis Steatosis Fibrosis [macrovesicular] Inflammation Ballooning Steatosis Mallory’s Hyaline Hepatocellular Cancer Fibrosis
![Current Treatment Algorithm Treat Comorbidity Confirm Histology Prognosis No Proven Treatment Vitamin E Current Treatment Algorithm Treat Co-morbidity Confirm Histology [Prognosis] No Proven Treatment [Vitamin E ?](https://slidetodoc.com/presentation_image_h/286c254bf19e51bf0fd73dd8471aa880/image-54.jpg)
Current Treatment Algorithm Treat Co-morbidity Confirm Histology [Prognosis] No Proven Treatment [Vitamin E ? ]

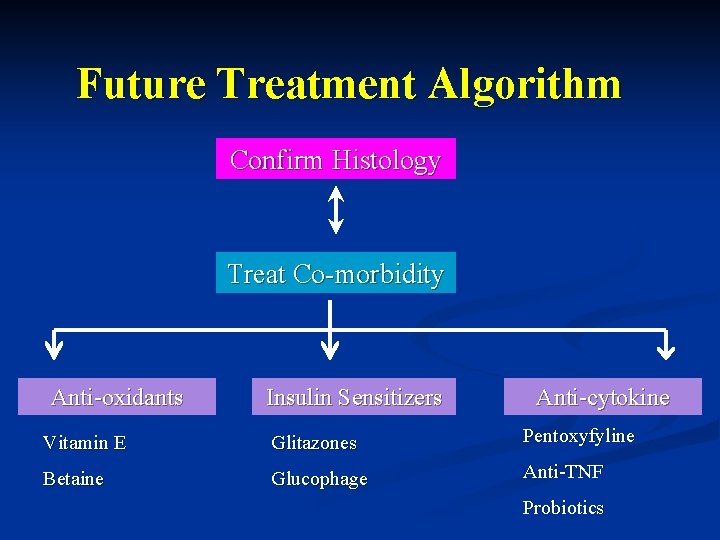
Future Treatment Algorithm Confirm Histology Treat Co-morbidity Anti-oxidants Insulin Sensitizers Anti-cytokine Vitamin E Glitazones Pentoxyfyline Betaine Glucophage Anti-TNF Probiotics

Steatosis & HCV

Steatosis & HCV Key Concepts n n Hepatic steatosis is a common finding in patients with HCV – 31% - 72% of such patients Hepatic steatosis more common in HCV G 3 [independent risk factor] development & progression of fibrosis in HCV patients Wt loss may diminish hepatic steatosis n n n Hickman et al Gut 2002 Eradication of HCV hepatic steatosis in G 3 pt but not G 1 [independent of any change in body weight] Degree of steatosis is inversely proportional to the likelihood of achieving response to therapy in the first 12 weeks n Poynard et al Hepatology 2003

Discussion