Nomenclature III Functional Groups These slides are best

Nomenclature III Functional Groups

• These slides are best when downloaded and run on powerpoint due to the animations.

Simple Names Base name: The name appears at the end of the name Suffix name: The name appears at the end of the name Prefix name: The name appears before the base name Some sound better than others If we can unambiguously draw a structure from your name, Then you will get a majority of the credit
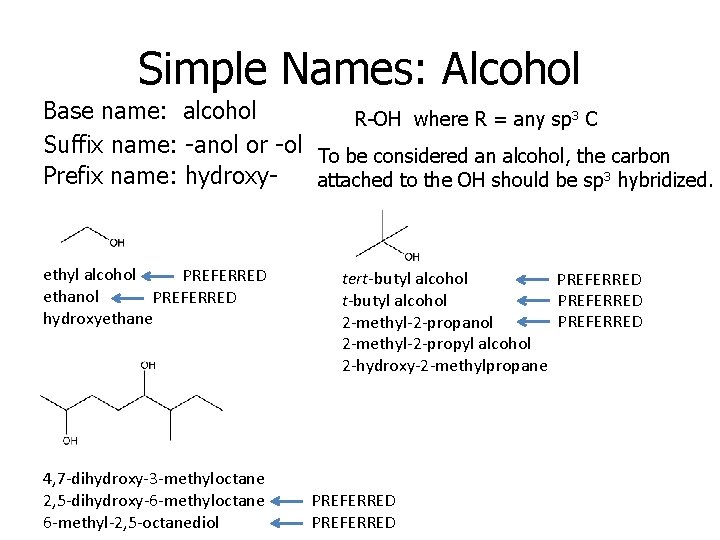
Simple Names: Alcohol Base name: alcohol R-OH where R = any sp 3 C Suffix name: -anol or -ol To be considered an alcohol, the carbon Prefix name: hydroxyattached to the OH should be sp 3 hybridized. ethyl alcohol PREFERRED ethanol PREFERRED hydroxyethane 4, 7 -dihydroxy-3 -methyloctane 2, 5 -dihydroxy-6 -methyloctane 6 -methyl-2, 5 -octanediol tert-butyl alcohol PREFERRED 2 -methyl-2 -propanol 2 -methyl-2 -propyl alcohol 2 -hydroxy-2 -methylpropane PREFERRED
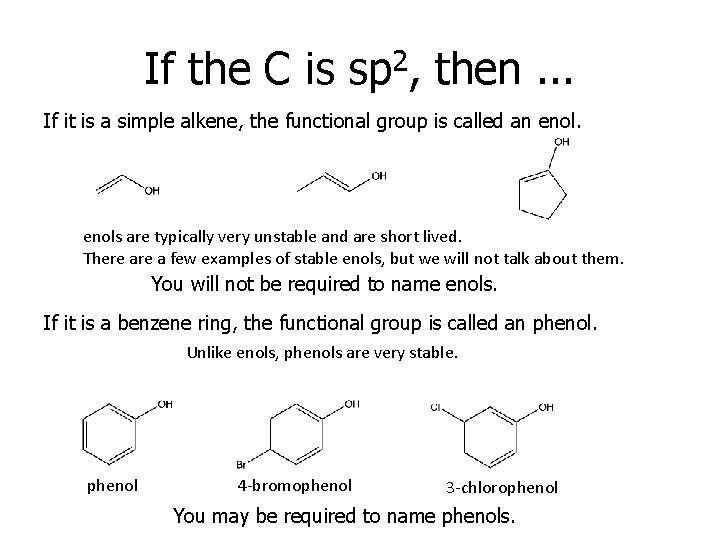
If the C is sp 2, then. . . If it is a simple alkene, the functional group is called an enols are typically very unstable and are short lived. There a few examples of stable enols, but we will not talk about them. You will not be required to name enols. If it is a benzene ring, the functional group is called an phenol. Unlike enols, phenols are very stable. phenol 4 -bromophenol 3 -chlorophenol You may be required to name phenols.

Simple Names: Amine Base name: amine Prefix name: amino. NH 3 To be considered an amine, the carbon attached to the N should be sp 3 hybridized. ammonia ethyl amine aminoethane where R = any sp 3 C or H PREFERRED N-isobutyl-methylamine N-methyl-2 -methylpropylamine N prefix is used to show group is attached to the nitrogen benzylamine N, N-diisopropylethylamine 1, 3 -diaminopropane
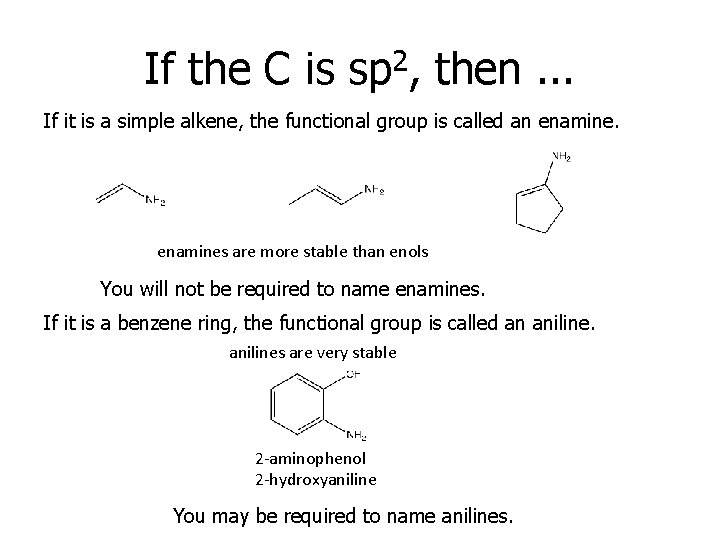
If the C is sp 2, then. . . If it is a simple alkene, the functional group is called an enamines are more stable than enols You will not be required to name enamines. If it is a benzene ring, the functional group is called an anilines are very stable 2 -aminophenol 2 -hydroxyaniline You may be required to name anilines.
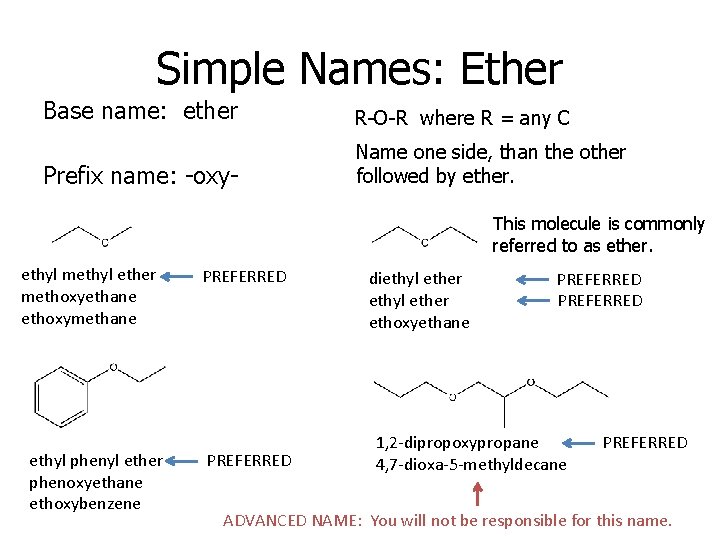
Simple Names: Ether Base name: ether R-O-R where R = any C Prefix name: -oxy- Name one side, than the other followed by ether. This molecule is commonly referred to as ether. ethyl methyl ether methoxyethane ethoxymethane ethyl phenyl ether phenoxyethane ethoxybenzene PREFERRED diethyl ether ethoxyethane PREFERRED 1, 2 -dipropoxypropane 4, 7 -dioxa-5 -methyldecane PREFERRED ADVANCED NAME: You will not be responsible for this name.

Simple Names: Carbonyls is a carbonyl. There are many functional groups that contain carbonyls: aldehydes, ketones, carboxylic acids, esters, amides
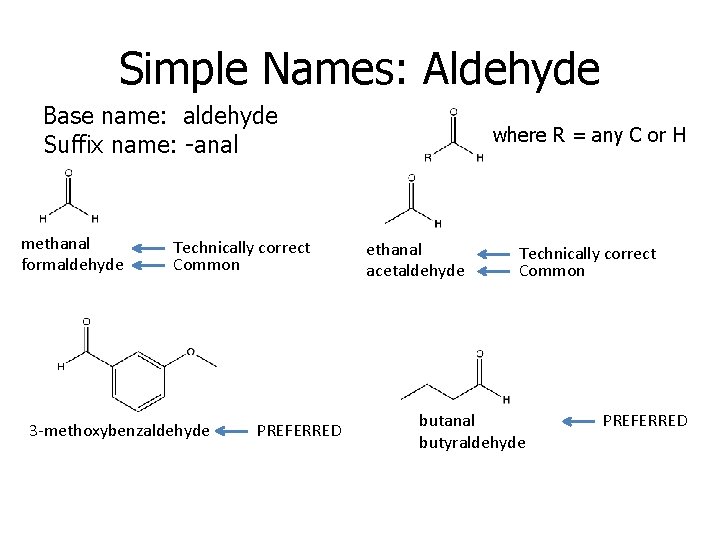
Simple Names: Aldehyde Base name: aldehyde Suffix name: -anal methanal formaldehyde Technically correct Common 3 -methoxybenzaldehyde PREFERRED where R = any C or H ethanal acetaldehyde Technically correct Common butanal butyraldehyde PREFERRED
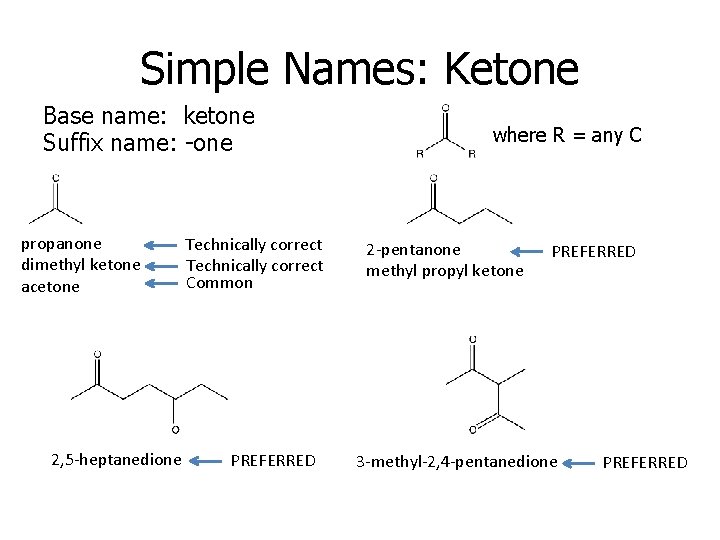
Simple Names: Ketone Base name: ketone Suffix name: -one propanone dimethyl ketone acetone 2, 5 -heptanedione Technically correct Common PREFERRED where R = any C 2 -pentanone methyl propyl ketone PREFERRED 3 -methyl-2, 4 -pentanedione PREFERRED
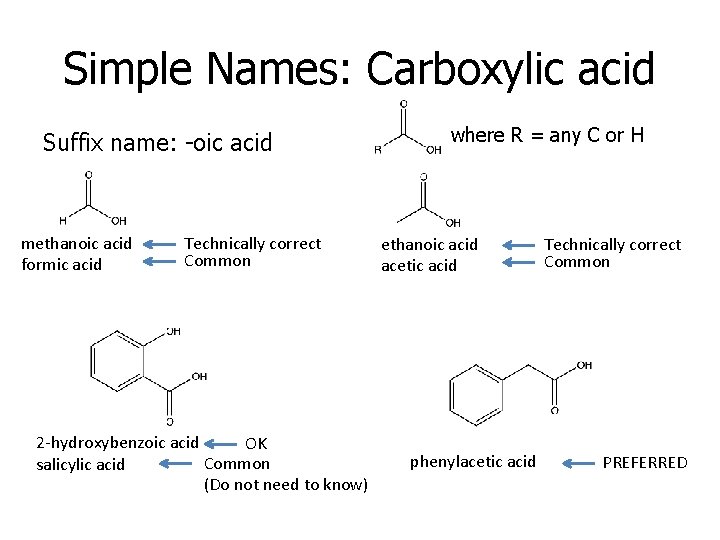
Simple Names: Carboxylic acid Suffix name: -oic acid methanoic acid formic acid Technically correct Common 2 -hydroxybenzoic acid OK Common salicylic acid (Do not need to know) where R = any C or H ethanoic acid acetic acid phenylacetic acid Technically correct Common PREFERRED
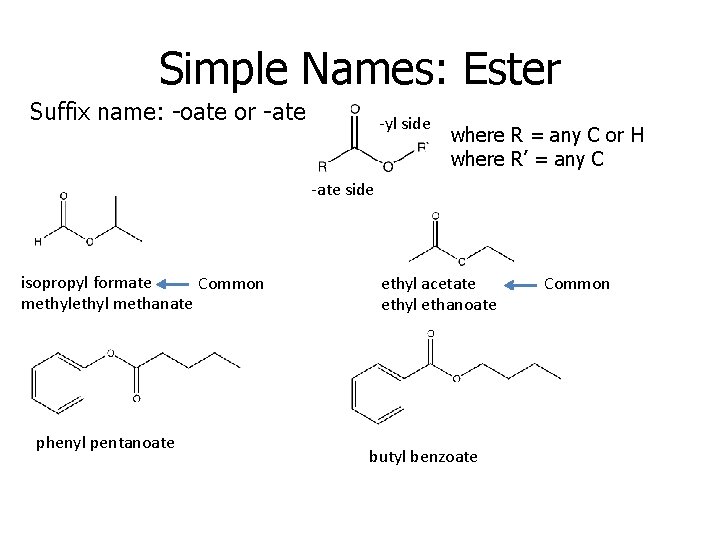
Simple Names: Ester Suffix name: -oate or -ate -yl side where R = any C or H where R’ = any C -ate side isopropyl formate Common methyl methanate phenyl pentanoate ethyl acetate ethyl ethanoate butyl benzoate Common
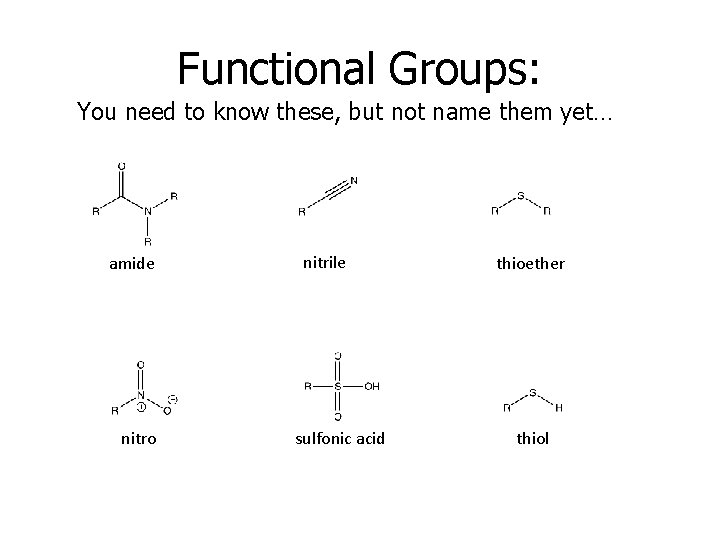
Functional Groups: You need to know these, but not name them yet… amide nitro nitrile sulfonic acid thioether thiol
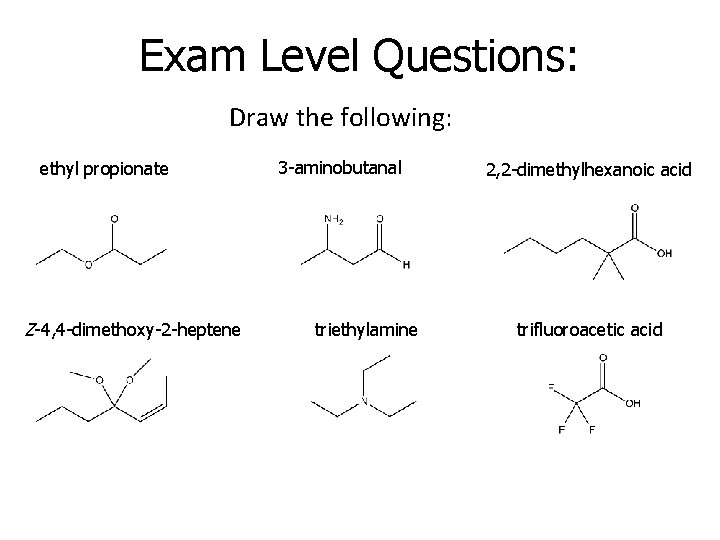
Exam Level Questions: Draw the following: ethyl propionate Z-4, 4 -dimethoxy-2 -heptene 3 -aminobutanal triethylamine 2, 2 -dimethylhexanoic acid trifluoroacetic acid
- Slides: 15