Nomenclature Chapter 5 Classifying Binary Compounds Compounds containing

Nomenclature Chapter 5

Classifying Binary Compounds • Compounds containing a metal and a nonmetal are – Type • Compounds containing two nonmetals – Type • Compounds containing H and a nonmetal =

Metal Cations • Type I – Metals that can only – Determine charge by • Type II – Metals that can have – Determine metal cation’s charge from
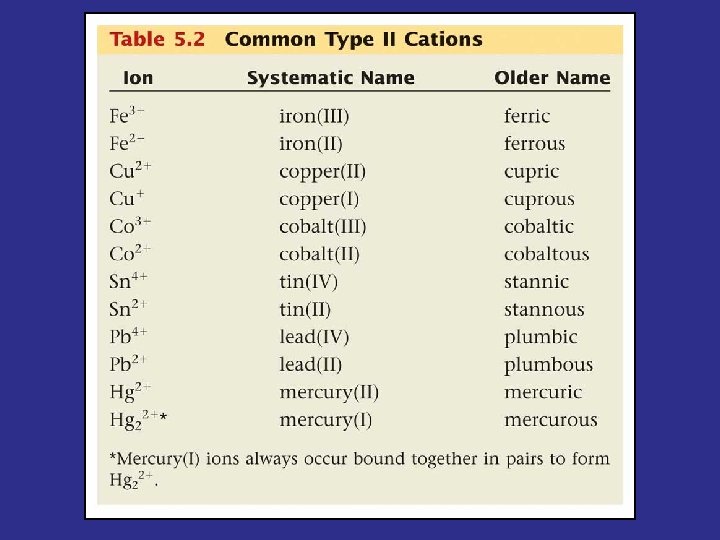

Naming Binary Type II Compounds • Cu+1 and N-3 • Iron (III) phosphide • Hg+2 and O-2 • Manganese (II) fluoride • Cr+3 and P-3 • Gold (I) telluride • Sn+4 and Cl-1 • Lead (IV) bromide • Ni+4 and S-2 • Cobalt (III) arsenide

Ionic Compounds • Sometimes you can use the reverse of the criss-cross method • When naming compounds,

Naming Ionic Compounds Examples • Au 2 S • Ni. S 2 • Mn. O • Cr 3 P 2 • Fe 3 N 2 • Pb. F 4 • Cu. Cl 2 • Hg. I

Type III - Binary Compounds of 2 Nonmetals • These binary compounds always start with a nonmetal or a metalloid. • Name first element in formula first, • Name the second element in the formula – However, remember

Type III Binary Molecular Compounds • Use a prefix in front of • Never use the prefix mono • Prefixes: 1 -mono, 2 -di, 3 -tri, 4 -tetra, 5 -penta, hexa, 7 -hepta, 8 -octa, 9 -nona, 10 -deca, 11 undeca, 12 -dodeca. • To write the formula for binary molecular compounds, 6 -

Binary Molecular Compounds Examples Naming Compounds Writing Formulas • Nitrogen trichloride • Si. F 2 • C 3 Cl 9 • Triphosphorus pentoxide • S 4 I 7 • Hexasulfur monofluoride • P 5 O 10 • Diselenium pentabromide
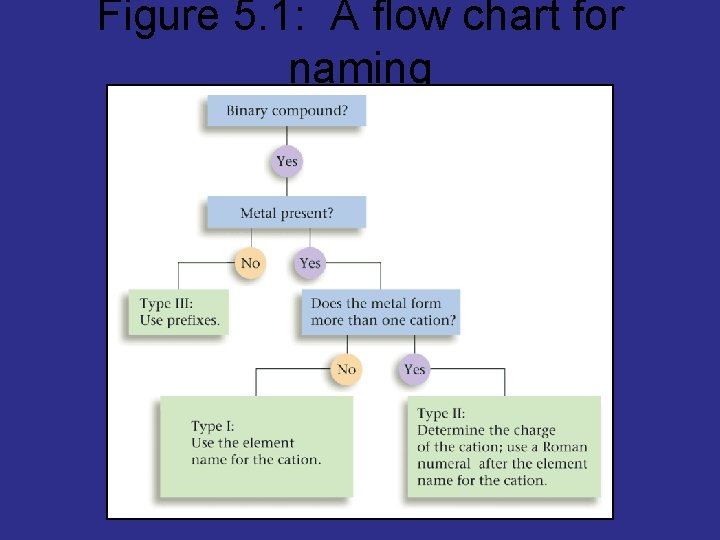
Figure 5. 1: A flow chart for naming binary compounds.

Ionic Compounds • Ternary ionic compounds – contain atoms of three or more different elements, usually a polyatomic ion. • Writing the formulas for ternary compounds is done in the same way as binary compounds. The polyatomic ions stays together though.

Ionic Compounds • When you need more than one polyatomic ion in your formula, put parentheses around the ion, and how many of them you need outside the parentheses as a subscript. • NEVER MOVE SUBSCRIPTS OF THE IONS, ONLY THE CHARGES!!!

Writing Formulas for Ternary Ionic Compound Examples • Calcium sulfate Ca+2 and SO 4 -2 • Sodium chlorate Na+1 and Cl. O 3 -1 • Magnesium hydroxide Mg+2 and OH-1 • Potassium phosphate K+1 and PO 4 -3 • Iron (III) carbonate Fe+3 and CO 3 -2 • Tin (IV) chromate Sn+4 and Cr. O 4 -2 • Nickel (II) dihydrogen phosphate Ni+2 and H 2 PO 4 -1 • Chromium (III) sulfate Cr+3 and SO 4 -2 • Copper (II) acetate Cu+2 and C 2 H 3 O 2 -1 • Iron (II) permanganate Fe+2 and Mn. O 4 -1

Ionic Compounds • When naming ternary compounds, name the cation (first symbol in the formula unless it is ammonium, NH 4+1) first, and then the rest of the formula, which will only have one name, unless it contains hydrogen.

Naming Ternary Ionic Compounds • Ca(NO 3)2 • Ni 3(PO 3)4 • KCl. O 3 • Fe(CN)2 • Ba. SO 3 • Mn(HCO 3)3 • Al. PO 4 • Au 2 CO 3 • Cu. OH • Cr 2 HPO 4


Acids Naming & Writing Formulas for Acids • All acids begin with a hydrogen, and are neutral compounds. • In all acids, the cation is the hydrogen ion, H+1. • Anions change their endings when they become acids.

Acids • -ide ions become hydro root ic acid • Ex: chloride becomes hydrochloric acid • -ate ions become root ic acid • Ex: nitrate becomes nitric acid • -ite ions become root ous acid • Ex: chlorite becomes chlorous acid

Acids Naming & Formula Writing Examples Naming Acids • H 3 PO 3 Writing Formulas for Acids • Sulfuric acid • HI • Hydroarsenic acid • HCN • Perchloric acid • H 2 C 4 H 4 O 6 • Hypoiodous acid

Hydrates Naming & Writing Formulas for Hydrates • Hydrates – compounds that have a certain number of water molecules attached to each formula unit. • To name hydrates, use the same prefixes you used for naming binary molecular compounds and add –hydrate after the prefix.

Hydrates • To write the formula for compounds containing hydrates you simply have to determine how many molecules are present by looking at the prefix written before the hydrate. • If you see a substance where it says it is anhydrous, that means it does not have any water molecules attached to each formula unit.

Naming and Formula Writing Examples for Hydrates • Cu. SO 4 5 H 2 O • Fe. Cl 2 3 H 2 O • Ca 3(PO 4)2 8 H 2 O • Sodium thiosulfate tetrahydrate • Magnesium hydroxide dihydrate • Stannic nitrate octahydrate
- Slides: 23