Nomenclature and Functional Groups Classifying organic compounds Hydrocarbons

Nomenclature and Functional Groups
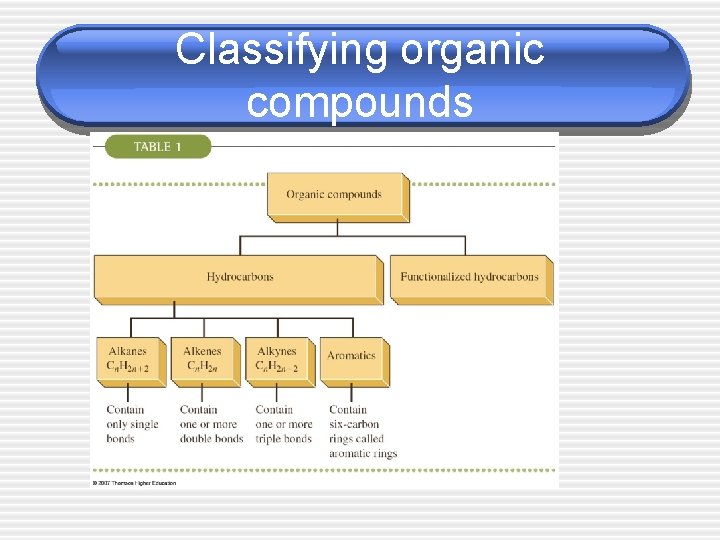
Classifying organic compounds

Hydrocarbons • _____ - Organic compounds that • • • contain only carbon & hydrogen _____ - contain only single covalent bonds _____ - contain one or more carbon - carbon double bond _____ - contain one or more carbon triple bond

Saturated & Unsaturated Hydrocarbons • Saturated hydrocarbons – contain only _____ carbon-carbon bonds (_____ ) • Unsaturated hydrocarbons – contain double carbon-carbon bonds (_____) or triple carbon-carbon (_____ ) bonds

Saturation bonding: Multiple bonds and unsaturation • Saturated: no more bonds can be • added Unsaturated: more bonds can be formed § More reactive compounds

Formulas • Alkanes = Cn. H 2 n+2 • Alkenes = Cn. H 2 n • Alkynes = Cn. H 2 n-2
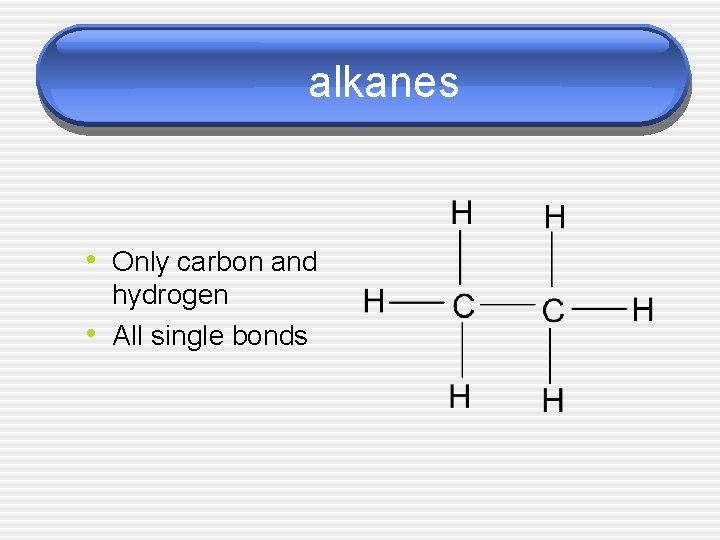
alkanes • Only carbon and • hydrogen All single bonds

Alkenes contain double bonds
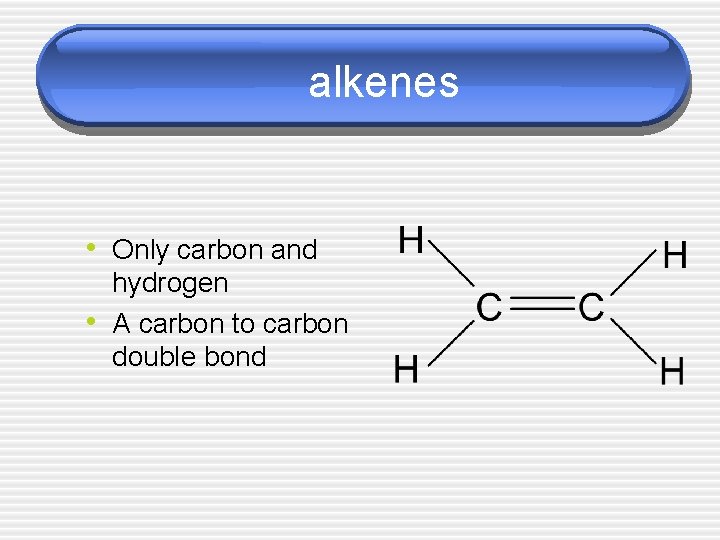
alkenes • Only carbon and • hydrogen A carbon to carbon double bond

Examples of alkynes

Isomerism • Same number and type of atoms • Different arrangements • Hydrocarbons can have straight and branched chains
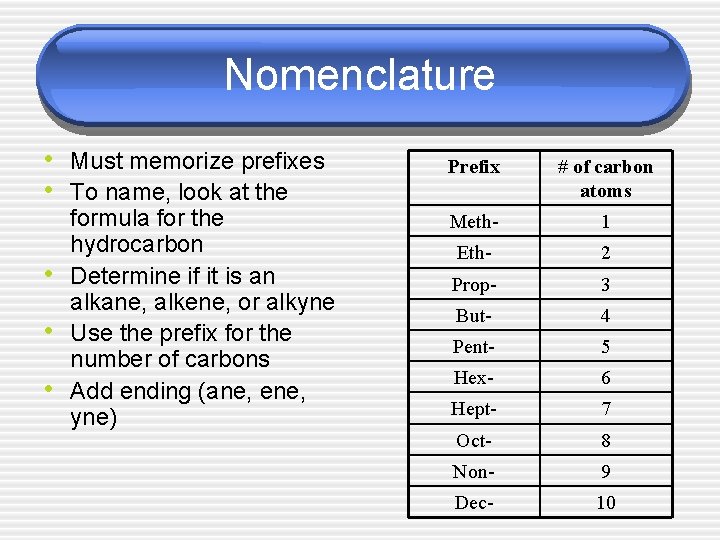
Nomenclature • Must memorize prefixes • To name, look at the • • • formula for the hydrocarbon Determine if it is an alkane, alkene, or alkyne Use the prefix for the number of carbons Add ending (ane, ene, yne) Prefix # of carbon atoms Meth- 1 Eth- 2 Prop- 3 But- 4 Pent- 5 Hex- 6 Hept- 7 Oct- 8 Non- 9 Dec- 10

Mnemonic for first four prefixes First four prefixes • • Meth. Eth. Prop. But- Monkeys Eat Peeled Bananas
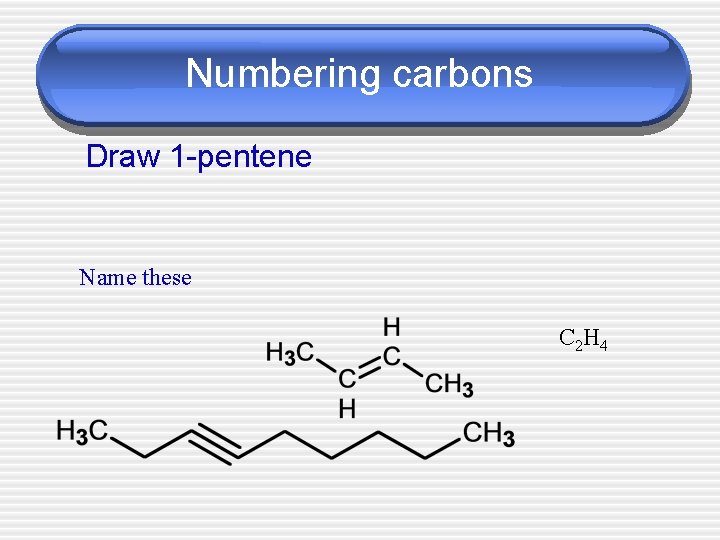
Numbering carbons Draw 1 -pentene Name these C 2 H 4
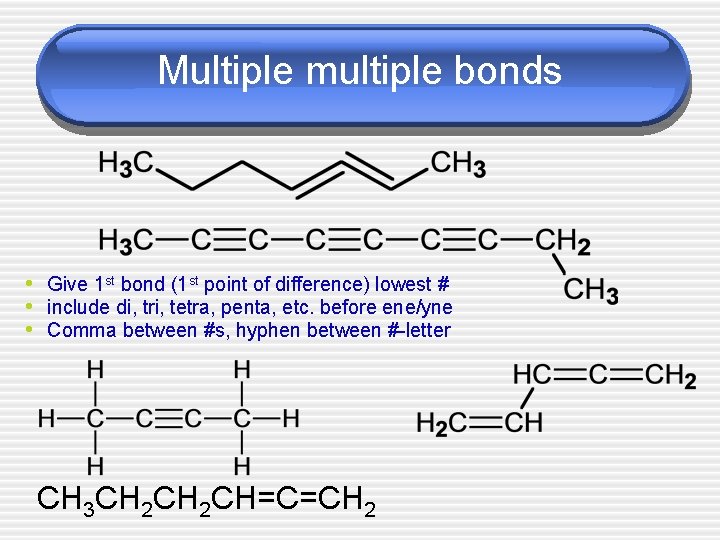
Multiple multiple bonds • Give 1 st bond (1 st point of difference) lowest # • include di, tri, tetra, penta, etc. before ene/yne • Comma between #s, hyphen between #-letter CH 3 CH 2 CH=C=CH 2
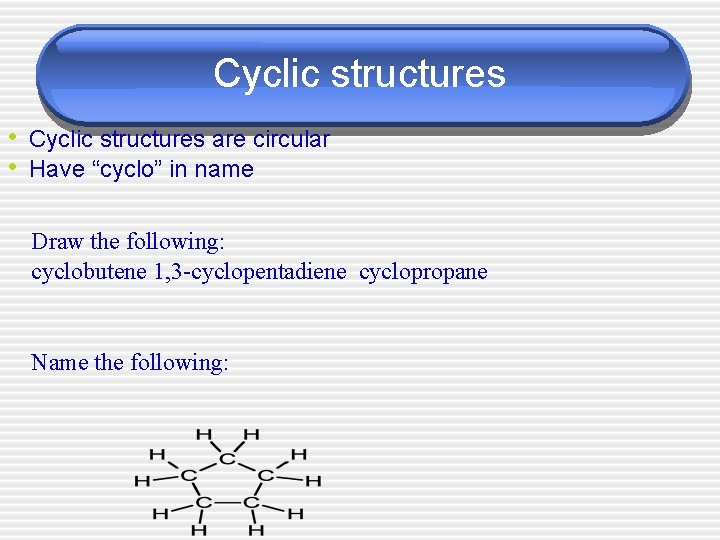
Cyclic structures • Cyclic structures are circular • Have “cyclo” in name Draw the following: cyclobutene 1, 3 -cyclopentadiene cyclopropane Name the following:
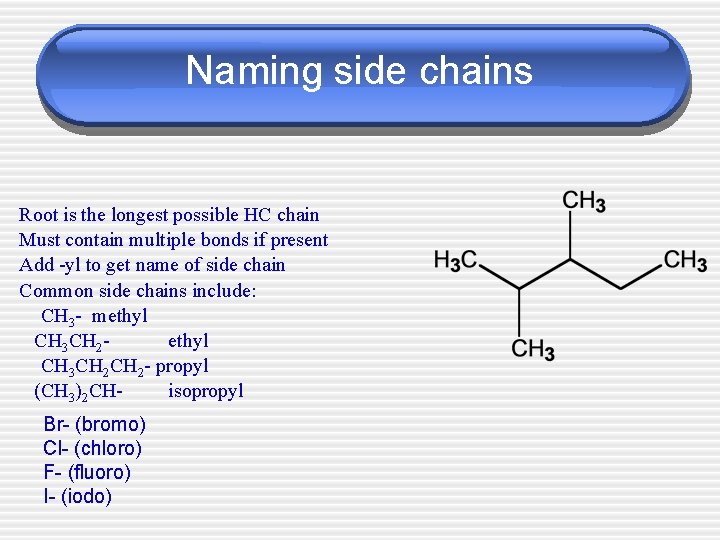
Naming side chains Root is the longest possible HC chain Must contain multiple bonds if present Add -yl to get name of side chain Common side chains include: CH 3 - methyl CH 3 CH 2 CH 2 - propyl (CH 3)2 CHisopropyl Br- (bromo) Cl- (chloro) F- (fluoro) I- (iodo)

Functional groups • Chemistry of organic compounds is • • determined by functional groups Functional group is an atom or group of atoms that are different from C Heteroatoms confer very different properties on the substance
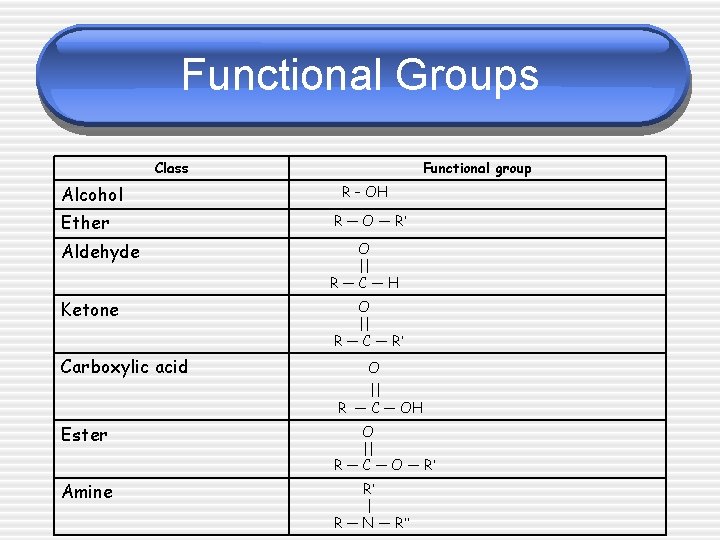
Functional Groups Class Alcohol Functional group R – OH Ether R — O — R’ Aldehyde O || R—C—H Ketone O || R — C — R’ Carboxylic acid O || R — C — OH Ester O || R — C — O — R’ Amine R’ | R — N — R’’
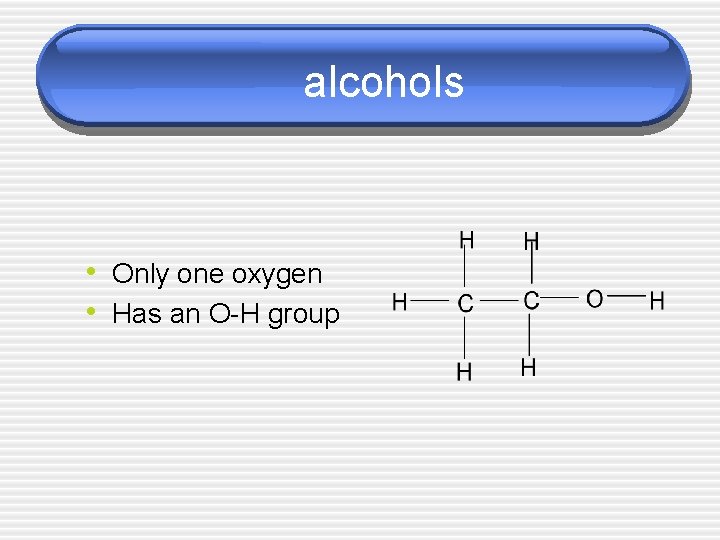
alcohols • Only one oxygen • Has an O-H group
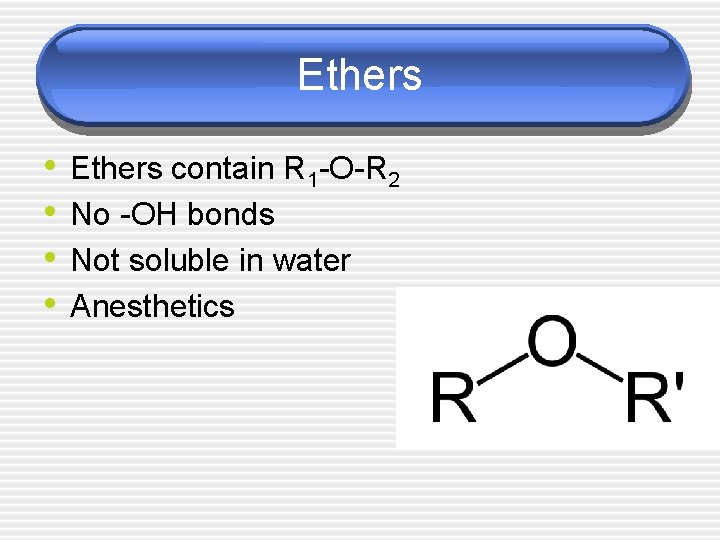
Ethers • • Ethers contain R 1 -O-R 2 No -OH bonds Not soluble in water Anesthetics
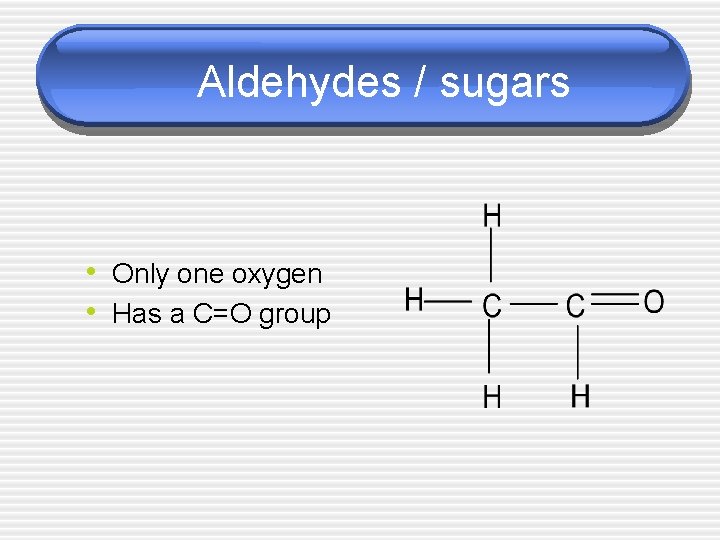
Aldehydes / sugars • Only one oxygen • Has a C=O group
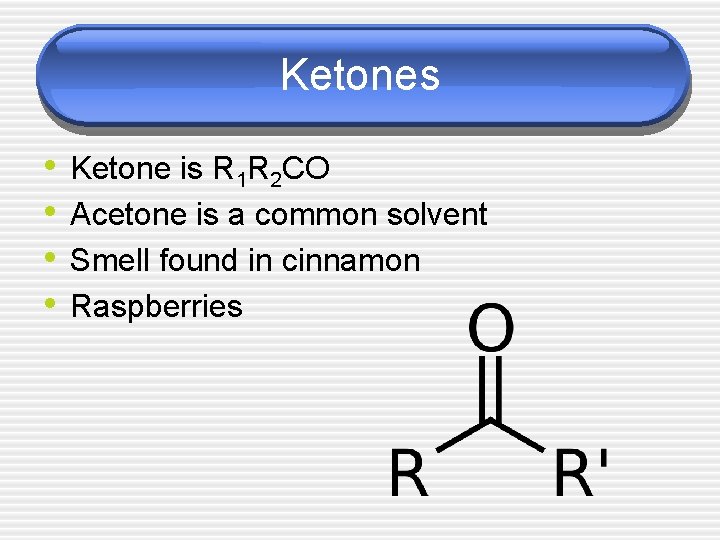
Ketones • • Ketone is R 1 R 2 CO Acetone is a common solvent Smell found in cinnamon Raspberries

Carboxylic acids • Commonly found in citrus fruits and any • sour foods Formic acid present in ant and bee stings

Esters • Has 2 oxygens • One oxygen is in a • carbon sandwich Ester loves to eat her carbon sandwich ?
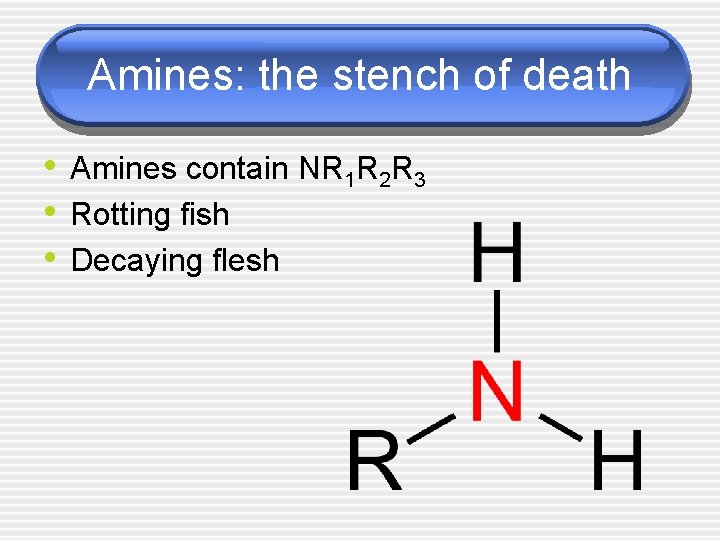
Amines: the stench of death • Amines contain NR 1 R 2 R 3 • Rotting fish • Decaying flesh

Alkyl halides • are a group of chemical compounds derived from alkanes containing one or more halogens.
- Slides: 27