Nomenclature aka Naming Compounds Unit 3 Binary Ionic

Nomenclature aka Naming Compounds Unit 3

Binary Ionic Compounds O A two-element compound consisting of a cation and an anion. O We will look at two different kinds of binary ionic compounds.

Type I Binary Ionic Compounds O

Rules for Naming Type I Ionic Compounds O

O

Examples: Type I Compounds Compound Ions Present Name Na. Cl Sodium Chloride KI Potassium Iodide Calcium Sulfide Ca. S Cs. Br Mg. O Cesium Bromide Magnesium Oxide

Practice O

Practice O Write the names for the following binary compounds. O Be. F 2 O Beryllium Fluoride O Na. I O Sodium iodide O Al. Br 3 O Aluminum bromide

Practice O Write the names for the following binary compounds. O Ra 3 N 2 O Radium nitride O K 2 S O Potassium sulfide O Sr. O O Strontium Oxide

Type II Binary Compounds O The metal present can form two or more cations that have different charges. O Follow the same rules for Type I compounds but include a Roman numeral O The Roman numeral tells the charge on the ion, not the number of ions present in the compound. O Metals that form only one cation do not need to be identified by a Roman numeral.

Ex: Type II Ionic Compounds O Cu. Cl O Cu = Cation (plus 1 charge) O Cl = Anion (negative 1 charge) O Name: Copper chloride

Ex: Type II Ionic Compounds O Hg. O O Hg = Cation (plus 2 charge) O O = Anion (negative 2 charge) O Name: Mercury (II) oxide

Rules for Naming: Type III Binary Compounds (Contain Only Nonmetals) The first element in the formula is named first, and the full element name is used. 2. The second element is named as though it were an anion 3. Prefixes are used to denote the numbers of atoms present. 1. O These prefixes are given in Table 4. 3 4. The prefix mono- is never used for naming the first element. O Ex. CO is called carbon monoxide, not monocarbon monoxide

Prefixes in Covalent Compounds Number of Atoms 1 2 3 4 5 Prefix monoditritetrapenta- Number of Atoms 6 7 8 9 10 Prefix hexaheptaoctanonadeca-

Naming Compounds That Contain Ployatomic Ions O You must memorize the names of the polyatomic ions O A polyatomic ion consisting of two or more atoms bound together. O Oxyanion – A polyatomic ion containing at least one oxygen atom and one or more atoms of at least one other element.

Naming Acids

O An acid is defined as a substance that gives off a hydrogen ion (H+) in water. Therefore all acids are made up of a hydrogen cation and either a monatomic or polyatomic anion. O Acid names are based on the anion ending.

Rules for Naming Acids 1. If the anion does not contain oxygen, the acid is named with the prefix hydro- and the suffix –ic attached to the root name for the element. O Example: Acid – HCl O Anion – Cl – O Name – hydrochloric acid
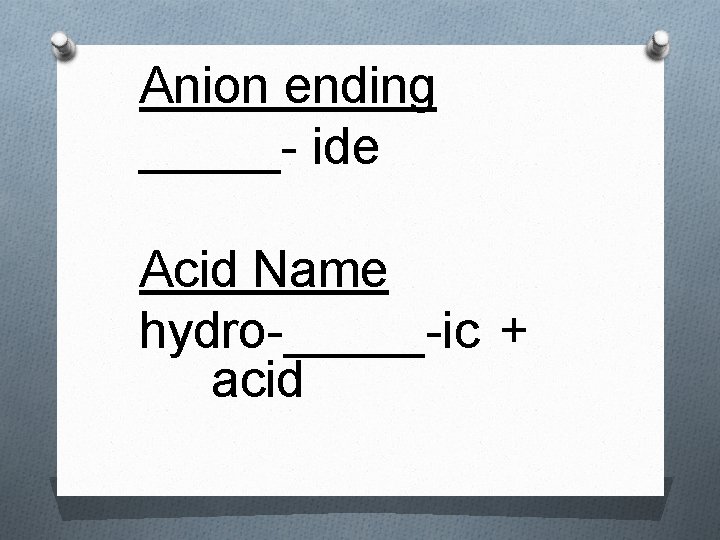
Anion ending _____- ide Acid Name hydro-_____-ic + acid
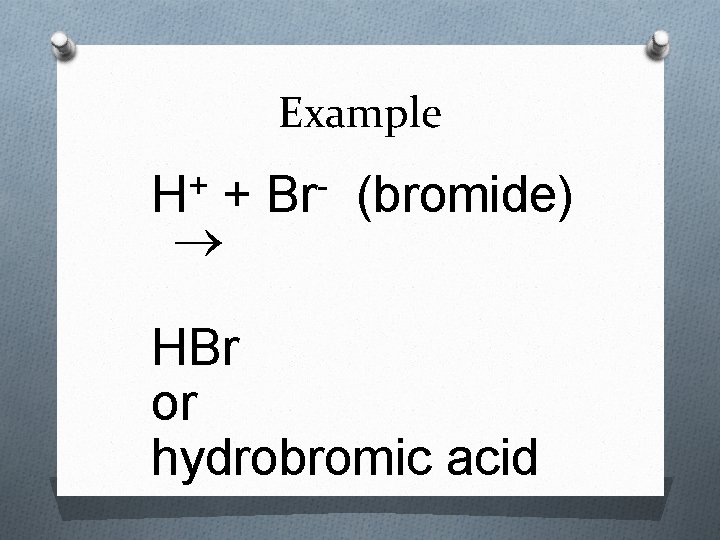
Example + H + Br (bromide) HBr or hydrobromic acid

Rules for Naming Acids 2. When the anion contains oxygen, the acid - name is formed from the root name of the central element of the anion or the anion name with a suffix of –ic or –ous. When the anion name ends in –ate, the suffix –ic is used. For example: Acid H 2 SO 4 Anion: sulfate Name: Sulfuric Acid
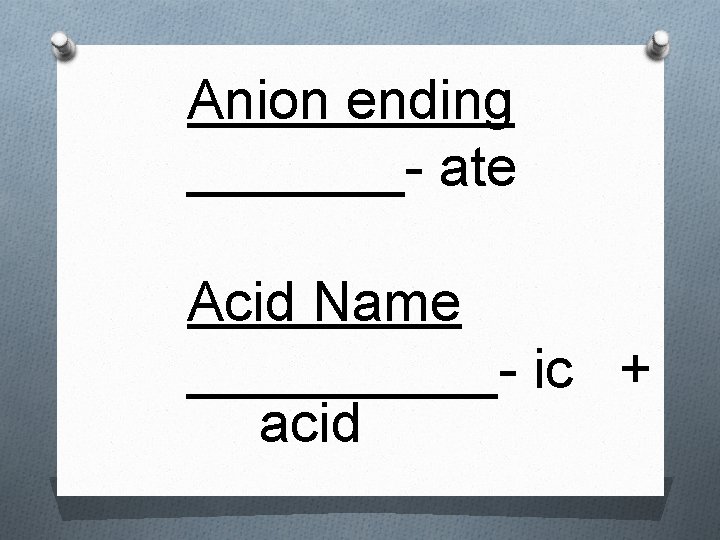
Anion ending _______- ate Acid Name _____- ic + acid
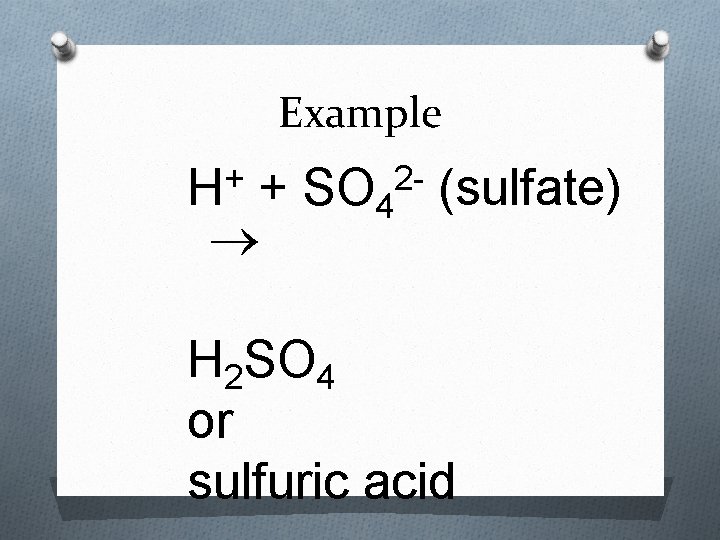
Example + H + SO 4 (sulfate) 2 - H 2 SO 4 or sulfuric acid

Rules for Naming Acids 3. When the anion name ends in –ite, the suffix –ous is used in the acid name - For example: Acid – H 2 SO 3 - Anion: sulfite - Name: Sulfurous acid
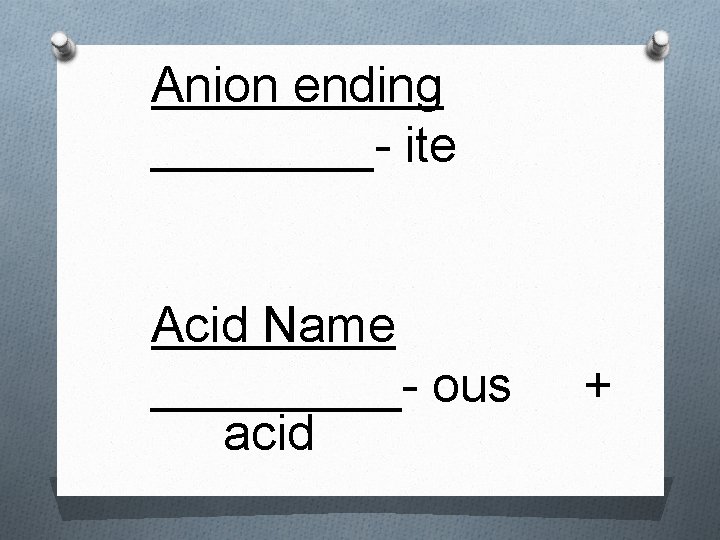
Anion ending ____- ite Acid Name _____- ous acid +
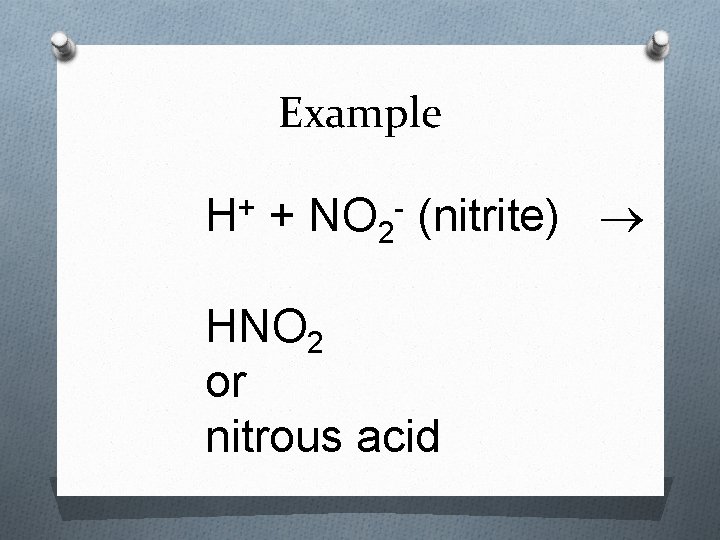
Example H+ + NO 2 - (nitrite) HNO 2 or nitrous acid
- Slides: 26