NOBLE GASES GROUP 8 A LINKS Helium Neon
NOBLE GASES GROUP 8 A LINKS Helium Neon Argon Krypton Xenon Radon
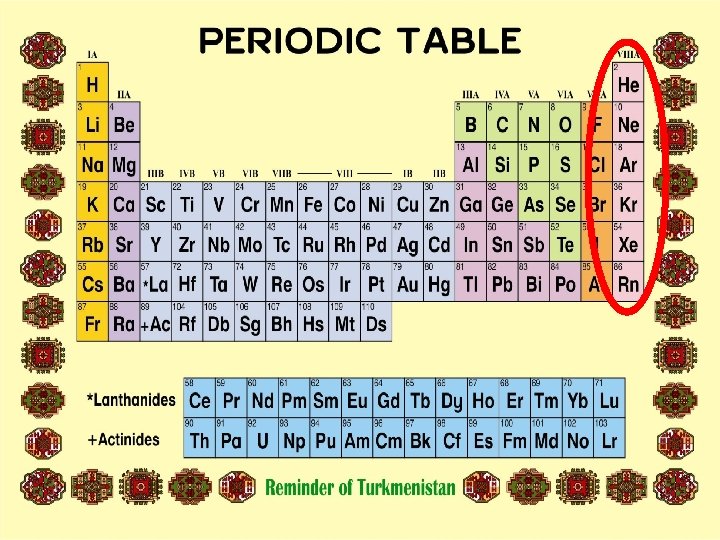

Group 8 A Helium 4 2 He Neon 20 10 Ne Argon 40 18 Ar Krypton 84 36 Kr Xenon 131 Radon 222 54 Xe 86 Rn

NOBLE GASES *They are all gases at room temperature. *They are colorless, odorless, water insoluble and monatomic. *Argon (Ar) is the most abundant. *Except He and Rn, they are produced by the fractional distillation of air. *Radon is obtained in small quantities from the radioactive decay of uranium. *They have no tendency to react chemically, that is why called as noble or inert gases. *Each period ends with noble gases in the periodic table.

NOBLE GASES *They have the most stable electron configuration. *They have the most ionization energy in the period, He have 1 st highest ionization energy and lowest freezing point in the periodic table. *They have different colors when exposed to electric current. *Neil Bartlett is the first chemist reacted Xe with Pt. F 6 and got the compound Xe. Pt. F 6.

They do not gain or lose electrons. e ee e More electrons, Sir ! Thanks, I am full

HELIUM (He) *Discovered in England in 1895 by Sir William Ramsay. *Used to inflate zeppelins, hovercrafts and weather balloons. *Used in artificial atmosphere, welding, mercury vapor lamps, rocket fuels, cooler and nuclear reactors.

NEON (Ne) *Discovered in England in 1898 by Sir William Ramsay. *No reaction. *Used in advertisement light, welding, lasers, lamps and cooler.

ARGON (Ar) *Discovered in Scotland in 1894 by Lord Rayleigh. *Has the most abundance in air. *No reaction. *Used in electric lamps and welding.

KRYPTON (Kr) *Discovered in Great Britain in 1898 by Sir William Ramsay. *Possible oxidation numbers, +2 and +4. *Used in photography, some special lamps and lasers.

XENON (Xe) *Discovered in England in 1898 by Sir William Ramsay. *Possible oxidation numbers, +2, +4 and +6. *Used in photography.

RADON (Rn) *Discovered in Germany in 1900 by Fredrich Ernst Dorm. *Has the smallest abundance in air. *Possible oxidation numbers, +2 and +4. *Radioactive.
- Slides: 12