Noble Gases Discovered He 1868 Ar 1894 Ne

Noble Gases

Discovered �He 1868; Ar 1894 �Ne, Kr and Xe in 1898 �Rn 1900 �Unreactive because they have a full outer shell of electrons �Called Noble then Inert now Noble

Uses Helium �Airships as it is lighter than air �Not as light as hydrogen [twice as heavy per volume] but does not burn [Hindenberg] �Used in atmosphere of deep sea divers �Less likely to cause the “bends” �Gives the diver “Mickey Mouse” voice because it has such a low density compared to air.

Argon �The most common Noble Gas. �Used to fill normal incandescent light bulbs to stop them imploding, to reduce evaporation of the filament and because it is unreactive �Used as inert atmosphere for welding

Trends in Noble gases �Don’t react in general because they have a full outer shell. �Down the Group – molecules get larger �Makes it easier to form temporary dipoles due to larger electron clouds �So van der Waal’s forces are greater �More energy required to break these. �So MP and BP get higher �Same trend in halogens

Octet Rule l. A chemical rule of thumb l. Octet Rule says atoms with 8 electrons in their outer shell are stable l. Atoms tend to combine in such a way that they each have eight electrons in their valence shells, giving them the same electron configuration as the nearest Noble gas

�The rule applies to the main-group elements, especially carbon, nitrogen, oxygen, the halogens, and also to metals such as sodium or magnesium. �In simple terms, molecules or ions tend to be most stable when the outermost electron shells of their constituent atoms contain 8 electrons.

Limitations Doesn’t allow for �H, He or Li �Stable with 2 e- in their outer shells Duet Rule �Transition elements - 18 electron rule �BF 3 which only has 6 e- in its outer shell

How can atoms get a full outer shell? 1. Give away electrons 2. Take in electrons 3. Share electrons 4. Don’t bother getting a full outer shell at all – (Rare but does happen [BF 3])

Ionic Bonding
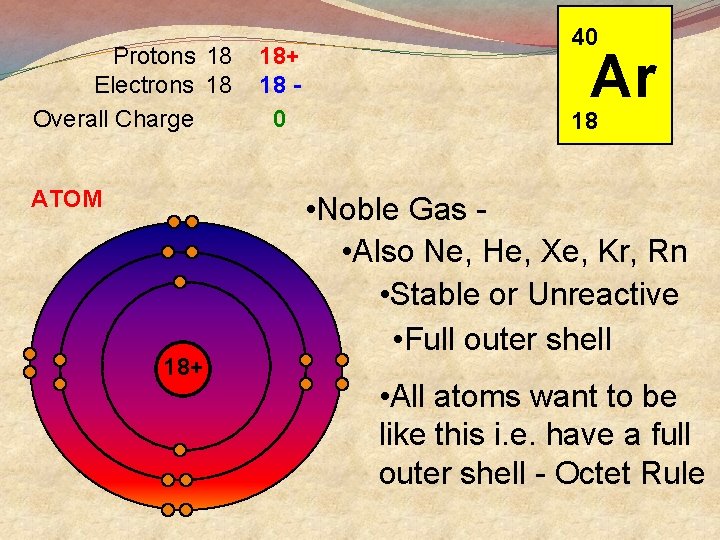
Protons 18 Electrons 18 Overall Charge ATOM 18+ 18 0 40 Ar 18 • Noble Gas • Also Ne, He, Xe, Kr, Rn • Stable or Unreactive • Full outer shell • All atoms want to be like this i. e. have a full outer shell - Octet Rule
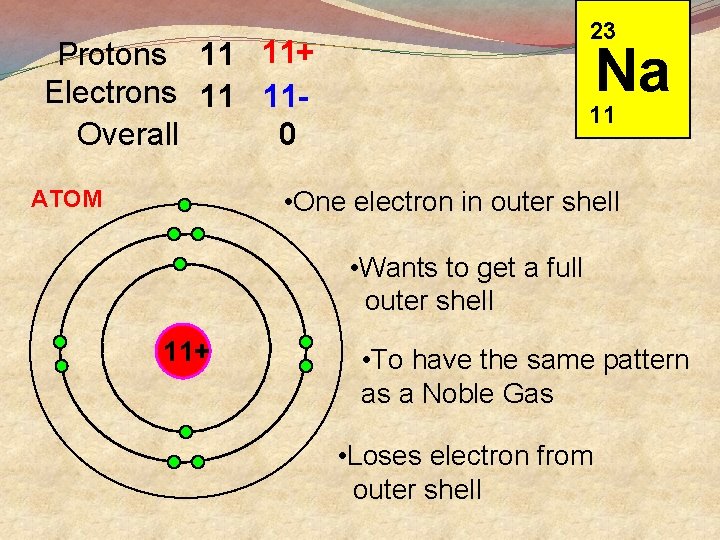
23 Protons 11 11+ Electrons 11 11 Overall 0 Na 11 • One electron in outer shell ATOM • Wants to get a full outer shell 11+ • To have the same pattern as a Noble Gas • Loses electron from outer shell
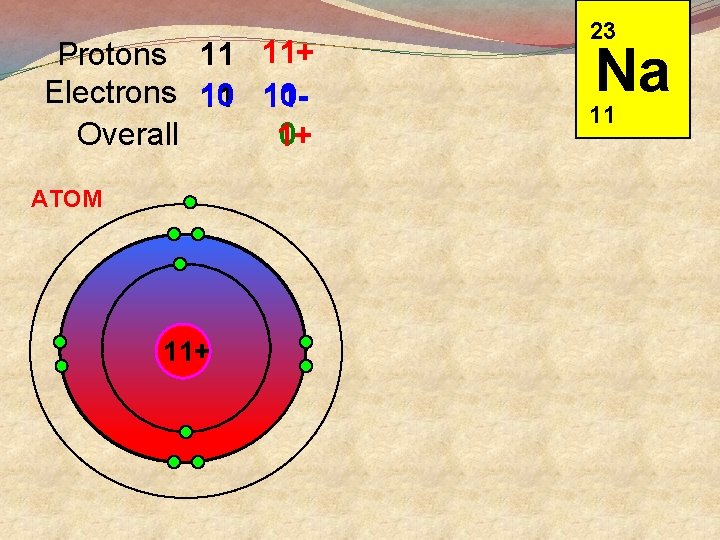
Protons 11 11+ Electrons 10 1011 11 Overall 0 1+ ATOM 11+ 23 Na 11
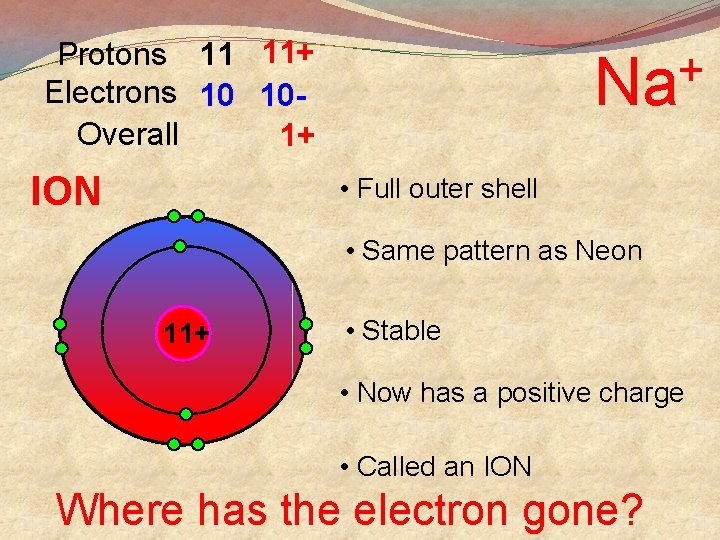
Protons 11 11+ Electrons 10 10 Overall 1+ ION + Na • Full outer shell • Same pattern as Neon 11+ • Stable • Now has a positive charge • Called an ION Where has the electron gone?
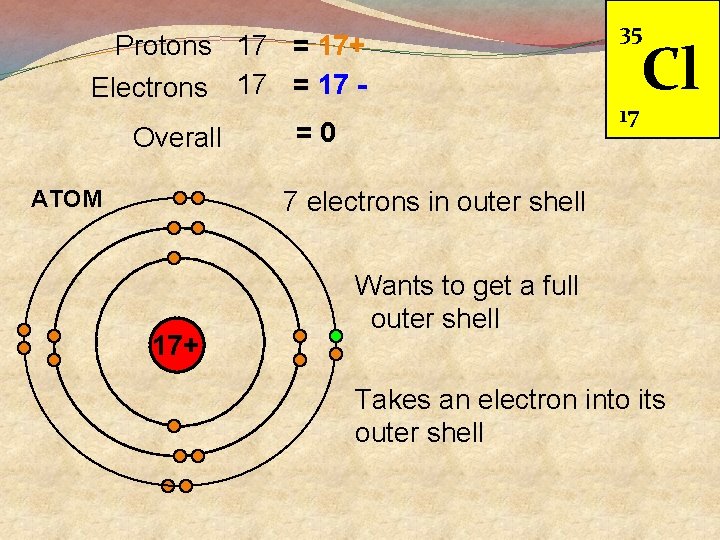
Protons 17 = 17+ Electrons 17 = 17 Overall ATOM =0 35 17 Cl 7 electrons in outer shell 17+ Wants to get a full outer shell Takes an electron into its outer shell

Protons 17 Electrons Overall 18 ION Cl 17+ 18 1 - Full outer shell Same pattern as Argon 17+ Stable Now has a - charge Called an ION
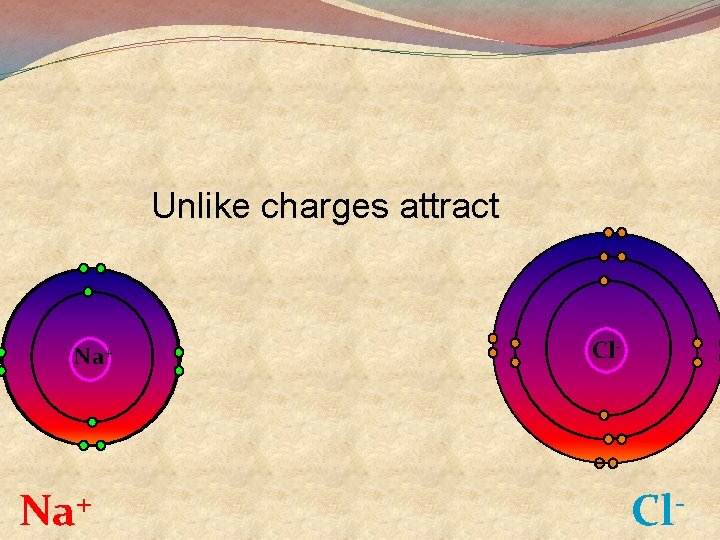
Unlike charges attract Na+ + Na Cl- Cl

Summary �Sodium atoms lose an electron to become sodium ions �Sodium ions have a +ve charge �Chlorine atoms gain an electron to become Chloride ions �Chloride ions have a − ve charge �Opposite charges attract so the Na+ and Cl - come together and stick to each other �This is called an ionic bond
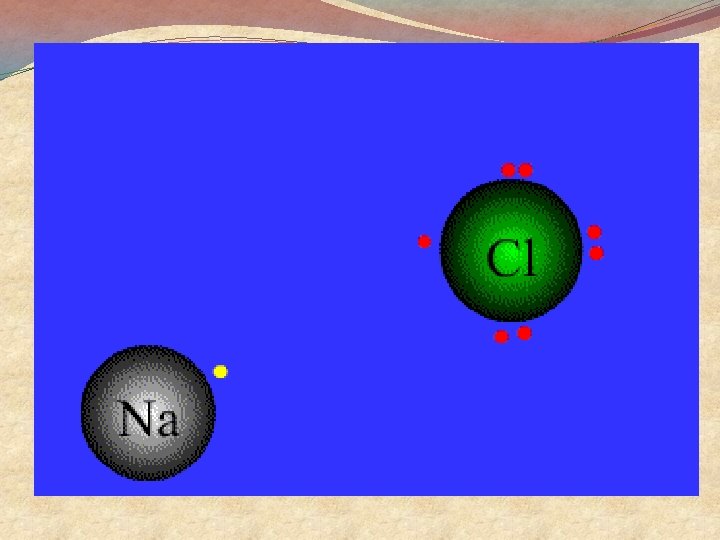

General Points �Ionic Bonding involves a transfer of electrons �It happens when the difference in electronegativity between atoms > 1. 7 �Metals lose electrons to form positive ions �They gain one plus charge for each electron lost �Non-metals gain electrons to form negative ions �They gain one minus charge for each electron gained �Total electron loss must equal electron gain
![Keeping Track of Electrons �Atoms in the same group [column] �Have the same outer Keeping Track of Electrons �Atoms in the same group [column] �Have the same outer](http://slidetodoc.com/presentation_image_h2/8bfc433e896cf63e082aa75085af4223/image-21.jpg)
Keeping Track of Electrons �Atoms in the same group [column] �Have the same outer electron configuration. �Have the same valence electrons. �Easily found by looking up the group number on the periodic table. �Group 2 A - Be, Mg, Ca, etc. � 2 valence electrons

General Points + M Group I lose 1 e- to become Group II lose 2 e- to become M 2+ Group III lose 3 e- to become M 3+ 3 Group V gain 3 e- to become X Group VI gain 2 e- to become X 2 Group VII gain 1 e- to become X There are exceptions
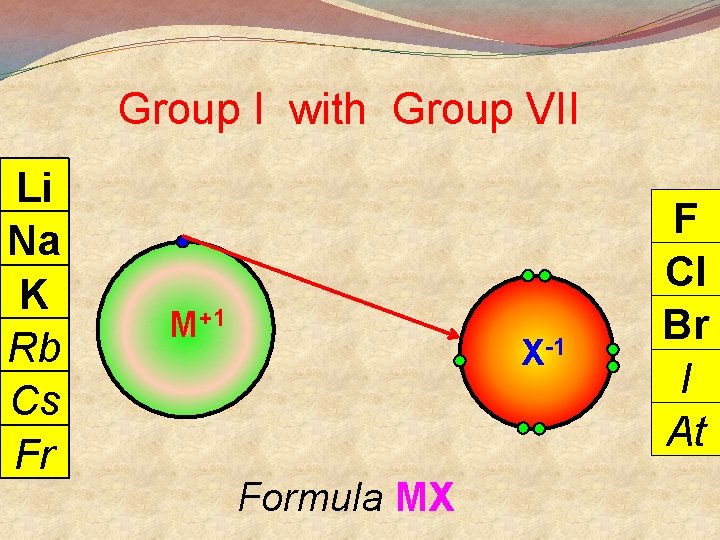
Group I with Group VII Li Na K Rb Cs Fr M+1 X-1 Formula MX F Cl Br I At
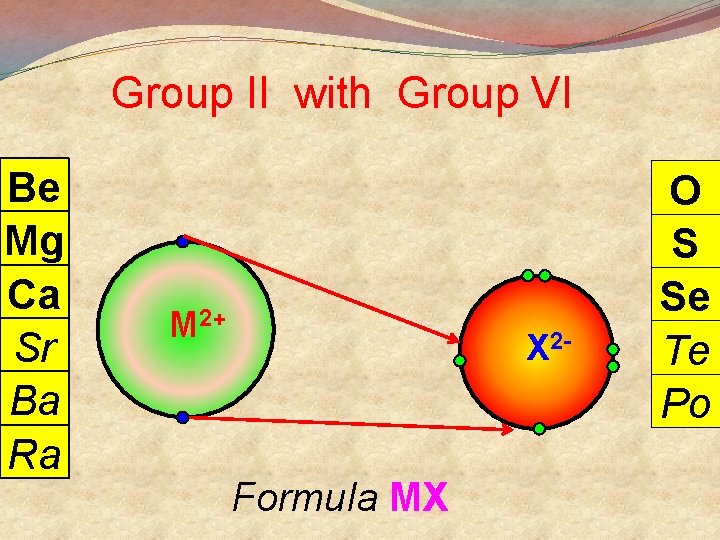
Group II with Group VI Be Mg Ca Sr Ba Ra M 2+ X 2 - Formula MX O S Se Te Po
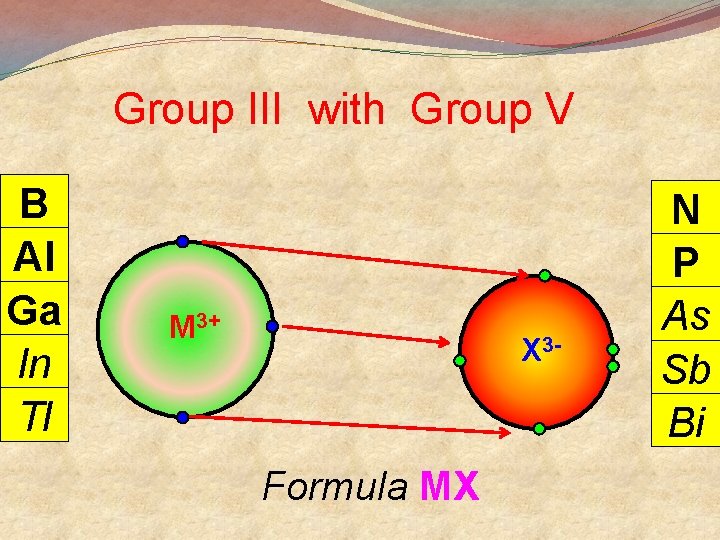
Group III with Group V B Al Ga In Tl M 3+ X 3 - Formula MX N P As Sb Bi
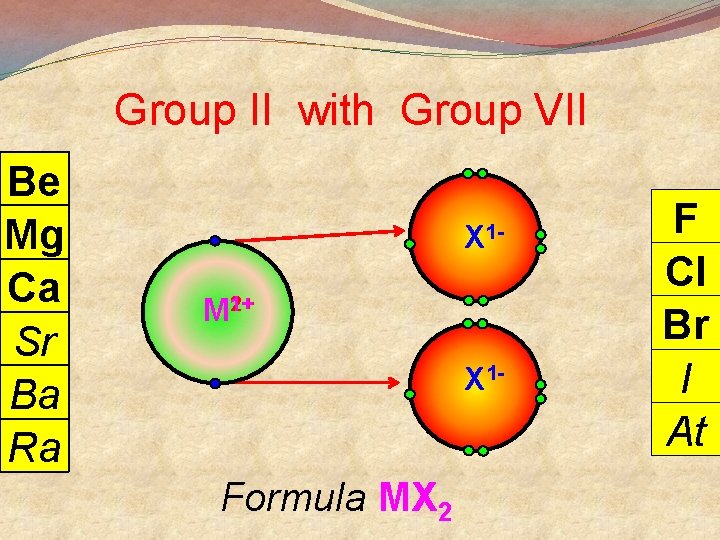
Group II with Group VII Be Mg Ca Sr Ba Ra X 12+ M 1+ X 1 - Formula MX 2 F Cl Br I At
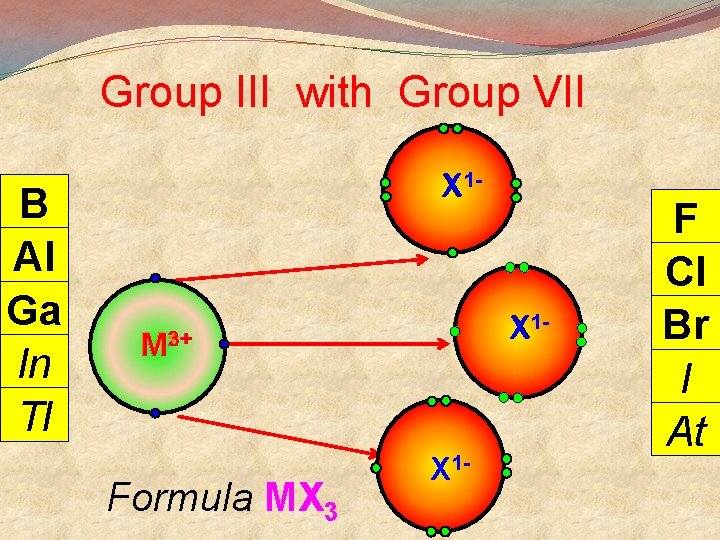
Group III with Group VII B Al Ga In Tl X 1 - 3+ M+2+ Formula MX 3 X 1 - F Cl Br I At
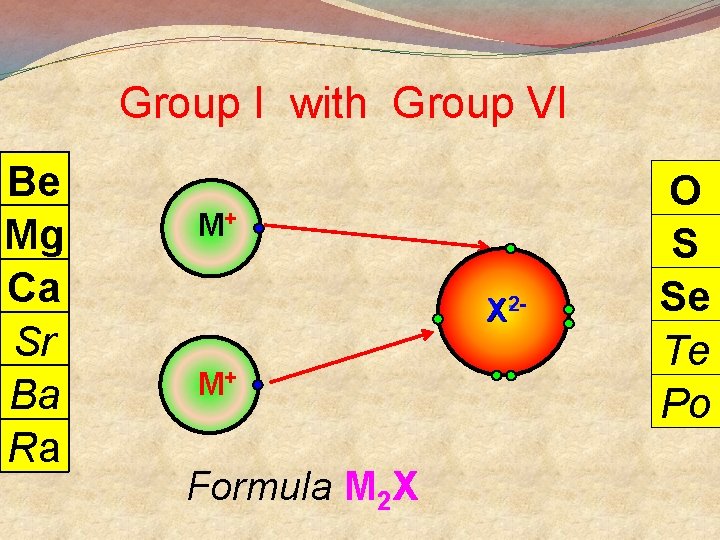
Group I with Group VI Be Mg Ca Sr Ba Ra M+ X 2 - M+ Formula M 2 X O S Se Te Po
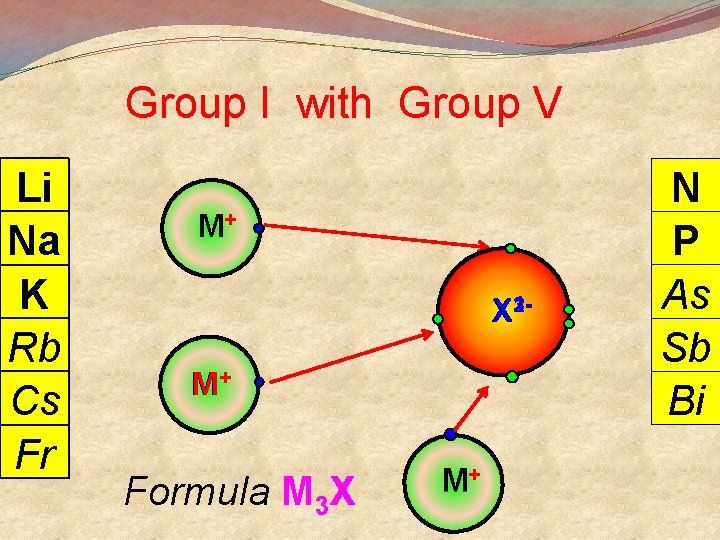
Group I with Group V Li Na K Rb Cs Fr M+ 23 X 1 - M+ Formula M 3 X M+ N P As Sb Bi
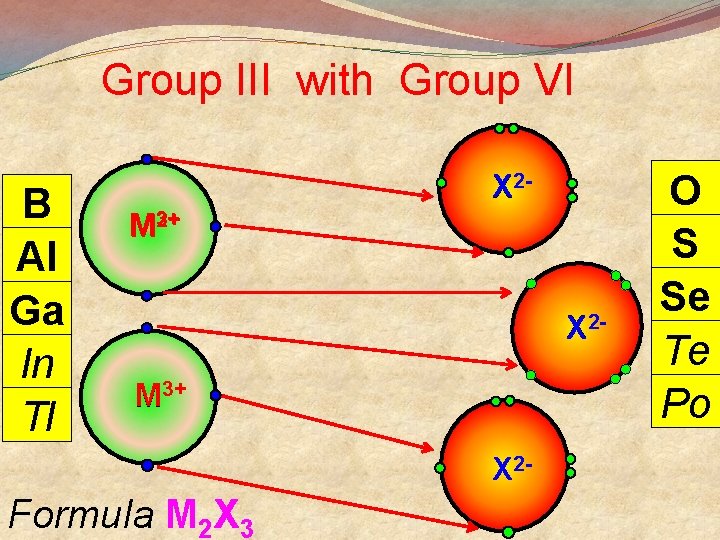
Group III with Group VI B Al Ga In Tl X 22+ M 3+ X 2 - Formula M 2 X 3 O S Se Te Po
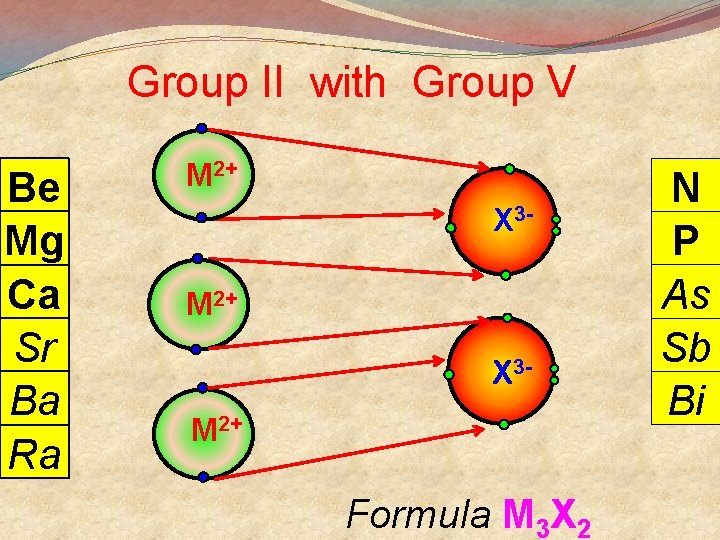
Group II with Group V Be Mg Ca Sr Ba Ra M 2+ X 3 M 2+ Formula M 3 X 2 N P As Sb Bi

Covalent Bonding Peter Jackson 32

General Rules �Involves a sharing of electrons �Electrons are shared in pairs �In each shared pair one electron comes from each atom �This type of bond occurs between elements of Groups IV, V, VI and VII �If there is a choice between ionic and covalent - covalent is preferred �There are exceptions 33
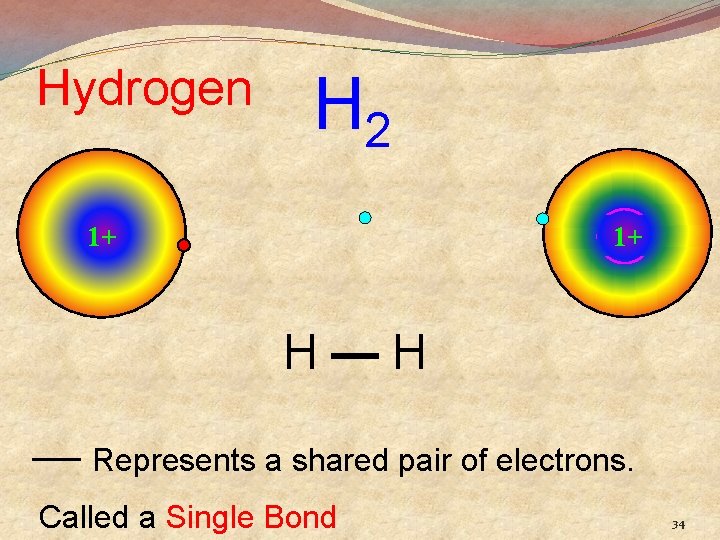
Hydrogen H 2 1+ 1+ H—H — Represents a shared pair of electrons. Called a Single Bond 34
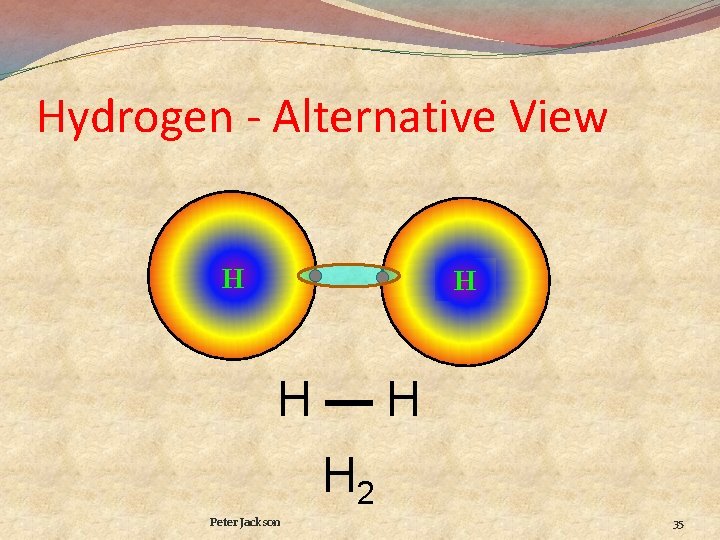
Hydrogen - Alternative View H H H—H H 2 Peter Jackson 35
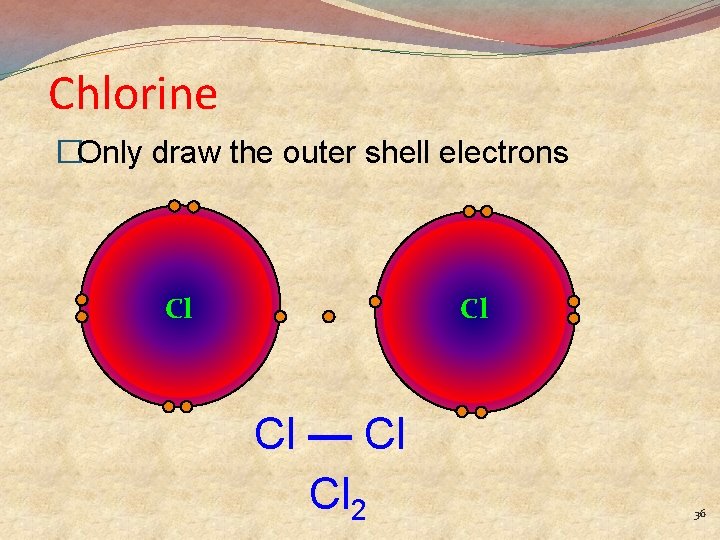
Chlorine �Only draw the outer shell electrons Cl Cl Cl — Cl Cl 2 36
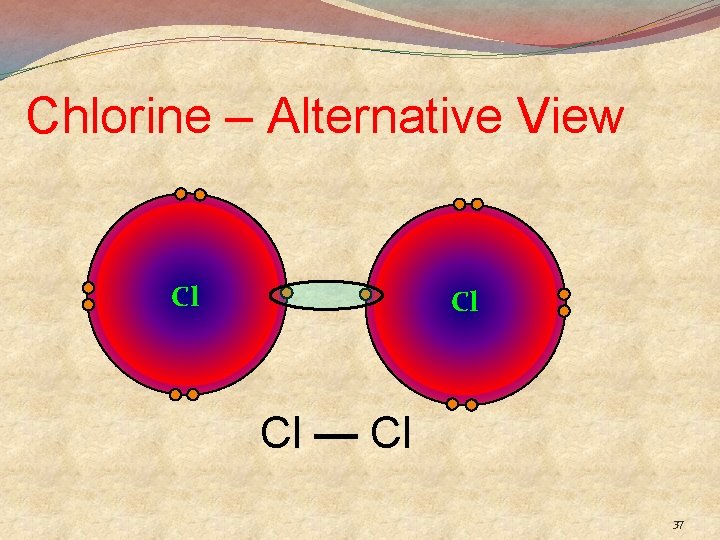
Chlorine – Alternative View Cl Cl Cl — Cl 37
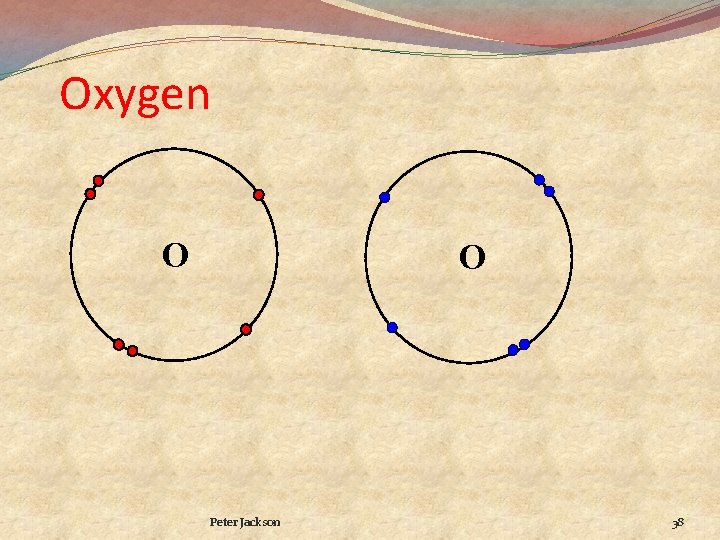
Oxygen O O Peter Jackson 38
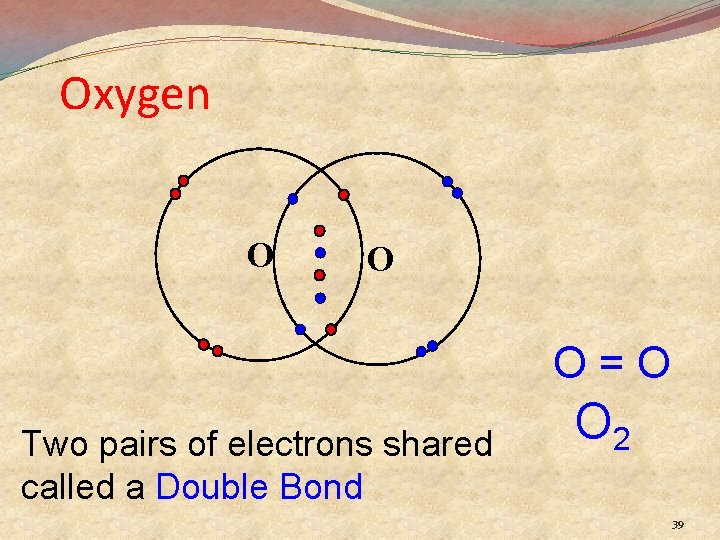
Oxygen O O O=O Two pairs of electrons shared called a Double Bond O 2 39
Oxygen – Alternative View O O O=O O 2 40
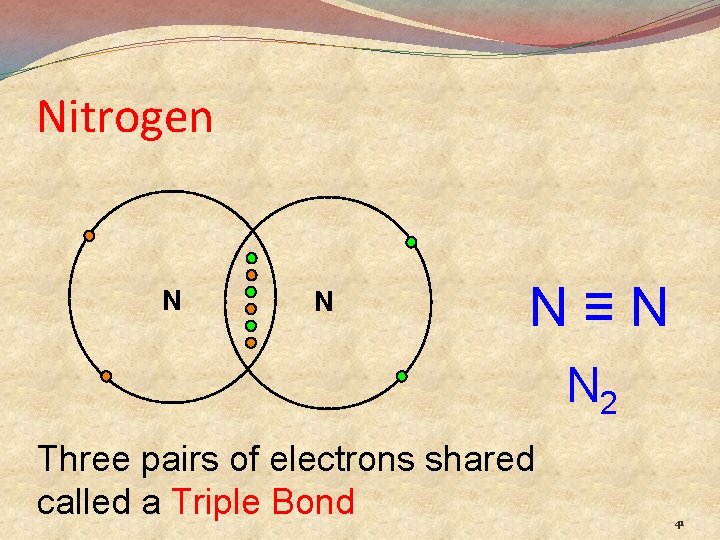
Nitrogen N N N≡N N 2 Three pairs of electrons shared called a Triple Bond 41
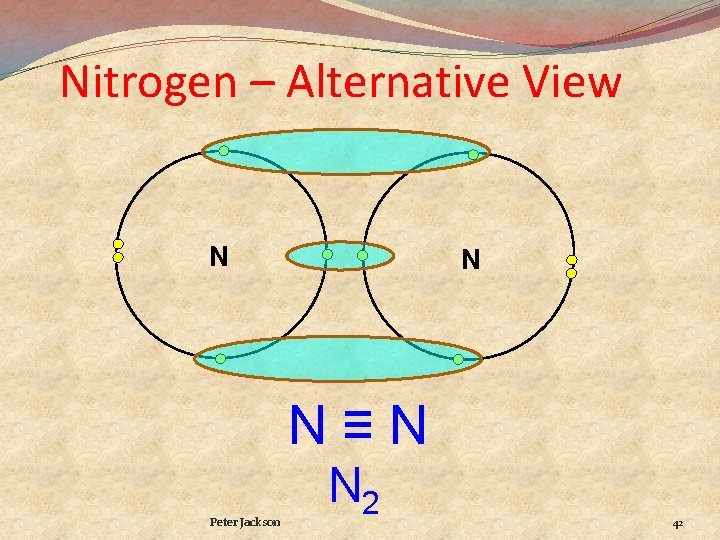
Nitrogen – Alternative View N N N≡N Peter Jackson N 2 42
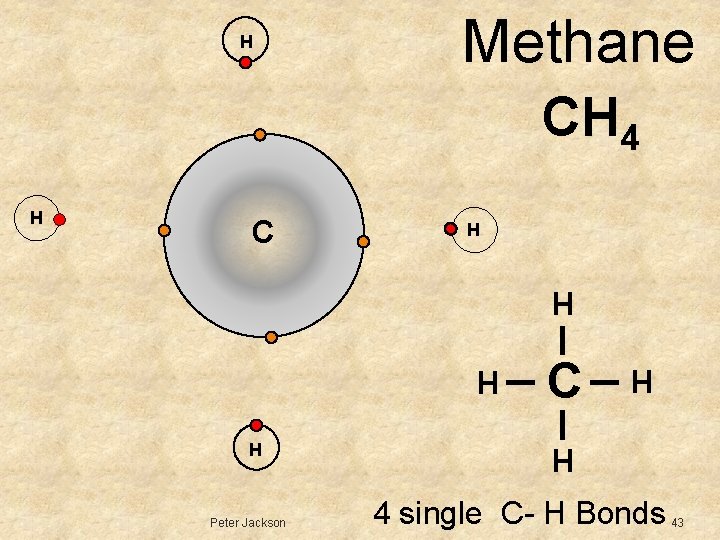
H Methane CH 4 C H H H Peter Jackson H H C H H 4 single C- H Bonds 43
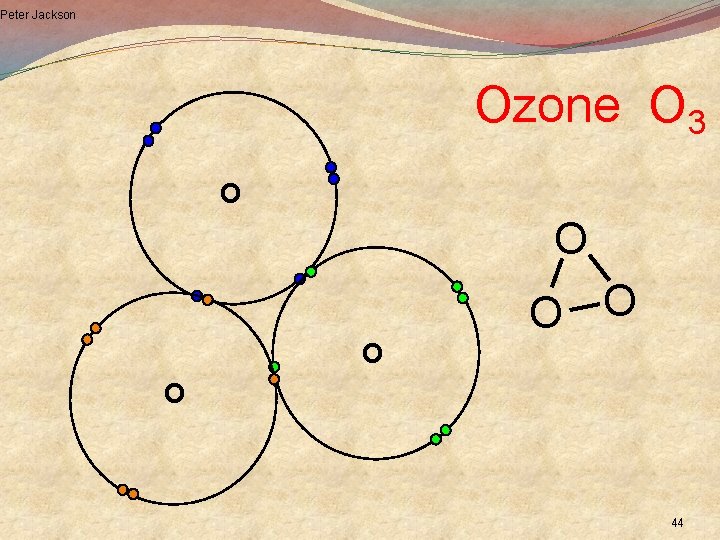
Peter Jackson Ozone O 3 O O O 44

Bonding

Electronegativity SCC Science Dept

�Is the relative attraction that an atom in a molecule has for the shared pair of electrons in a single covalent bond between two atoms of the same element �In a bond between two identical atoms the pair of electrons are shared equally �Chemists have found that in many bonds the pair of electrons are attracted to one of the atoms more than to the other. SCC Science Dept

Hydrogen and Chlorine �Electrons attracted to chlorine more than to hydrogen [bigger, but more +ve nucleus] �Electrons spend more time near the chlorine than near the hydrogen �This gives the chlorine a slightly negative charge δ- delta minus �This gives the hydrogen a slightly positive charge δ+ delta plus SCC Science Dept
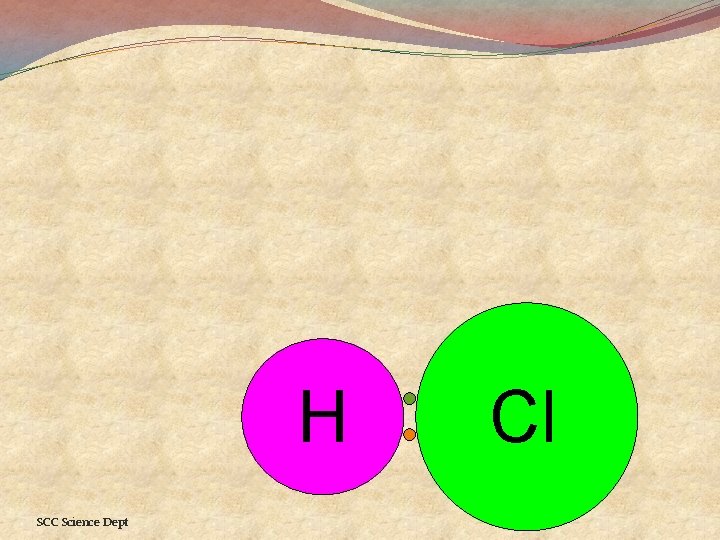
H SCC Science Dept Cl

Linus Pauling measured the electronegativity of each element and put them in a table Noble gases are not in the table because they do not form bonds + SCC Science Dept H Cl –
![Differences and Bond Type � Difference 0 to 0. 45 [Pure] Covalent Bond � Differences and Bond Type � Difference 0 to 0. 45 [Pure] Covalent Bond �](http://slidetodoc.com/presentation_image_h2/8bfc433e896cf63e082aa75085af4223/image-51.jpg)
Differences and Bond Type � Difference 0 to 0. 45 [Pure] Covalent Bond � Difference > 0. 45 but < 1. 7 Polar Covalent Bond � Difference is = or > 1. 7 then Ionic Bond SCC Science Dept

Trends in Electronegativity Across a period �Goes up 1. Bigger nuclear charge 2. Smaller atomic radius [distance from nucleus] Increased effective nuclear charge SCC Science Dept

Down a Group �Gets less �Bigger nuclear charge �But! 1. Increased Atomic Radius - electron is much further from nucleus 2. Increased shielding by more inner electron shells Decreased effective nuclear charge SCC Science Dept

Intra and Intermolecular Forces �Intramolecular forces are the forces within a molecule �They are essentially �Ionic Bonds and Covalent Bonds. �They are powerful forces

Intermolecular Forces �Forces of attraction between molecules �They are generally weak �Three types 1. Dipole – Dipole [permanent] 2. Hydrogen Bonding [special type of permanent dipole – dipole] 3. Van der Waals’ Forces [transient or temporary dipole]

�What is dipole? �Charge caused by the separation of the centres of + and - charge within an atom or molecule �Dipole-Dipole [Polar Bonding] caused by difference in electronegativity. Dipole permanent e. g. δ+ C=Oδ�Hydrogen Bonding special dipole-dipole where H bonded to N, O or F. e. g. δ- O-H δ+ in water [1/10 th] �Van der Waals’ Forces occurs in atoms or molecules where the difference in electronegativity is 0 and there should be no polarity. They are caused by temporary dipoles produced by random movement of electrons. E. g. H-H or He [about 1/1000 of covalent]

�Why is NH 3 soluble in water but PH 3 is not? �NH 3 is polar, PH 3 is not. NH 3 forms hydrogen bonds with the water, PH 3 does not. �Metallic Bonds a lattice of +vely charged nuclei in a sea of electrons

Let us look at a Helium atom + 1. Centre of + and – charges are in the same place [centre] so there is an even distribution of charge
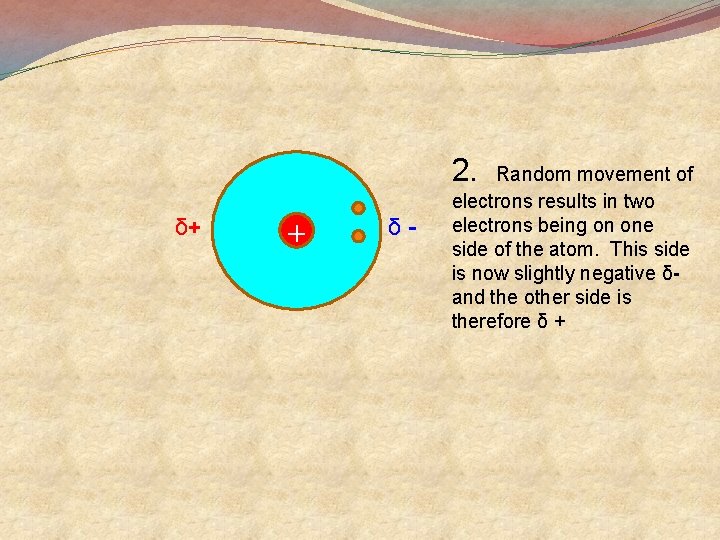
2. δ+ + δ- Random movement of electrons results in two electrons being on one side of the atom. This side is now slightly negative δand the other side is therefore δ +

3. Random movement of electrons results in two electrons being on another side of the atom. This side is now slightly negative δand the other side is therefore δ + 4. δ- It is unlikely that the two electrons will be exactly opposite each other so the atom spends most of it time with one part slightly negative δ-and the other side slightly positive δ + + δ+ This is called a Temporary Dipole

Let us now look at a Neon atom �The bigger the atom the more electrons it has so there is more scope for a bigger charge differential between the two sides. �This is due to the possibility of more electrons being at one side.
![+ 1. Centre of + and – charges are in the same place [centre] + 1. Centre of + and – charges are in the same place [centre]](http://slidetodoc.com/presentation_image_h2/8bfc433e896cf63e082aa75085af4223/image-62.jpg)
+ 1. Centre of + and – charges are in the same place [centre] so there is an even distribution of charge
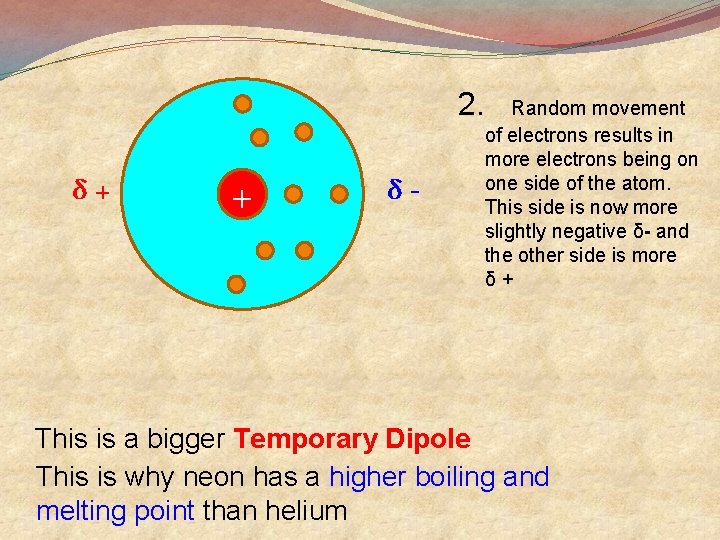
2. δ+ + δ- Random movement of electrons results in more electrons being on one side of the atom. This side is now more slightly negative δ- and the other side is more δ+ This is a bigger Temporary Dipole This is why neon has a higher boiling and melting point than helium

What is the effect of these Temporary Dipoles? These atoms haves NO temporary dipoles at the moment. They have a limited attraction to each other

This atom has developed a temporary dipole δ+ δ- This atom still has NO temporary dipole
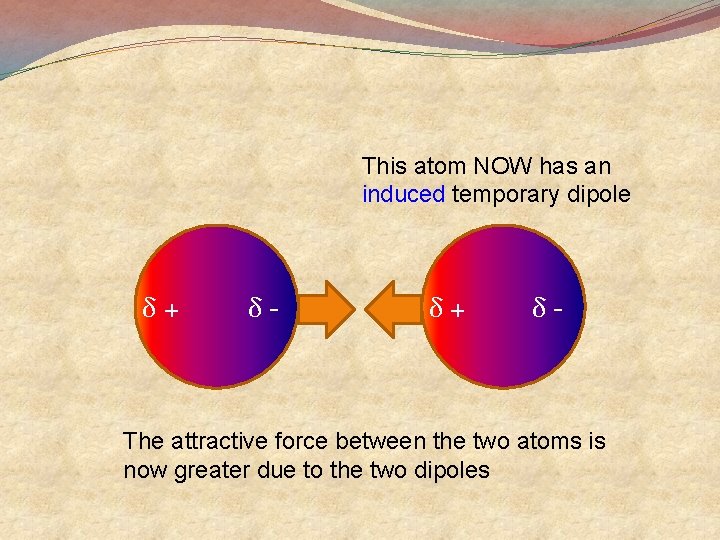
This atom NOW has an induced temporary dipole δ+ δ- The attractive force between the two atoms is now greater due to the two dipoles

• Since the force of attraction is greater it is more difficult to remove one of the atoms from the other. • The heat needed to give one enough energy to evaporate is greater • Thus the boiling point is greater than if there were no dipoles • Thus neon has a higher boiling point than helium. • Melting points are also greater for the same reason

• Neon has a higher boiling point than helium because the atoms are bigger and therefore can produce greater temporary dipoles. • The boiling point of O 2 is higher than H 2 for the same reason • Molecules with dipoles tend to be soluble in water – like dissolves like

Everday experience of Van der Walls’ Forces
![�Graphite is a form of carbon [allotrope] �Graphite is pencil lead �Made up of �Graphite is a form of carbon [allotrope] �Graphite is pencil lead �Made up of](http://slidetodoc.com/presentation_image_h2/8bfc433e896cf63e082aa75085af4223/image-70.jpg)
�Graphite is a form of carbon [allotrope] �Graphite is pencil lead �Made up of sheets of carbon atoms in hexagons �Carbons are joined to each other by covalent bonds �Sheets are held to each other by van der Walls’ forces.

�When you put the pencil on the paper the force of attraction between the paper and the graphite is greater than the attraction between the grapite sheets. �Thus when you move the pencil the bottom graphite sheet sticks to the paper �The pencils slides along and drops to the paper to repeat the process. �Pencils can write underwater �The american spent millions developing a pen that could write in space �The Russians were smart !!!!!!!! �They brought pencils
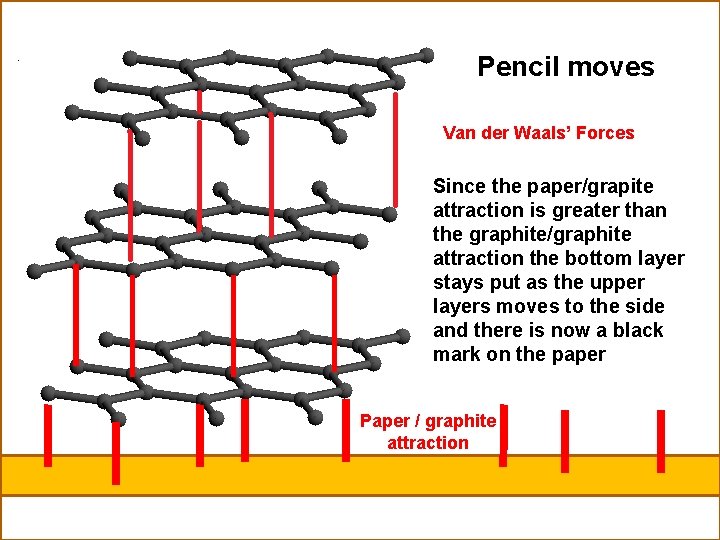
Pencil moves Van der Waals’ Forces Since the paper/grapite attraction is greater than the graphite/graphite attraction the bottom layer stays put as the upper layers moves to the side and there is now a black mark on the paper Paper / graphite attraction
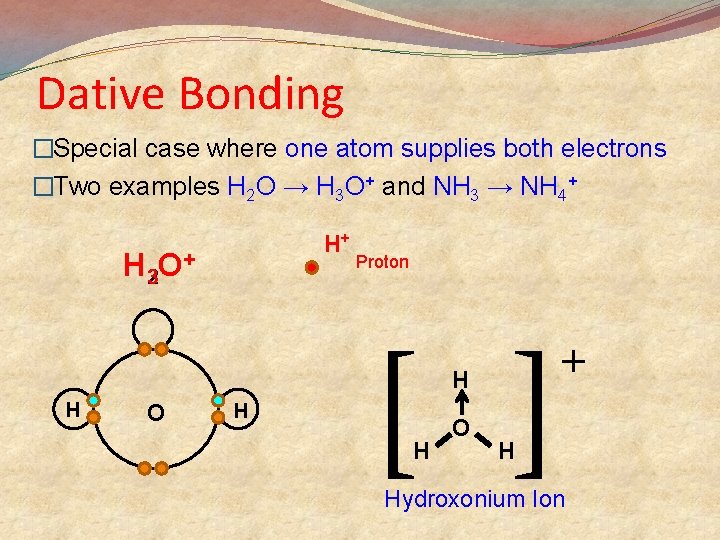
Dative Bonding �Special case where one atom supplies both electrons �Two examples H 2 O → H 3 O+ and NH 3 → NH 4+ H+ H 32 O+ Proton [ ] H H O + H Hydroxonium Ion
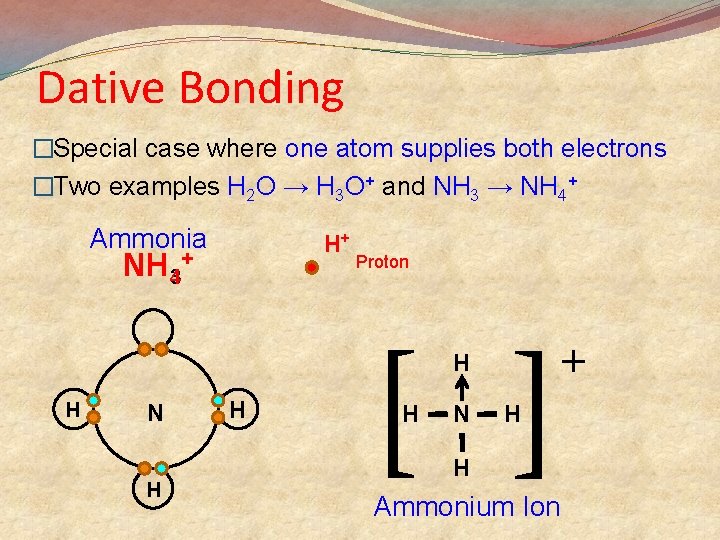
Dative Bonding �Special case where one atom supplies both electrons �Two examples H 2 O → H 3 O+ and NH 3 → NH 4+ Ammonia H+ NH 43+ Proton [ ] H H N H H H N + H H Ammonium Ion
- Slides: 74