Nitrogen cycling in the ocean Deborah Bronk Department

Nitrogen cycling in the ocean Deborah Bronk Department of Physical Sciences

bronk@vims. edu

Nitrogen Revolution mid-1990 s-present N • Nitrification • Denitrification/anammox N N • N 2 fixation • Anthropogenic N inputs
Why nitrogen is so cool! SEAWIFs chl a image

Outline: 1. Types & distribution 2. The Redfield Ratio 3. The nitrogen cycle 4. Liebig’s Law of the Minimum 5. New & regenerated production 6. Is the N cycle in steady state?

Types: Gaseous – N 2 gas Dissolved + Inorganic: ammonium – NH 4 nitrite – NO 2 nitrate – NO 3 Organic – DON Particulate – PN (cells & detritus)

Types: Gaseous – N 2 gas Dissolved + Inorganic: ammonium – NH 4 nitrite – NO 2 nitrate – NO 3 Organic – DON Particulate – PN (cells & detritus)
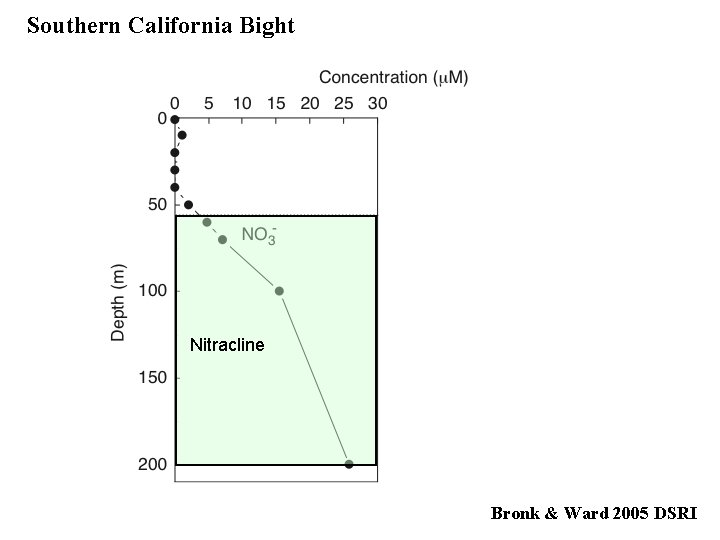
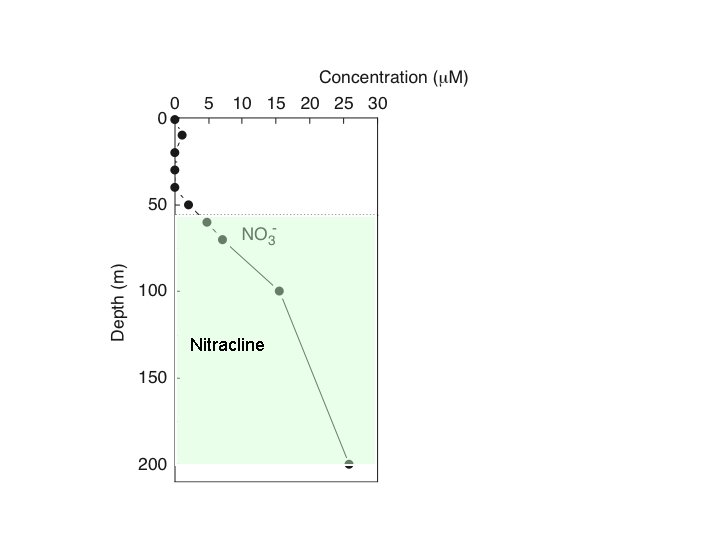
Southern California Bight Nitracline Bronk & Ward 2005 DSRI
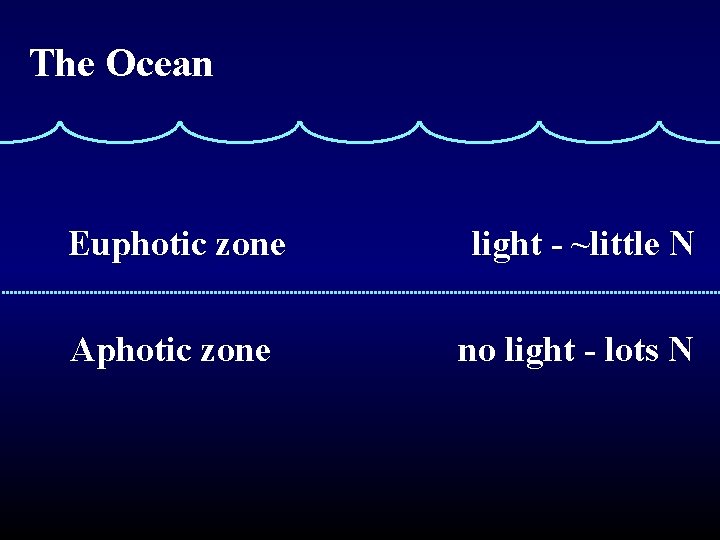
The Ocean Euphotic zone light - ~little N Aphotic zone no light - lots N
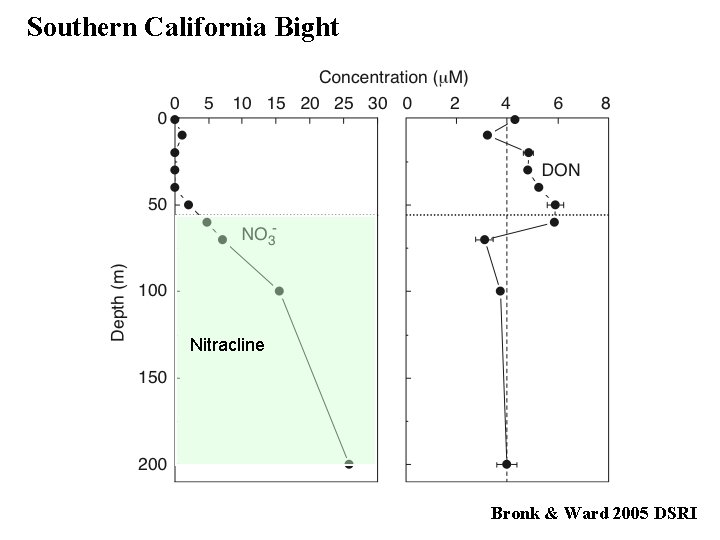
Southern California Bight Nitracline Bronk & Ward 2005 DSRI
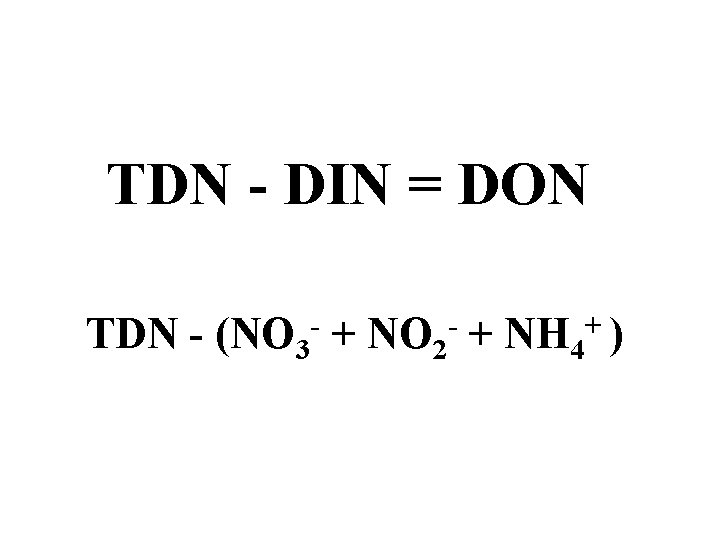
TDN - DIN = DON TDN - (NO 3 - + NO 2 - + NH 4+ )
Karl & Björkman 2002 DOM book
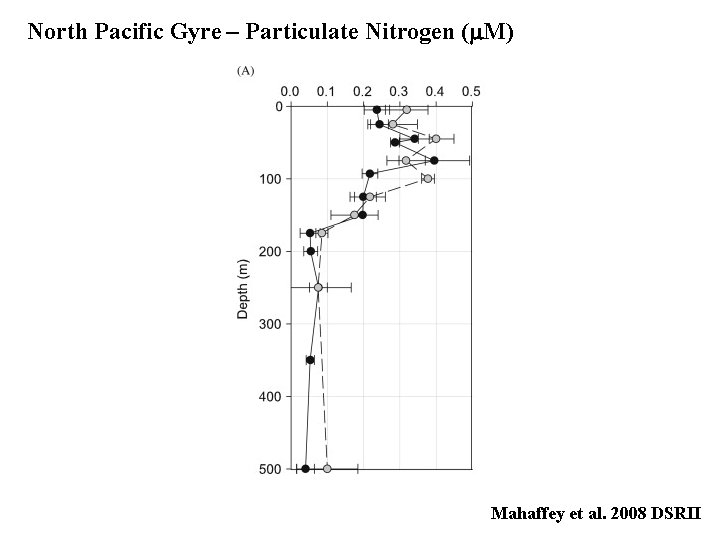
North Pacific Gyre – Particulate Nitrogen (m. M) Mahaffey et al. 2008 DSRII

Redfield 1934 James Johnstone Memorial Volume

Read the original papers!

The Redfield Ratio C: N: P = 106: 1 106 CO 2 + 122 H 2 O + 16 HNO 3 + H 3 PO 4 (CH 2 O)106(NH 3)16(H 3 PO 4) detritus vs. phyto vs. bacteria?
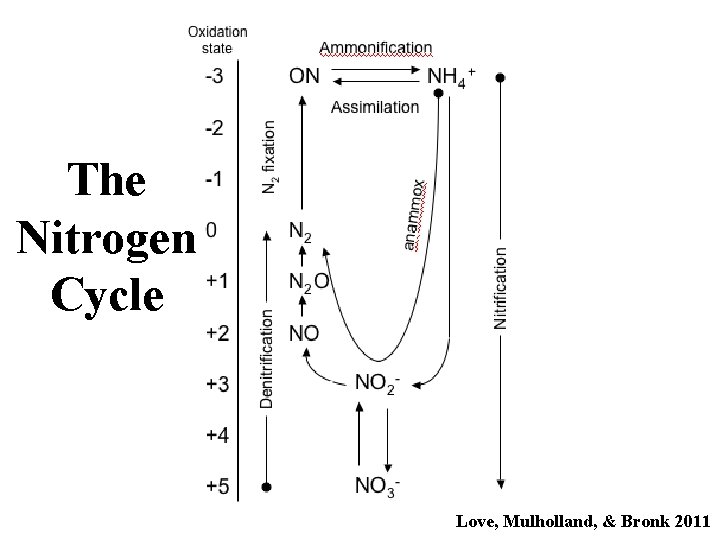
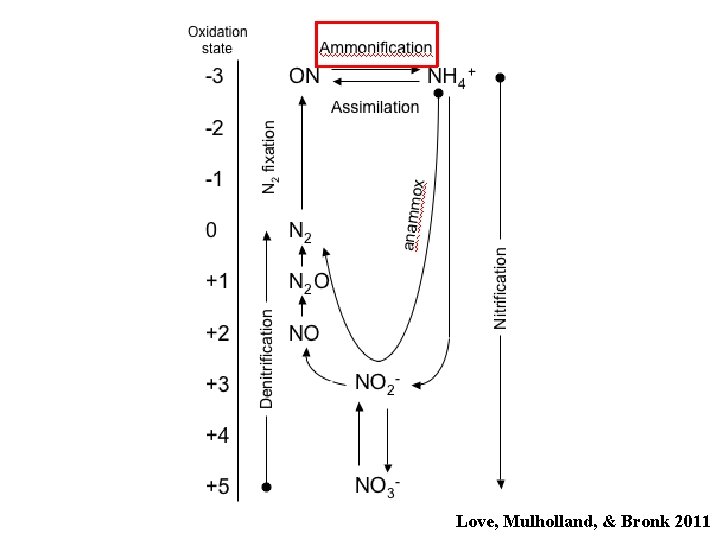
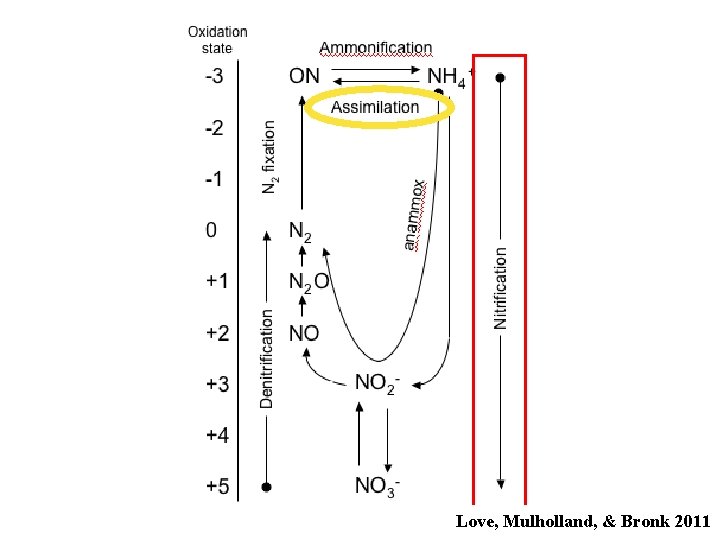
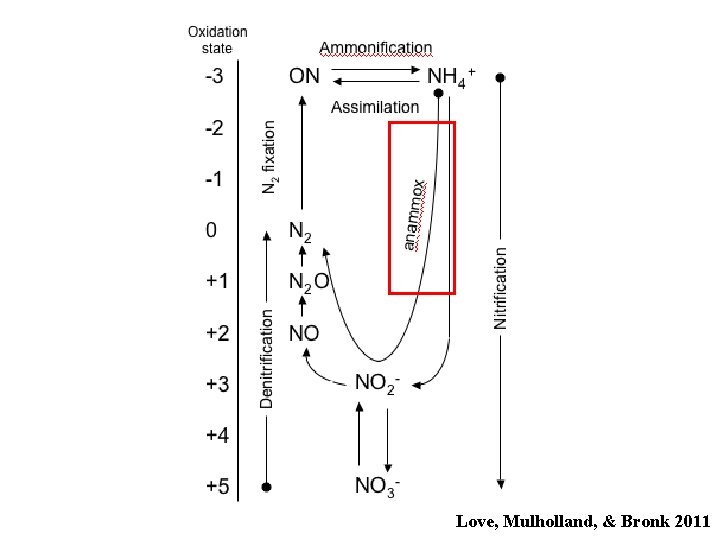
The Nitrogen Cycle Love, Mulholland, & Bronk 2011
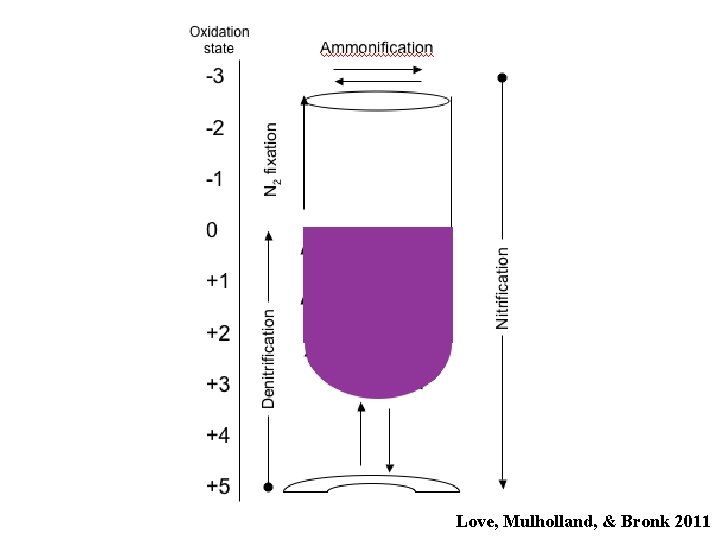
Love, Mulholland, & Bronk 2011
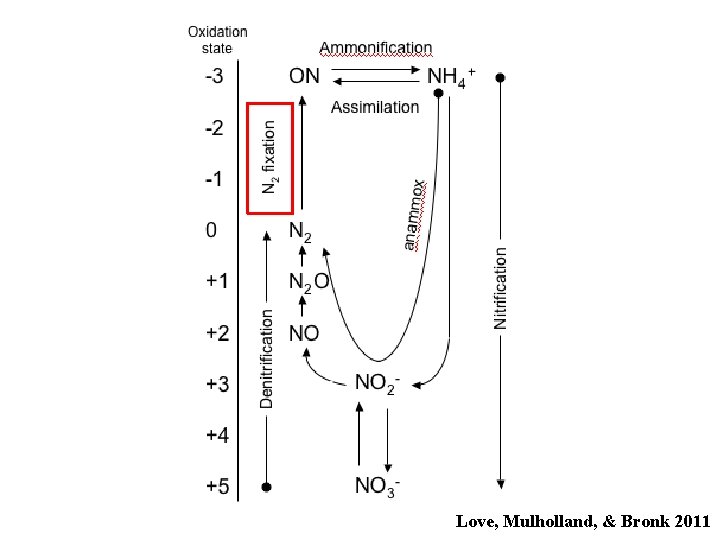
Love, Mulholland, & Bronk 2011

Nitrogen fixation N 2 + 8 H+ + 8 e- + 16 ATP 2 NH 3 + H 2 + 16 ADP + 16 Pi Nitrogenase • “Fixing” broken N 2 • Energetically very expensive – N 2 has triple bond • Nitrogenase is irreversibly inactivated by oxygen • Most N 2 fixers form heterocysts • Requires P, Fe, and trace metals (Mo, Co, V)

N 2 • • N 2 fixation Trichodesmium Two morphological forms Colonial – 100 s cells/trichome and 100 s trichomes/colony Traditionally considered the dominant N fixer in the ocean Found in tropical and subtropical waters (NOT!)
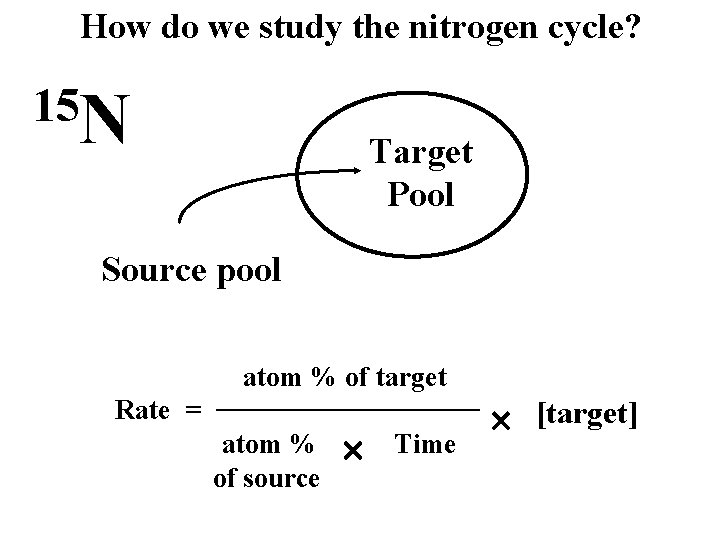
How do we study the nitrogen cycle? 15 N Target Pool Source pool atom % of target Rate = atom % of source Time [target]
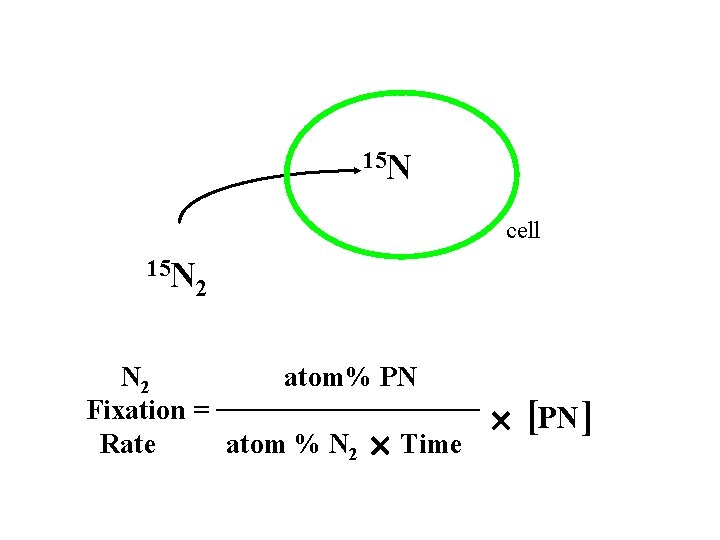
15 N cell 15 N 2 atom% PN Fixation = Rate atom % N 2 Time [PN ]

Trichodesmium mystery Non-heterocystous Photosynthesis CO 2 N 2 fixation N 2 NH 4+ nitrogenase Temporal or spatial segregation?
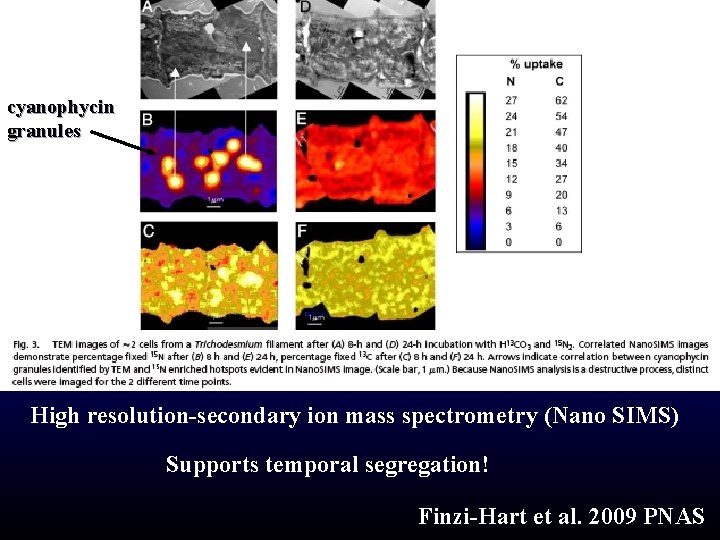
cyanophycin granules High resolution-secondary ion mass spectrometry (Nano SIMS) Supports temporal segregation! Finzi-Hart et al. 2009 PNAS

N 2 fixation N 2 Zehr et al. 2001 Nature Unicellular cyanobacteria that expressed nitrogenase at HOT Montoya et al. 2004 Nature Rates of N fixation by the single cell forms can equal or exceed rates by Trichodemsium
Love, Mulholland, & Bronk 2011
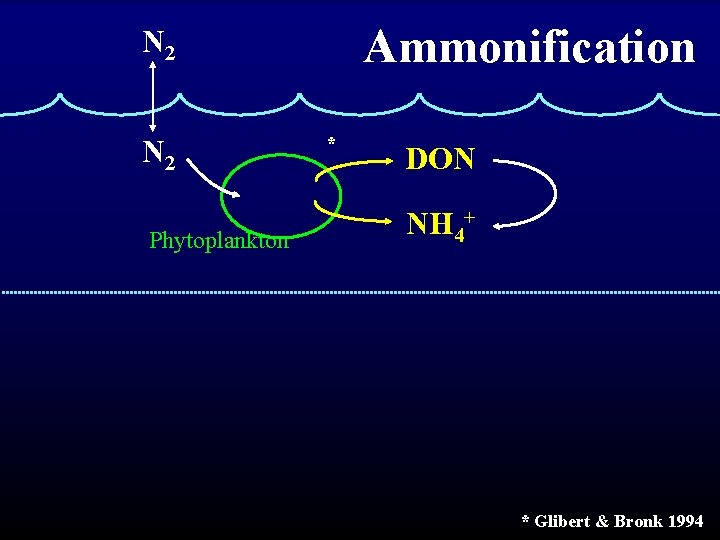
Ammonification N 2 Phytoplankton * DON NH 4+ * Glibert & Bronk 1994

Ammonification the conversion of DON or PON to NH 4+ Two types: 1. Bacterial a. Traditional view of bacterial decomposition b. Was considered primary source of NH 4+ (Now primary source if believed to be grazers) 2. Photochemical (abiotic!) a. More recently recognized source b. Importance of process is still debated
Love, Mulholland, & Bronk 2011

Nitrification NH 4+ ---------> NO 2 - ---------> NO 3 Ammonium oxidizers: Nitrite oxidizers: very slow growing faster growing sensitive to light more sensitive to light • Nitrifiers are chemolithoautotrophs. • Maximum rates occur near the base of the euphotic zone.
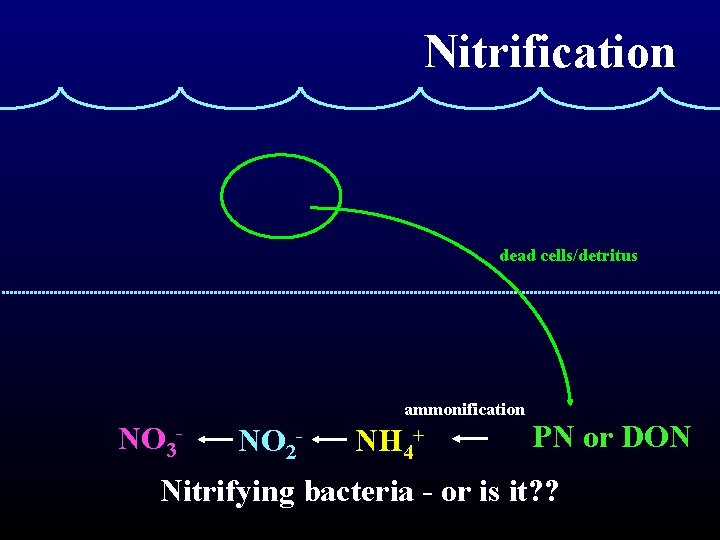
Nitrification dead cells/detritus ammonification NO 3 - NO 2 - NH 4+ PN or DON Nitrifying bacteria - or is it? ?
Nitracline
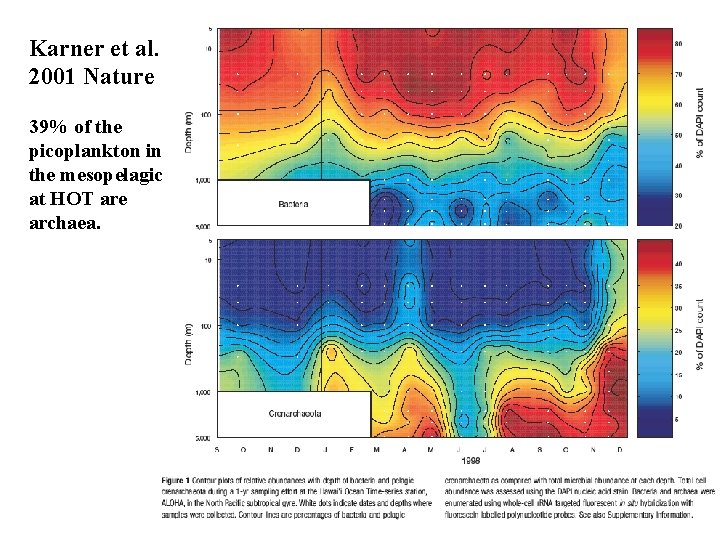
Karner et al. 2001 Nature 39% of the picoplankton in the mesopelagic at HOT are archaea.

Könneke et al. 2005 Nature Isolated a marine crenarchaeota that can grow by aerobically oxidizing NH 4+ to NO 2 -. Ingalls et al. 2006 PNAS An isotopic mass balance of radiocarbon signatures of archaeal membrane lipids indicates that 83% of their carbon is obtained autotrophically at depth.
Love, Mulholland, & Bronk 2011

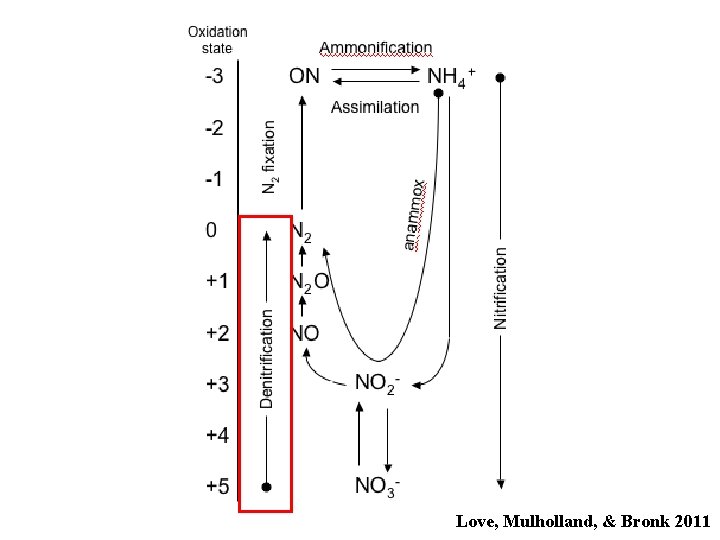
Lost to system Denitrification NO 3 - NO 2 - nitrate reductase NO nitrite reductase N 2 O nitric ox. reductase nitrous oxide • N is used as an electron acceptor, not as a N source • Lots of organisms can reduce NO 3 • Fewer can reduce NO 2 • All the enzymes are induced by anoxia • NO is very labile and does not accumulate
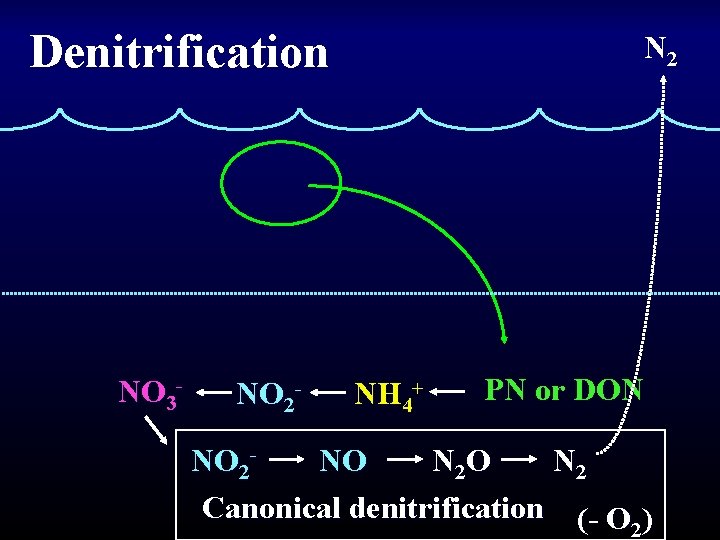
Denitrification NO 3 - NO 2 - N 2 NH 4+ PN or DON NO 2 N 2 NO N 2 O Canonical denitrification (- O ) 2
Love, Mulholland, & Bronk 2011

ANAMMOX ANaerobic AMMonium OXidation NO 2 - + NH 4+ N 2 • First described in a wastewater treatment plant in the Netherlands in 1995. • Oxygen inhibition is reversible. • Thandrup & Dalsgaard (2002) published the first marine data (sediment study)

Look to other disciplines
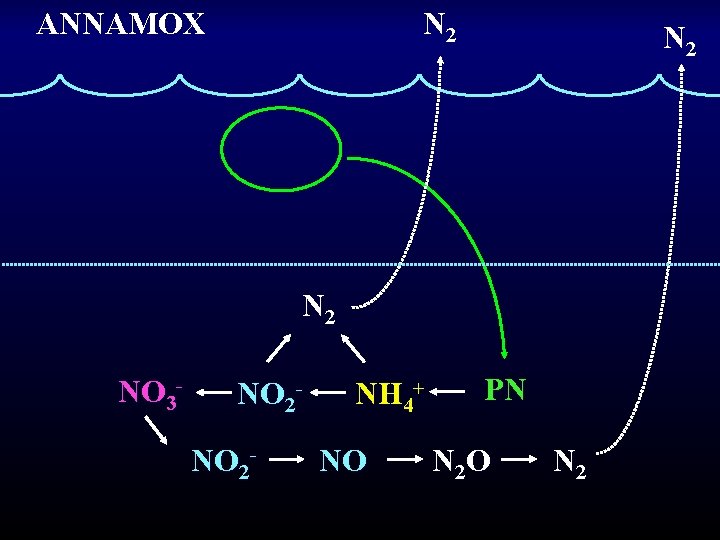
ANNAMOX N 2 N 2 NO 3 - NO 2 - NH 4+ NO PN N 2 O N 2
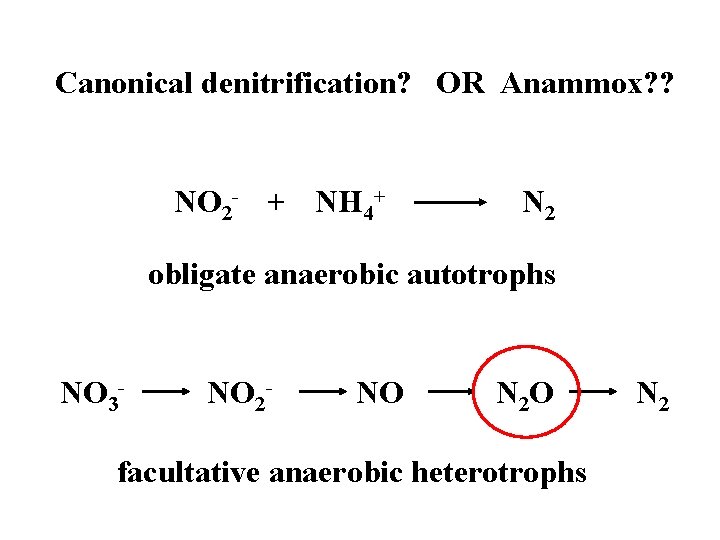
Canonical denitrification? OR Anammox? ? NO 2 - + NH 4+ N 2 obligate anaerobic autotrophs NO 3 - NO 2 - NO N 2 O facultative anaerobic heterotrophs N 2
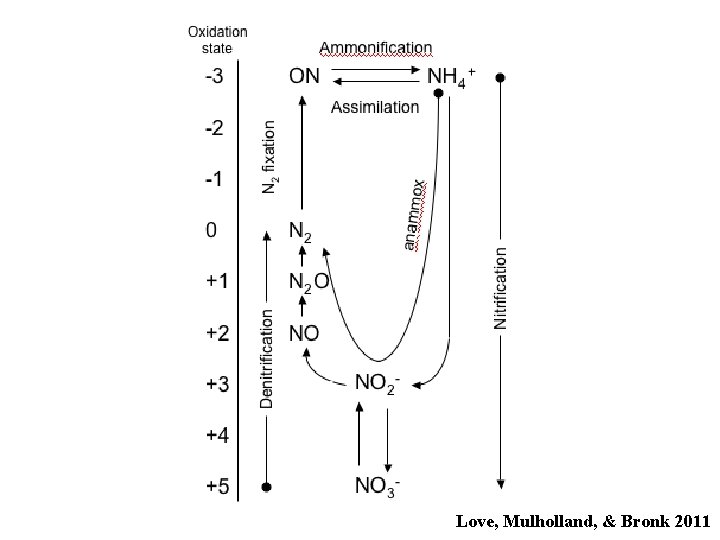
Love, Mulholland, & Bronk 2011

What’s the best way to make a living in a given place? Reaction Energy yield (kcal) Aerobic respiration 686 Denitrification 545 (-O 2) Nitrification N 2 fixation NH 4+ oxidation NO 2 - oxidation 66 17 -147
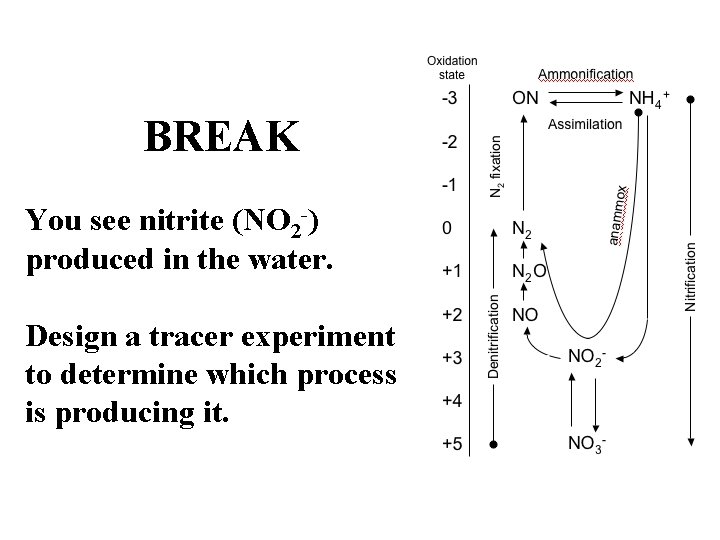
BREAK You see nitrite (NO 2 -) produced in the water. Design a tracer experiment to determine which process is producing it.
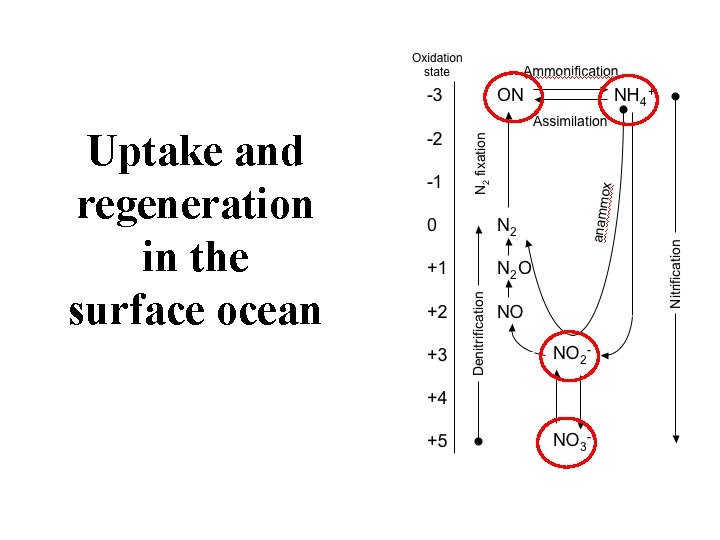
Uptake and regeneration in the surface ocean

Liebig’s Law of the Minimum (1840) the resource in smallest supply relative to what the organism needs is the limiting factor. Growth versus biomass
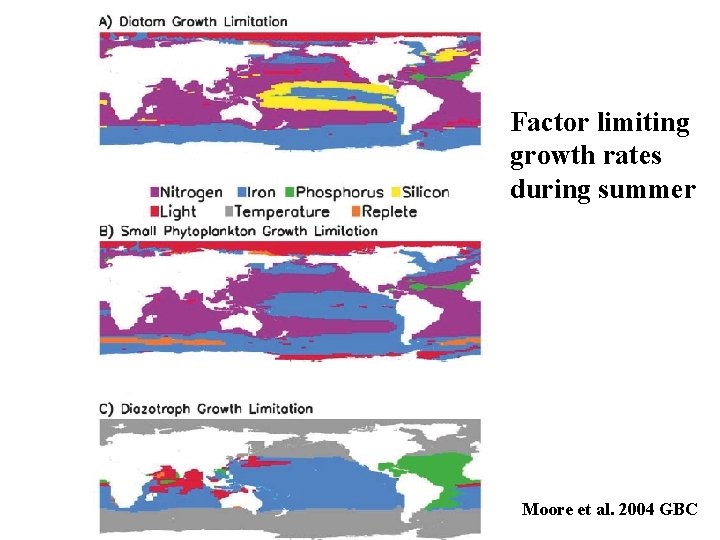
Factor limiting growth rates during summer Moore et al. 2004 GBC
SEAWIFs chl a image

Production can be defined as new or regenerated based on the source of the nitrogen that fueled it. Dugdale & Goering 1967 L&O
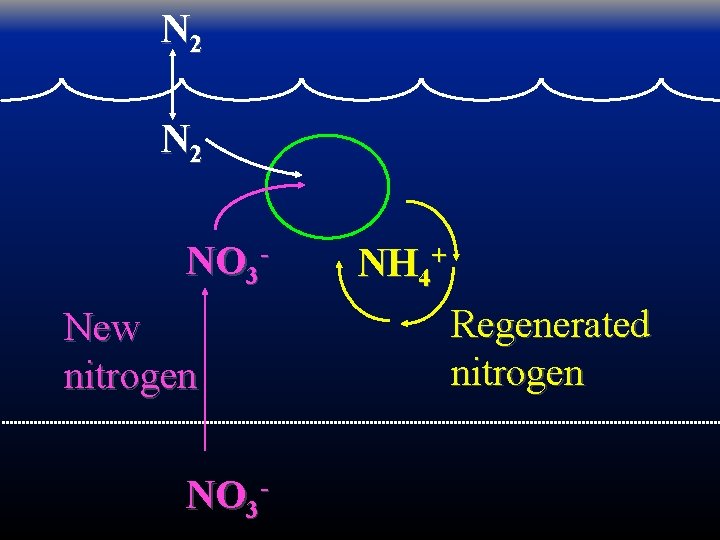
N 2 NO 3 New nitrogen NO 3 - NH 4+ Regenerated nitrogen

New production over appropriate spatial and temporal scales equals export flux. Eppley & Peterson 1979 Nature
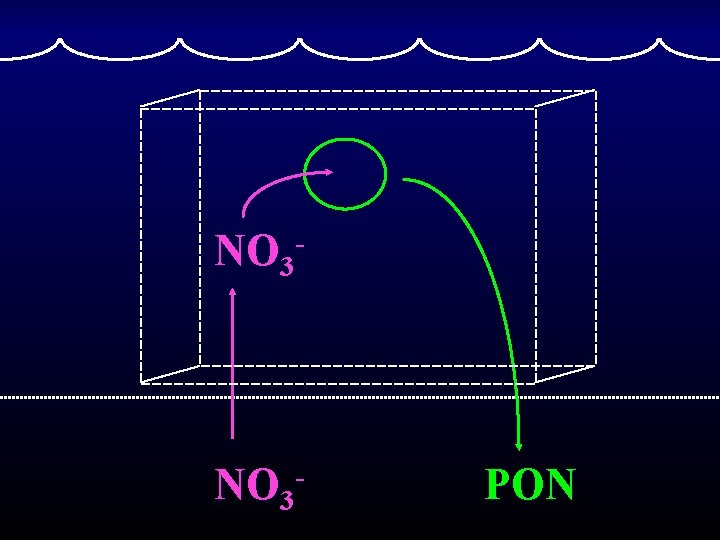
NO 3 - PON
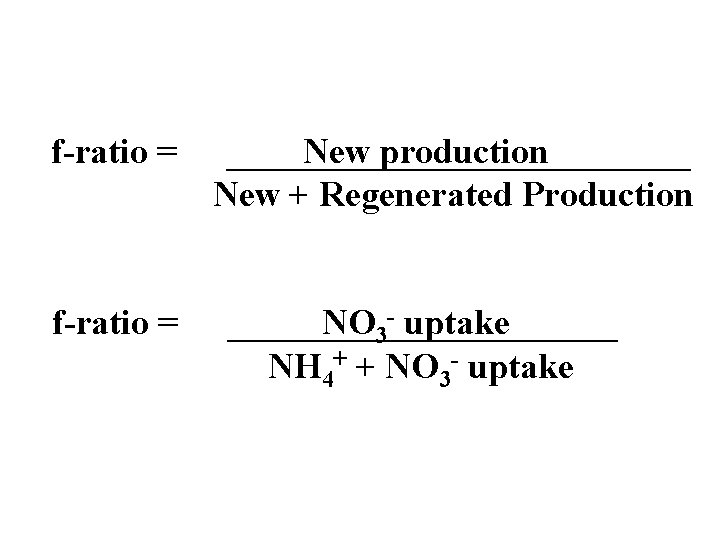
f-ratio = New production New + Regenerated Production NO 3 - uptake NH 4+ + NO 3 - uptake
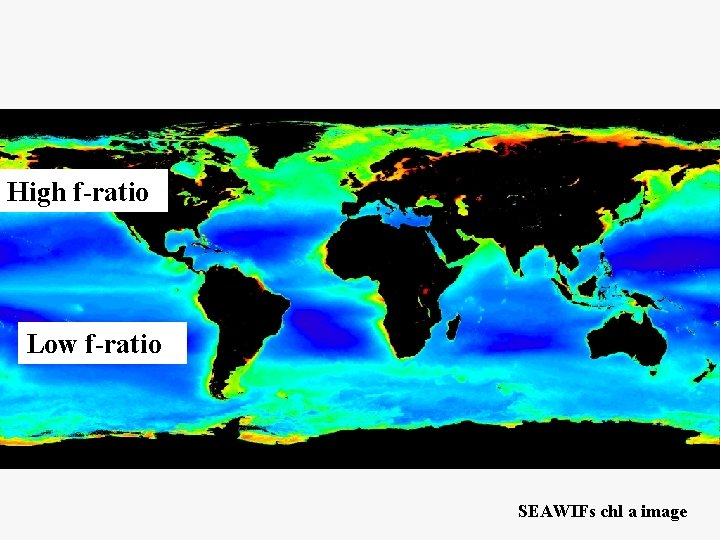
High f-ratio Low f-ratio SEAWIFs chl a image

Is the amount of N in the ocean in steady state?
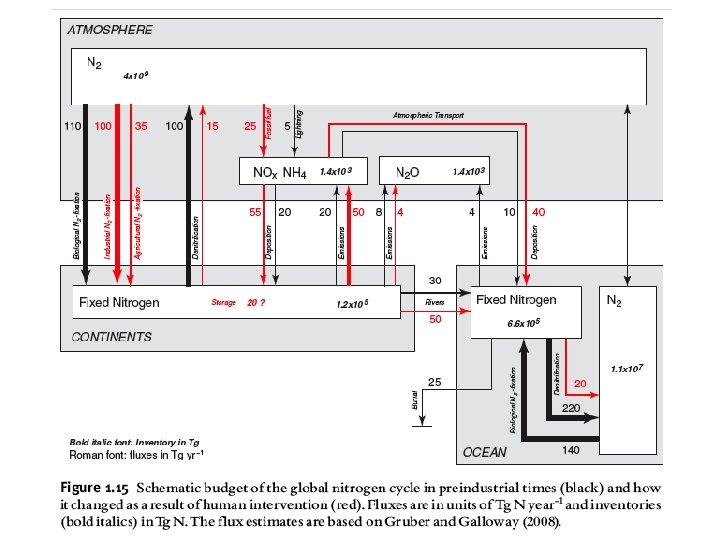
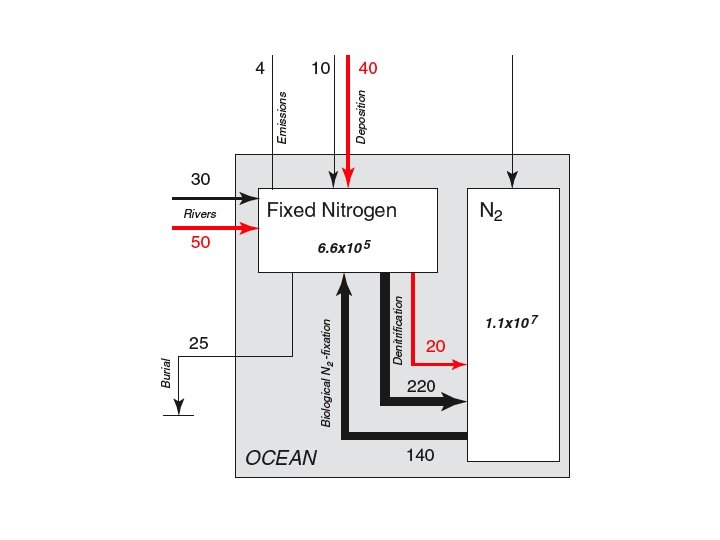
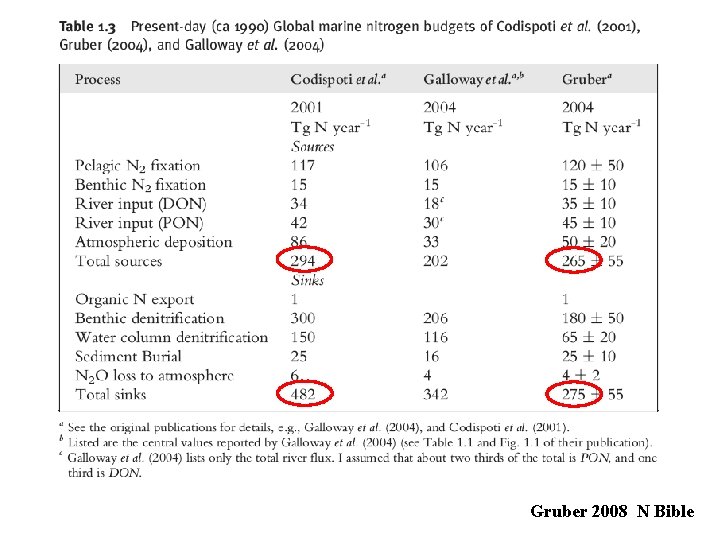
Gruber 2008 N Bible

Outline: 1. Types & distribution 2. The Redfield Ratio 3. The nitrogen cycle 4. Liebig’s Law of the Minimum 5. New & regenerated production 6. Is the N cycle in steady state?

% Degrees earned by women 1966 -2006 (US) Bach. Ph. D Tenured Biol/Agricultural 60 48 14 Chemistry 52 35 21 Earth, Atm. Ocean 51 34 22 Why So Few? 2010. AAUW

Discrimination? Hiring Publishing Funding Williams & Ceci (2011) PNAS
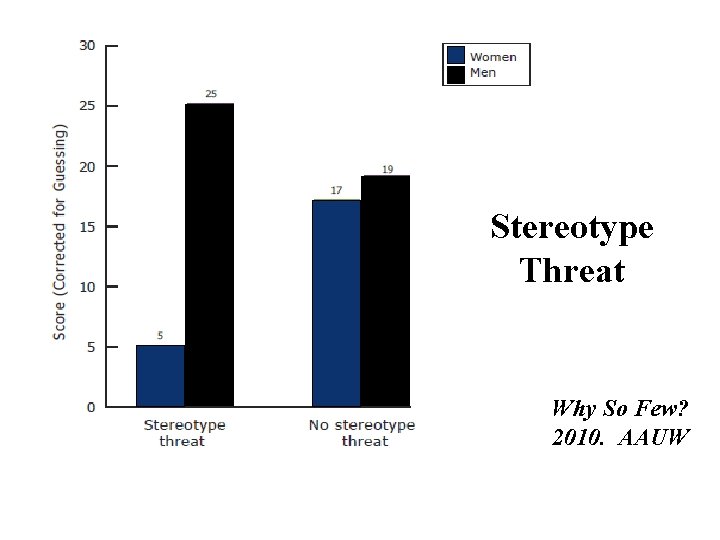
Stereotype Threat Why So Few? 2010. AAUW

- Slides: 65