Nitrogen Cycle Nitrogen n Most abundant element in

Nitrogen Cycle

Nitrogen n Most abundant element in the atmosphere n 78% of the air we breathe is N₂
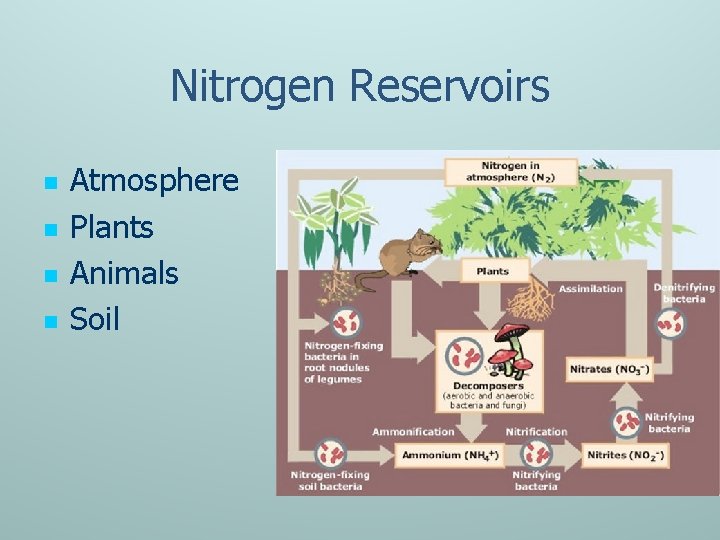
Nitrogen Reservoirs n n Atmosphere Plants Animals Soil
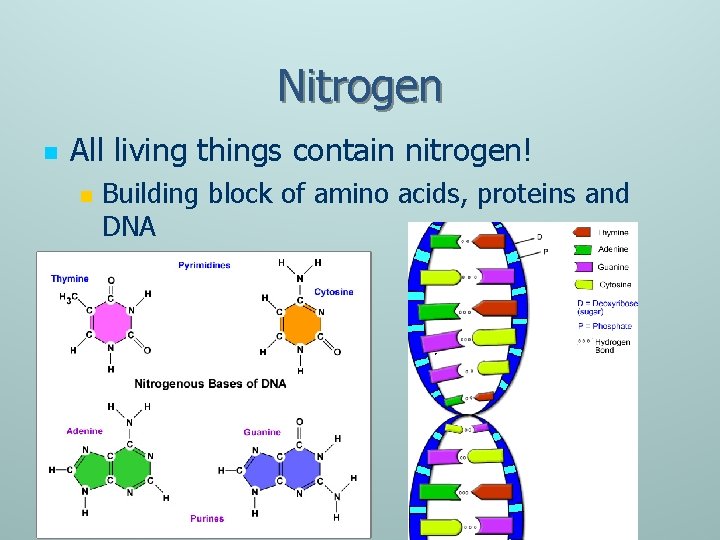
Nitrogen n All living things contain nitrogen! n Building block of amino acids, proteins and DNA

Nitrogen n Organisms cannot use Atmospheric nitrogen (N₂) n N₂ has to become “fixed” so organisms can use it

Nitrogen Fixation 1. Biological: Bacteria and fungi n Bacteria forms a mutualistic relationships with plants

Nitrogen Fixation 2. Atmospheric: Lightning n Lightning has enough energy to break the bonds in N₂ creating NO₂

Nitrogen Fixation 3. Industrial: Fertilizer § Ammonia is formed under high pressure

Haber - Bosch n n German scientists who developed fertilizer Same process created chemical warfare

Chemical Warfare n n WWI 15, 000 died in 1 st battle Mustard gas

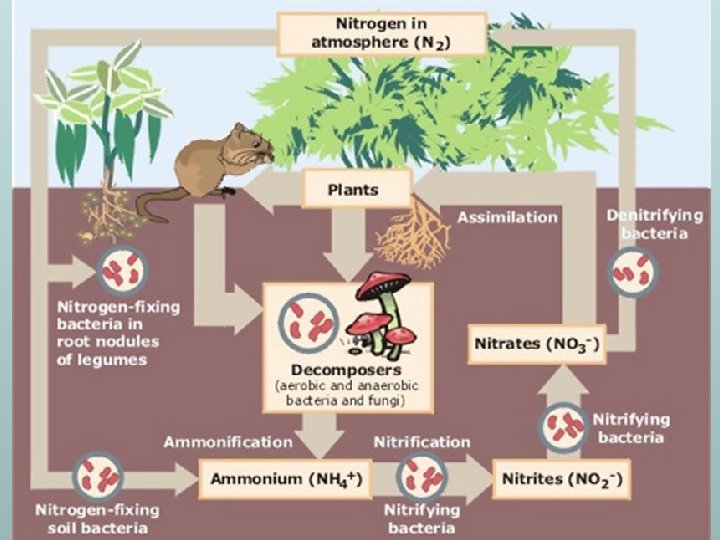
n n Usable: Ammonia: NH 3 Ammonium: NH₄ Nitrates: NO₃ Unusable: Atmospheric: N₂ Nitrites: NO₂ LIFE ON EARTH DEPENDS ON BACTERIA AND FUNGI THAT TRANSFORM NITROGEN INTO USEABLE FORMS n Plants cannot use atmospheric nitrogen N ₂ n Animals must get their nitrogen from eating plants and other animals

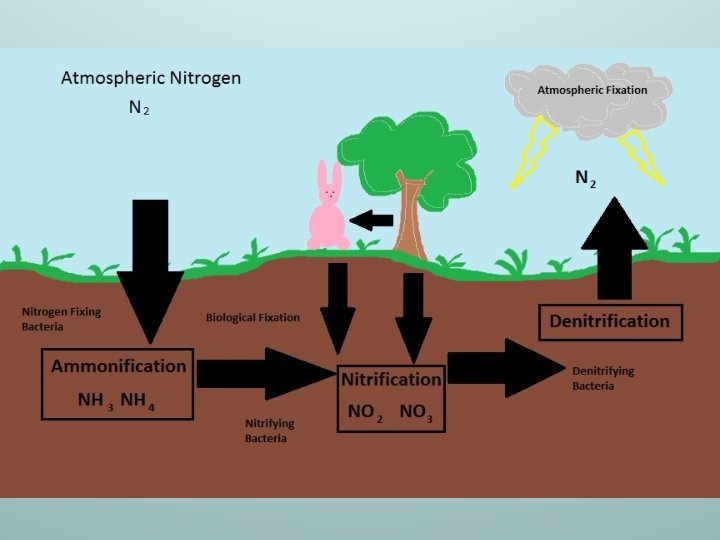
Ammonification n n The process of converting atmospheric nitrogen (N₂) to ammonia NH 3 and ammonium NH₄ N₂ NH 3 OR NH₄
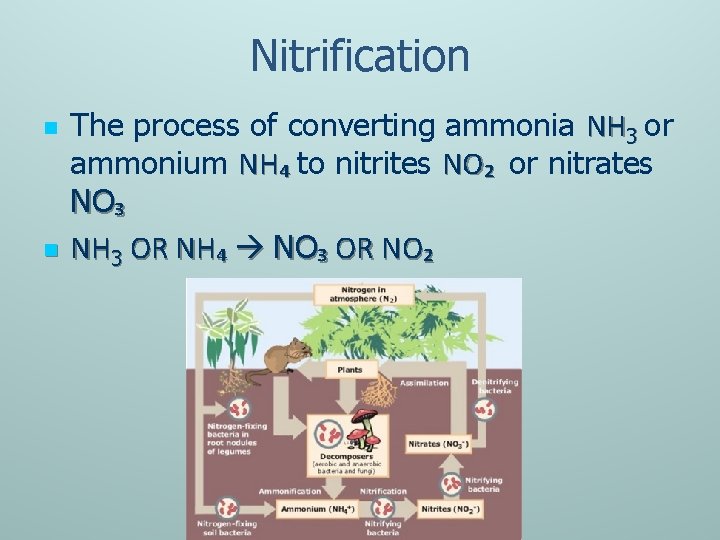
Nitrification n n The process of converting ammonia NH 3 or ammonium NH₄ to nitrites NO₂ or nitrates NO₃ NH 3 OR NH₄ NO₃ OR NO₂
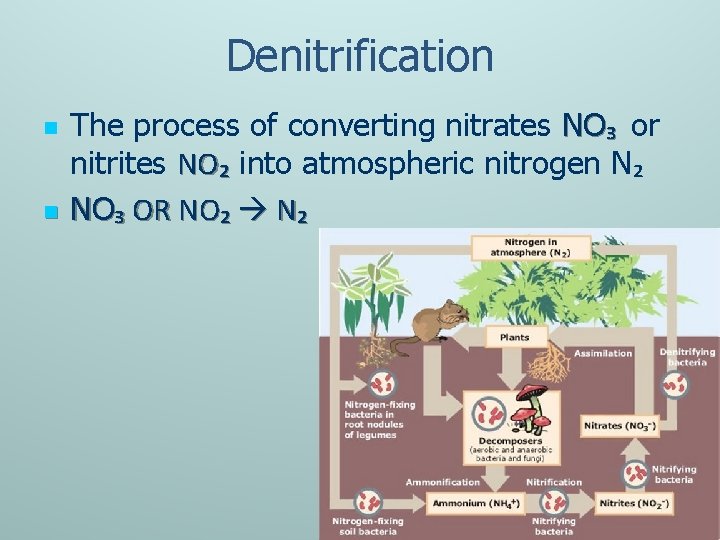
Denitrification n n The process of converting nitrates NO₃ or nitrites NO₂ into atmospheric nitrogen N₂ NO₃ OR NO₂ N₂
- Slides: 16