Nitration of Methyl Benzoate Multistep Synthesis A 50
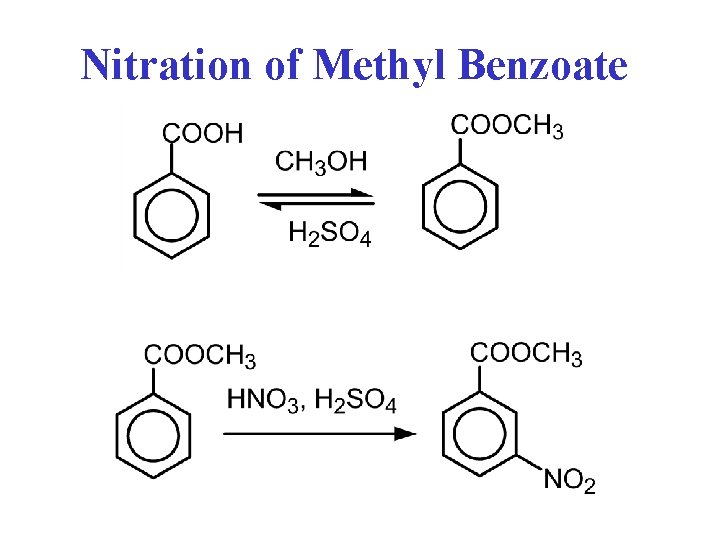
Nitration of Methyl Benzoate

Multistep Synthesis A 50% B 50% Yield = 25% C
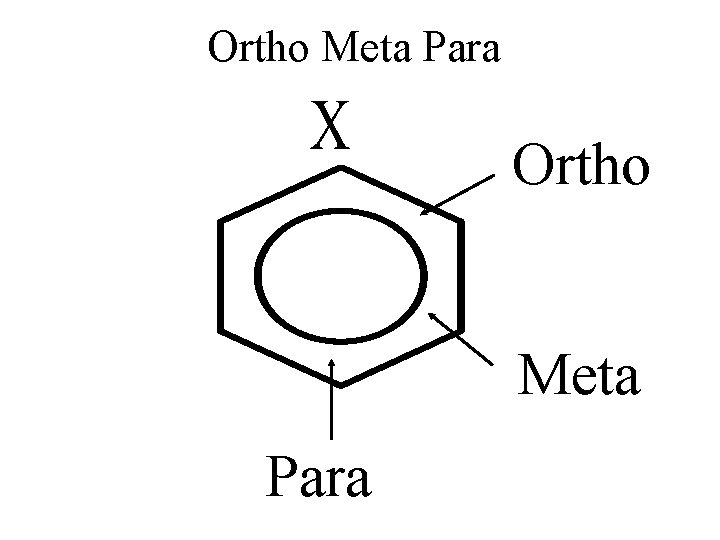
Ortho Meta Para
Number Ring 6 2 5 3 4
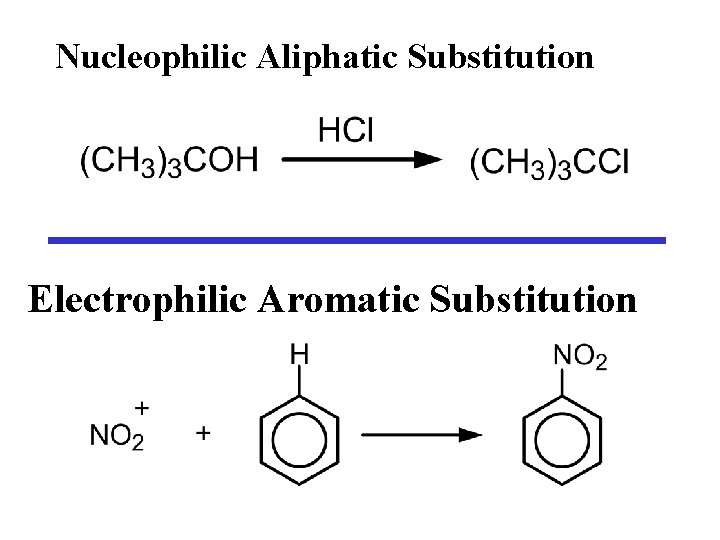
Nucleophilic Aliphatic Substitution Electrophilic Aromatic Substitution
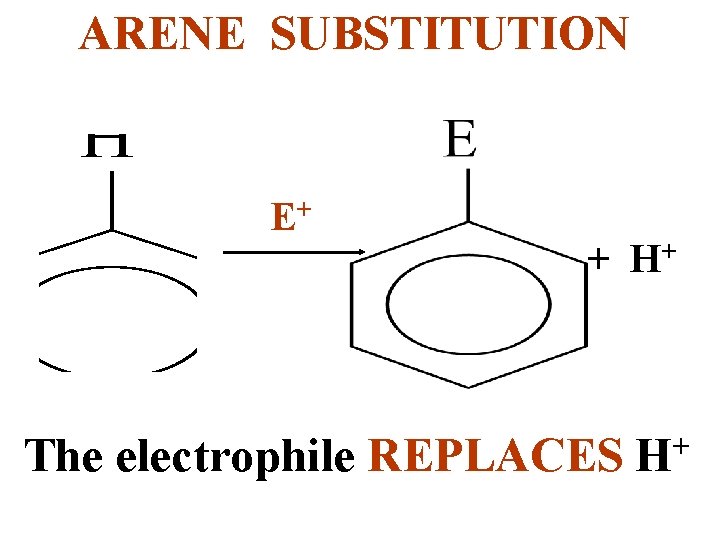
ARENE SUBSTITUTION E+ + H+ The electrophile REPLACES H+
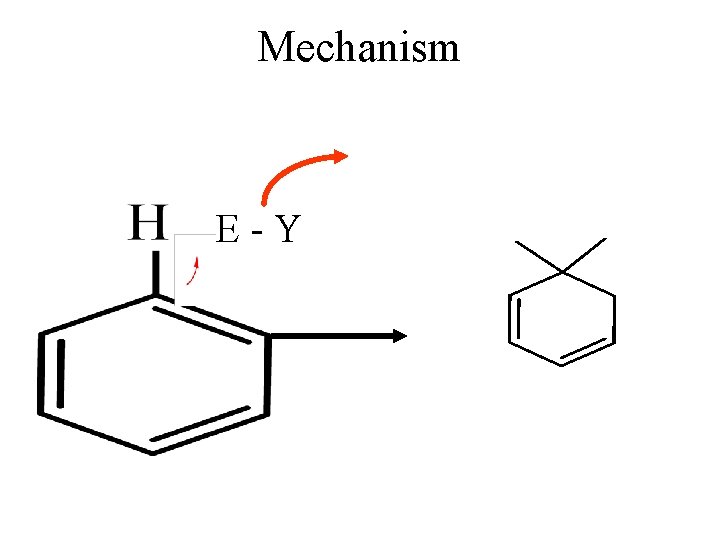
Mechanism E-Y
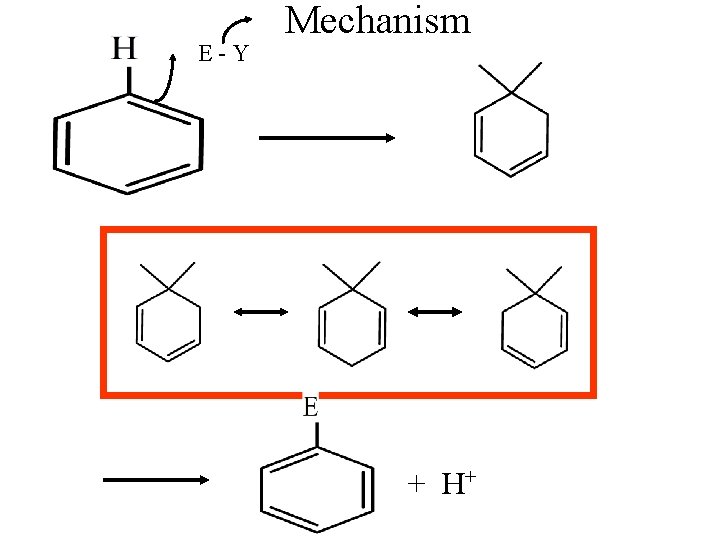
E-Y Mechanism + H+
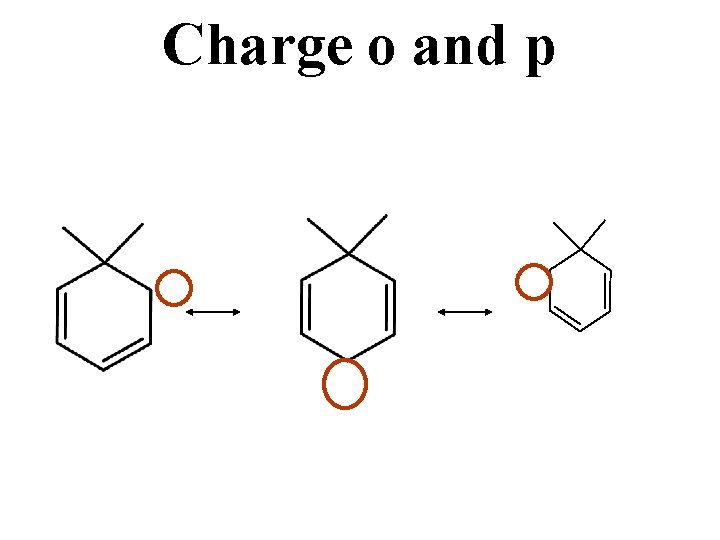
Charge o and p
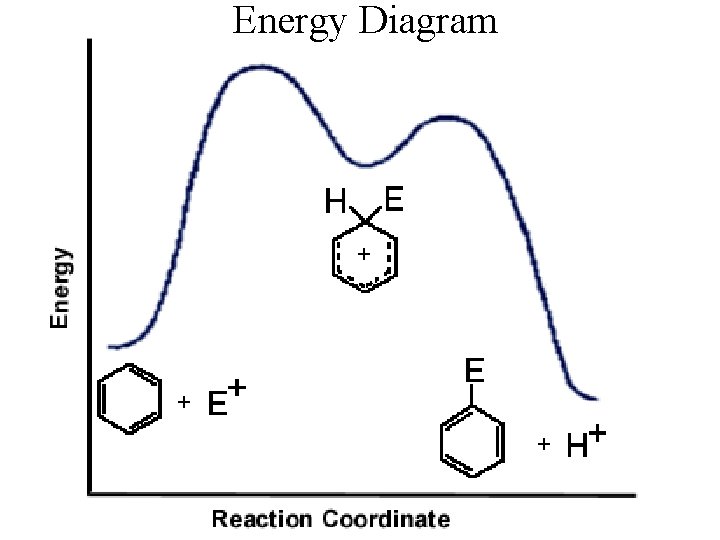
Energy Diagram
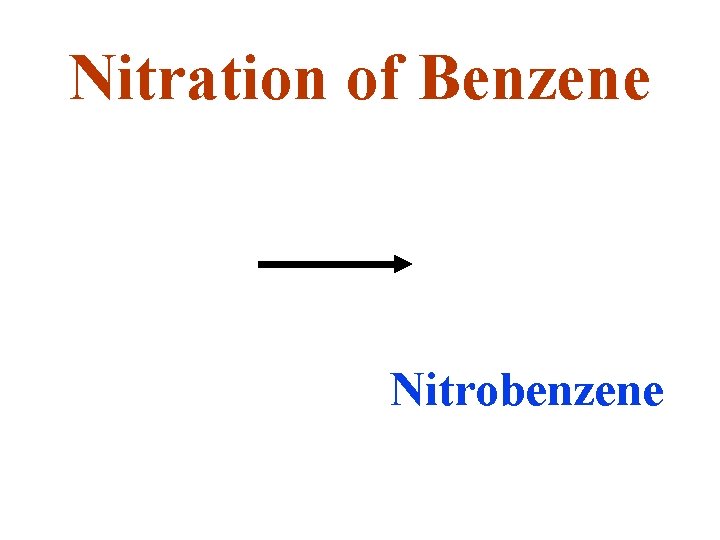
Nitration of Benzene Nitrobenzene
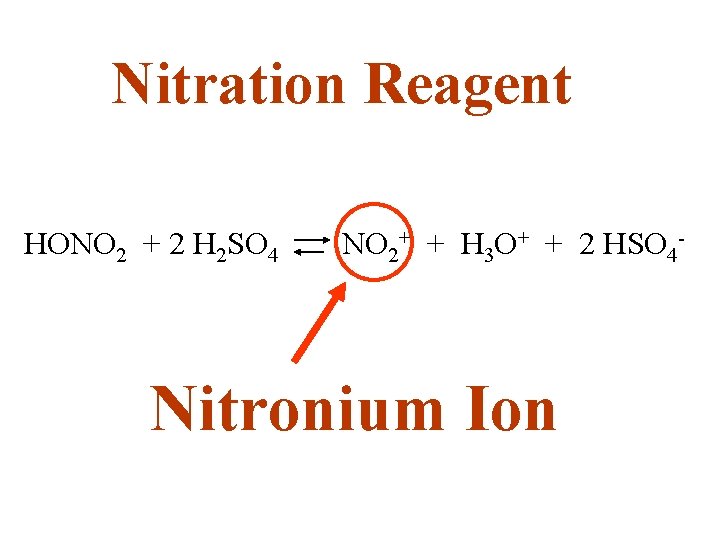
Nitration Reagent HONO 2 + 2 H 2 SO 4 NO 2+ + H 3 O+ + 2 HSO 4 - Nitronium Ion
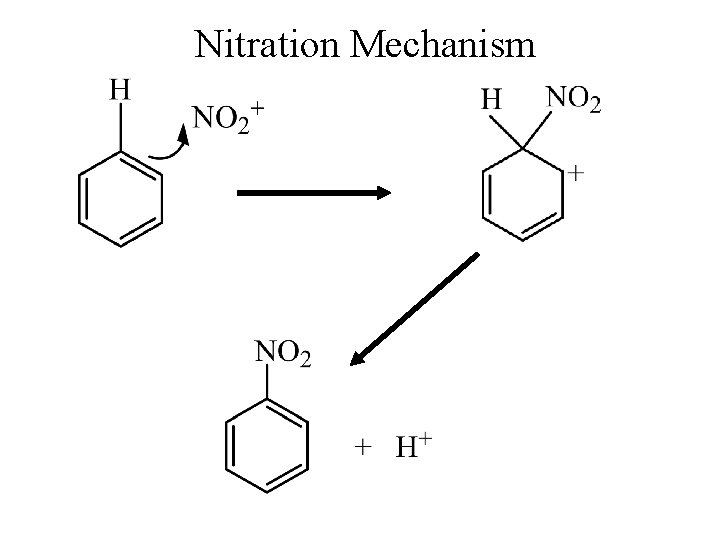
Nitration Mechanism
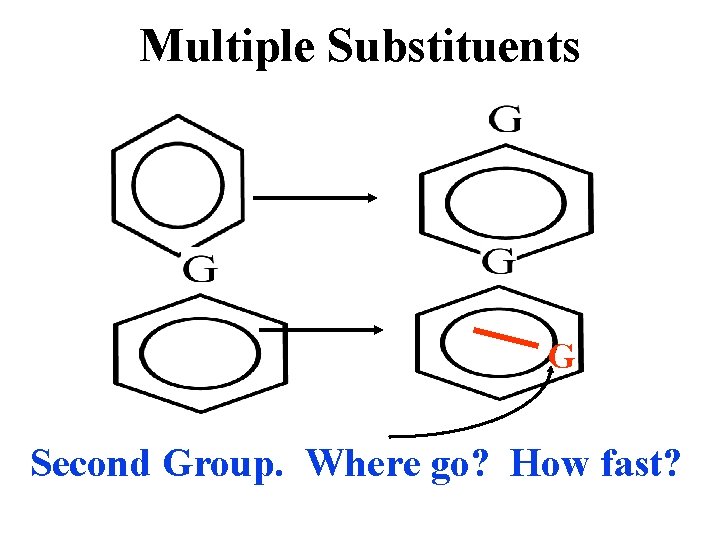
Multiple Substituents G Second Group. Where go? How fast?
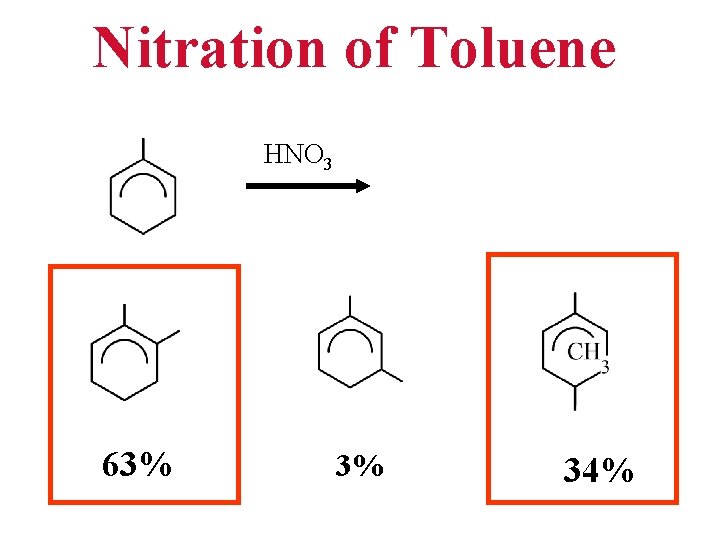
Nitration of Toluene HNO 3 63% 3% 34%
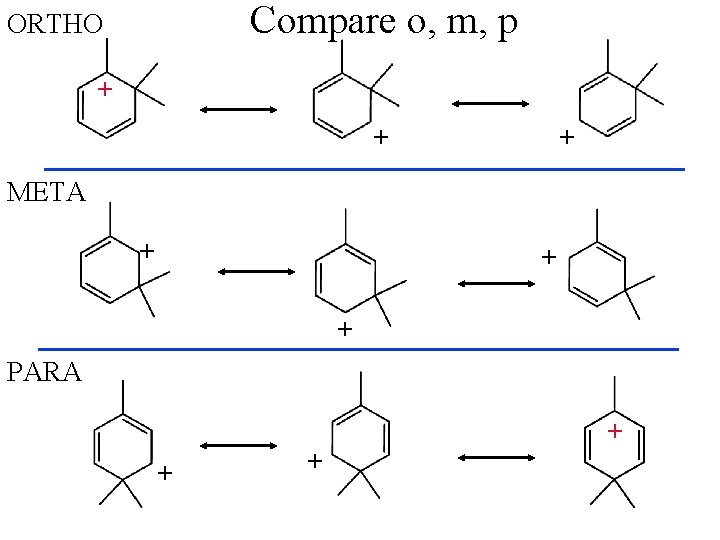
Compare o, m, p ORTHO + + + META + + + PARA + + +

Electron Donating Groups Favor: ORTHO and PARA. Electron Donating Ortho Para
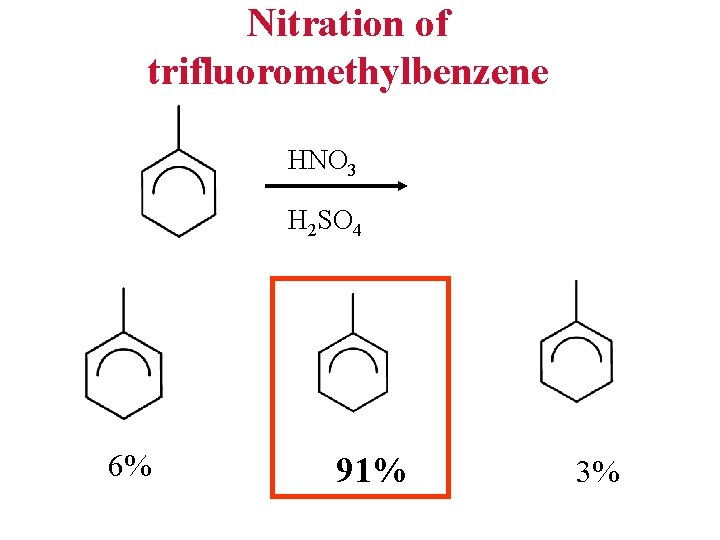
Nitration of trifluoromethylbenzene HNO 3 H 2 SO 4 6% 91% 3%
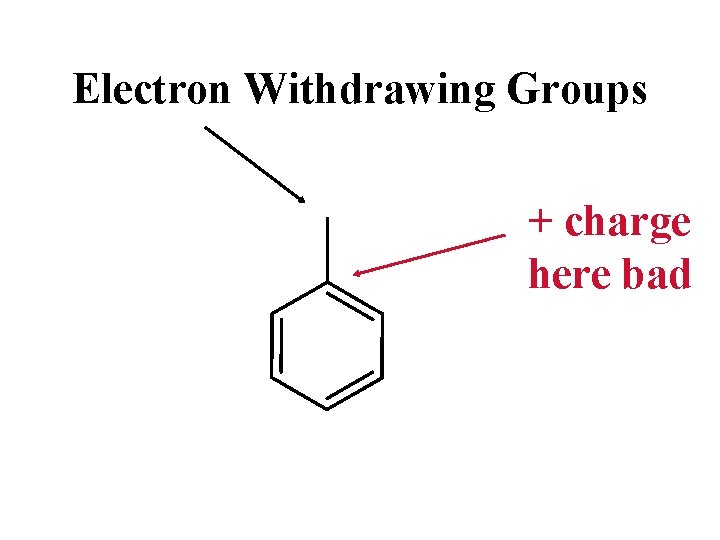
Electron Withdrawing Groups + charge here bad
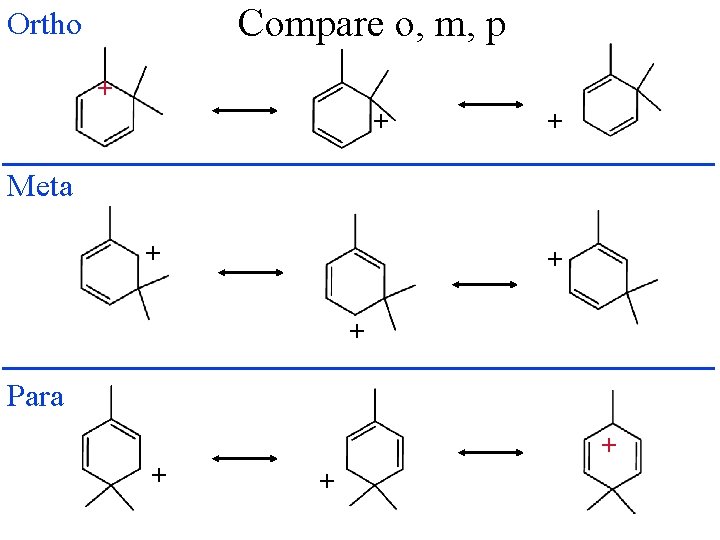
Compare o, m, p Ortho + + + Meta + + + Para + + +
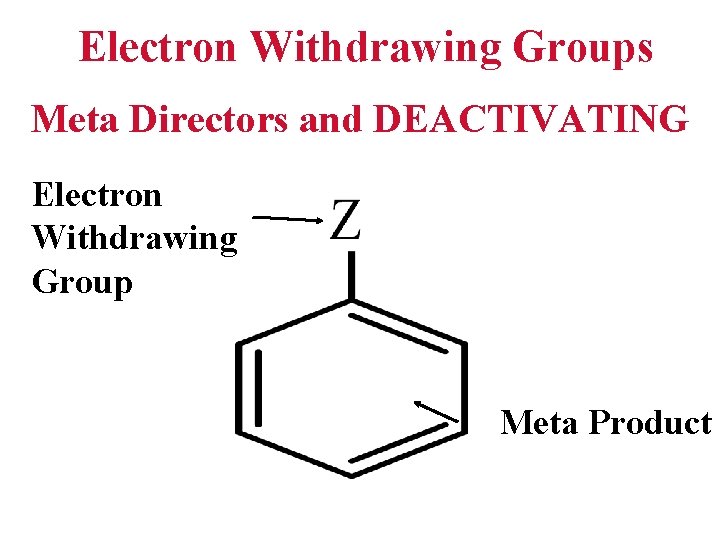
Electron Withdrawing Groups Meta Directors and DEACTIVATING Electron Withdrawing Group Meta Product

Ortho-Para Directors -NH 2 -NHR -OH -OR O -NHCR O -OCR Very Activating

Ortho-Para Directors -R Activating -Ar Activating -Halogens Deactivating
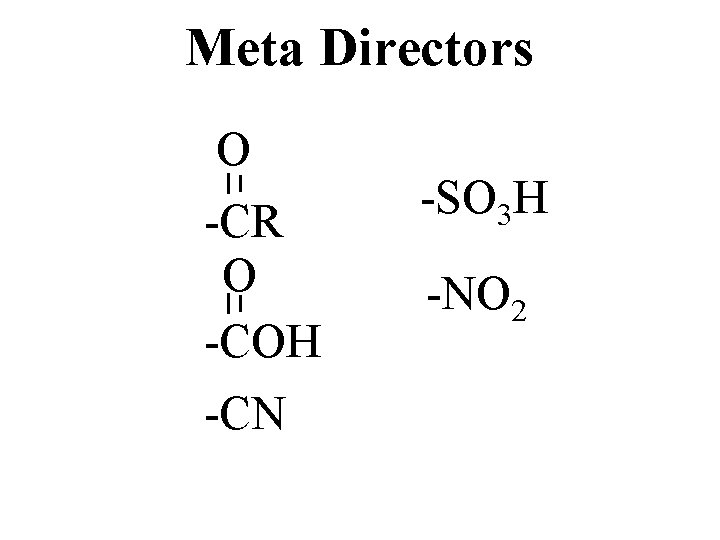
Meta Directors O -CR O -COH -CN -SO 3 H -NO 2

Main Reaction meta Side Products

Procedure 1. Dissolve methyl benzoate in H 2 SO 4 2. Mix HNO 3 and H 2 SO 4 at 0 o. C 3. Add HNO 3 / H 2 SO 4 dropwise to methyl benzoate at 0 o. C

Procedure 4. Let stand at room temperature 10 minutes 5. Pour onto ice

Filter

Wash Recrystallize from methanol Dry Weigh m. p.
- Slides: 29