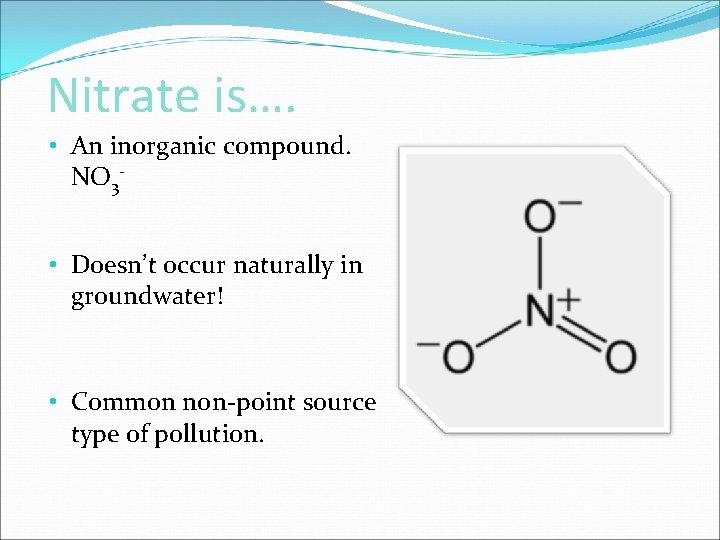
Nitrate Nitrate is An inorganic compound NO 3










- Slides: 15

Nitrate

Nitrate is…. • An inorganic compound. NO 3 • Doesn’t occur naturally in groundwater! • Common non-point source type of pollution.

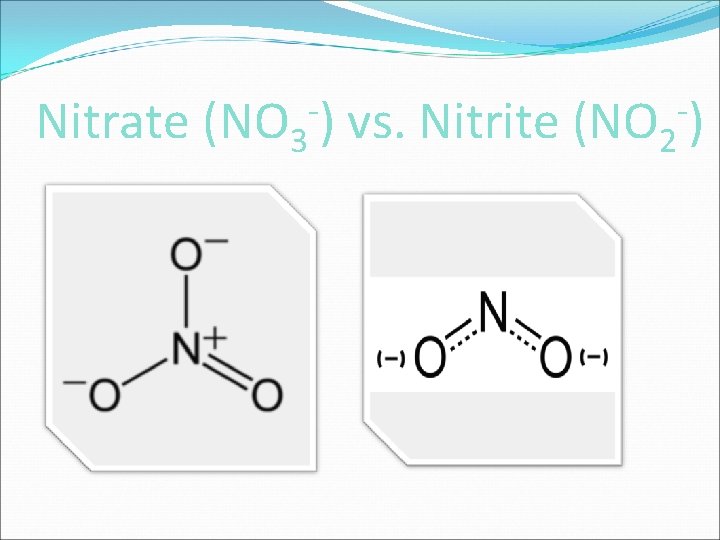
-) Nitrate (NO 3 vs. Nitrite (NO 2 -)

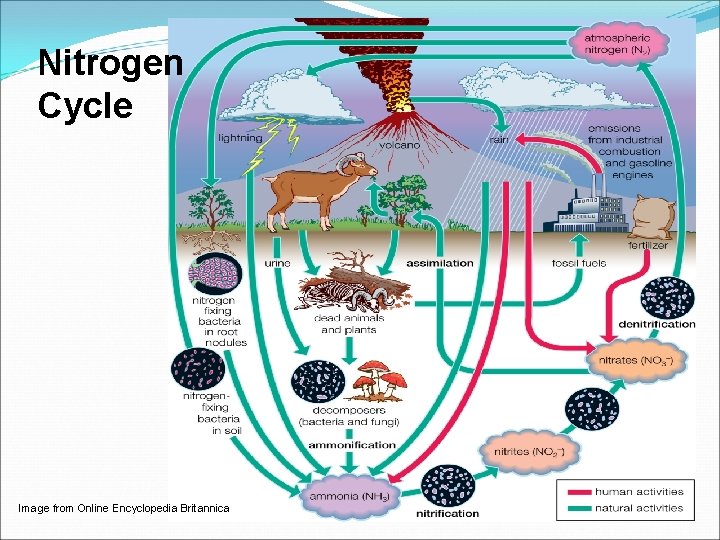
Nitrogen Cycle Image from Online Encyclopedia Britannica

Nitrite in the news • Sodium Nitrite (Na. NO 2) is used commonly as a food preservative. • Foods that contain the highest amounts of Na. NO 2 are smoked and cured meats.

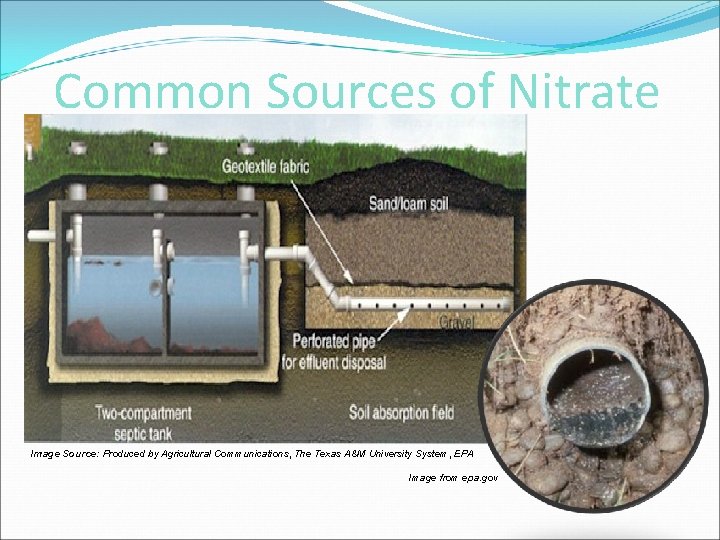
Common Sources of Nitrate Tom Gentle, Oregon State University EESC

Common Sources of Nitrate Image Source: Produced by Agricultural Communications, The Texas A&M University System, EPA Image from epa. gov

Common Sources of Nitrate Images from Utah State Image from epa. gov

How does nitrate get into drinking water? Used in plants when it is in the root zone. Nitrate is a negatively charged ion Isn’t adsorbed (held) by negatively charged soil particles. Moves easily with water. Once it is past the root zone it moves rapidly downward into groundwater.

What is a safe level of nitrate in drinking water? Health Standard for PUBLIC DRINKING WATER SUPPLIES is 10 ppm nitrate nitrogen No regulation of PRIVATE DRINKING WATER SUPPLIES If nitrate is detected, learn more about risk to you.

What are the health issues with nitrate in drinking water? Methemoglobinemia or “Blue Baby Syndrome” High level of nitrate consumption cause a reduction in the oxygen-carrying capacity of blood by increasing the amount of methemoglobin. Methemoglobin is a non-oxygen-carrying enzyme Infants will start to show obvious symptoms of cyanosis – a bluish color to the skin.

What are the health issues with nitrate in drinking water? Unconfirmed as a contributing factor in: Diabetes Reproductive difficulties Thyroid Dysfunction Decreased liver function Respiratory infections Cancer

Nitrate in the groundwater now! Treatment options Reverse osmosis Distillation Protect groundwater supplies from further contamination What are some steps that can be taken?


Nitrate Screening Screen your samples from home, a control, and a mystery sample.