Nitinol Copyright 2012 Board of Trustees University of

Nitinol Copyright © 2012 Board of Trustees, University of Illinois. All rights reserved.

Objectives • To investigate the unique properties of an alloy called Nitinol • To design an experiment using Nitinol • To understand the connection between Nitinol’s atomic structure and its physical characteristics • To learn about Nitinol’s applications 2

Nanotechnology • Some scientists and engineers develop smart materials 3

Smart materials • Any material engineered at the nanoscale to perform specific tasks 4

Smart fabrics 5

Self-healing materials • Studied and made at University of Illinois • Can repair themselves • Can be used in cell phones and other devices 6
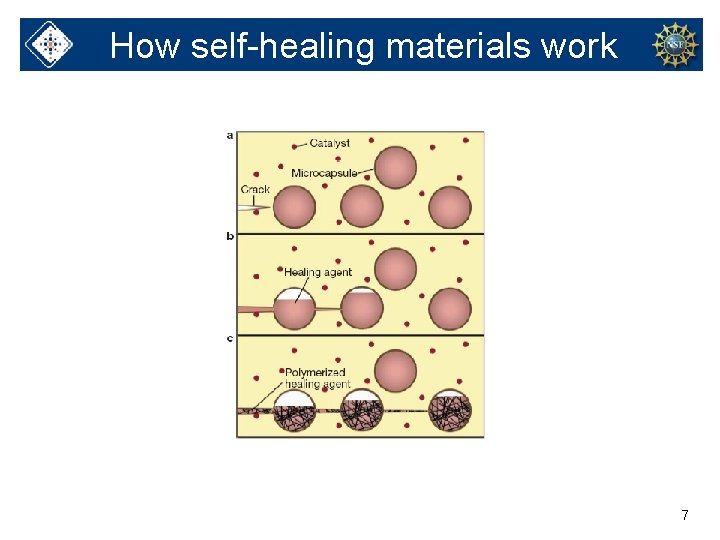
How self-healing materials work 7

Similar to epoxy 8
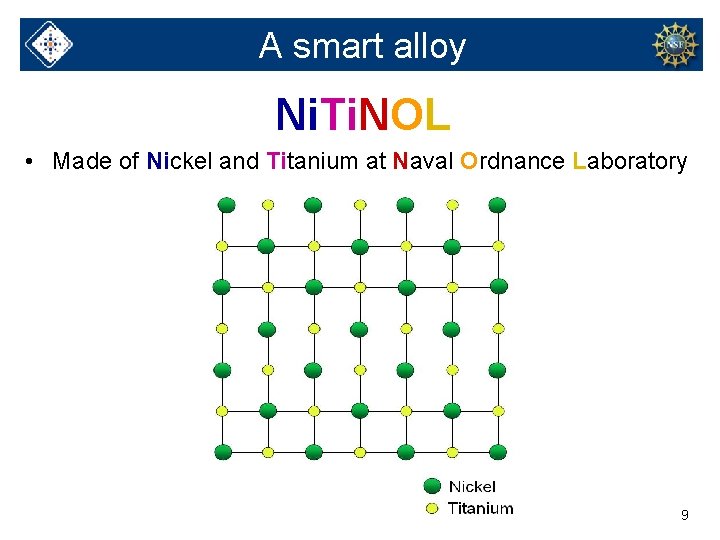
A smart alloy Ni. Ti. NOL • Made of Nickel and Titanium at Naval Ordnance Laboratory 9

Nitinol—a smart material? • Bend it • Heat it • What happens? 10
Shape Memory Alloy (SMA) • Nitinol is a “shape memory alloy” • What does that mean? • What is happening at the nanoscale? 11
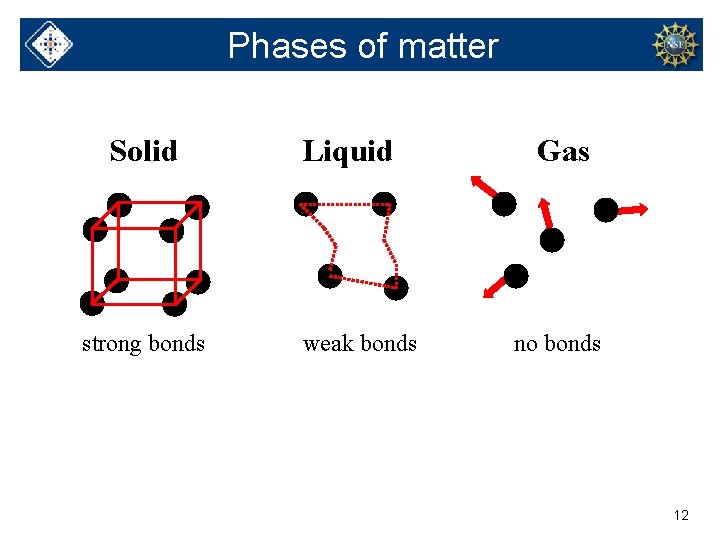
Phases of matter Solid strong bonds Liquid weak bonds Gas no bonds 12
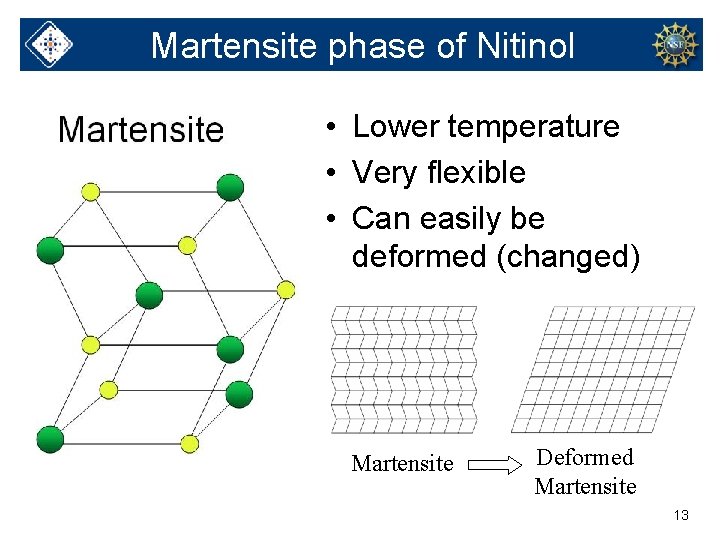
Martensite phase of Nitinol • Lower temperature • Very flexible • Can easily be deformed (changed) Martensite Deformed Martensite 13
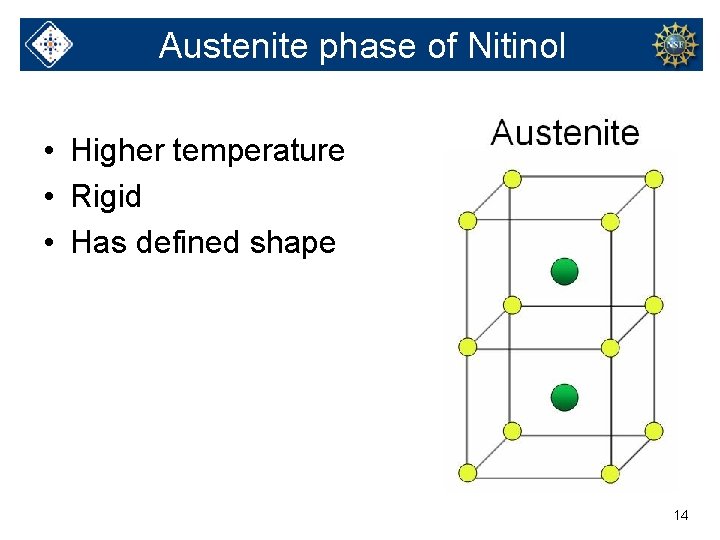
Austenite phase of Nitinol • Higher temperature • Rigid • Has defined shape 14
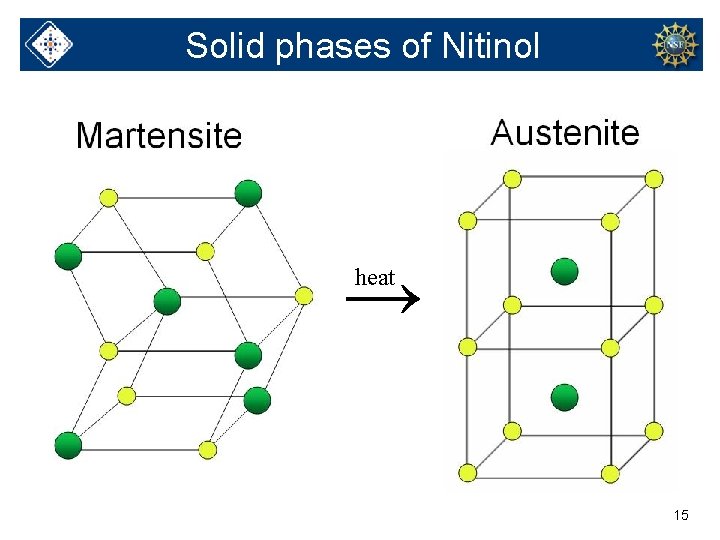
Solid phases of Nitinol → heat 15
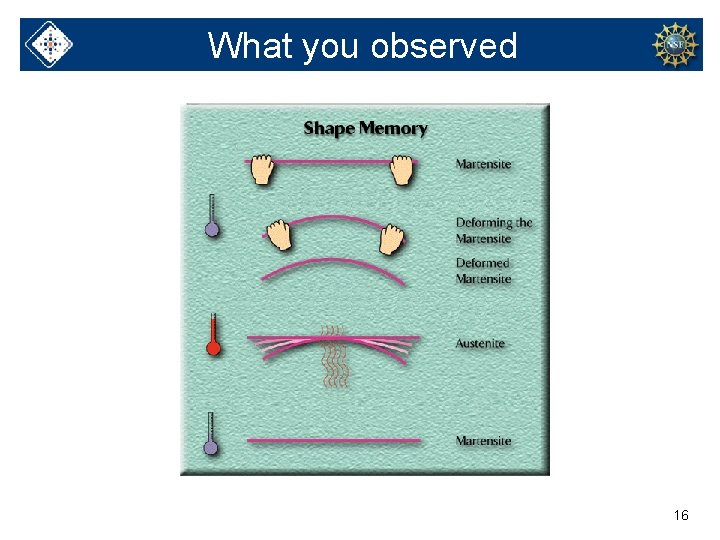
What you observed 16
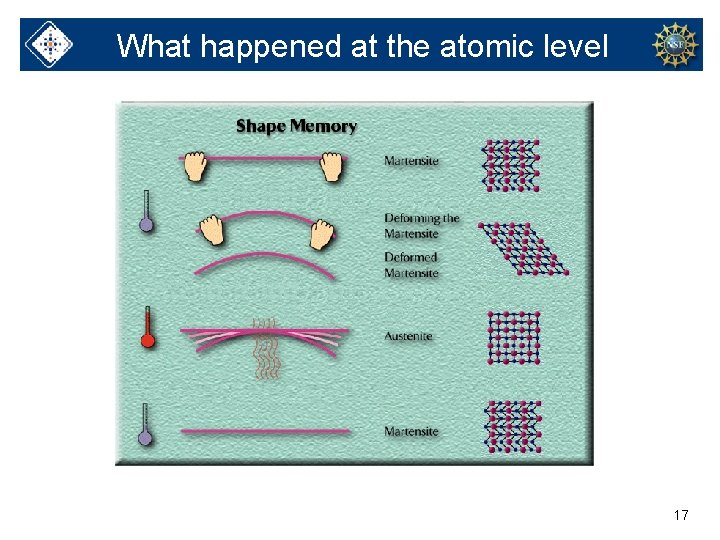
What happened at the atomic level 17
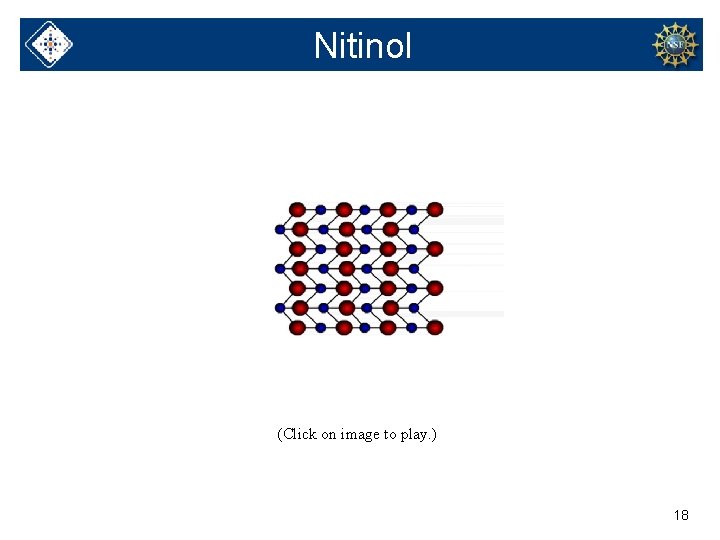
Nitinol (Click on image to play. ) 18
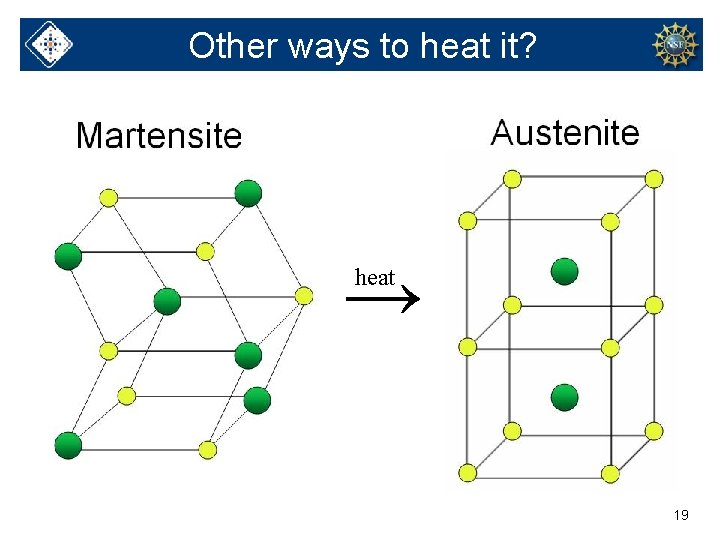
Other ways to heat it? → heat 19
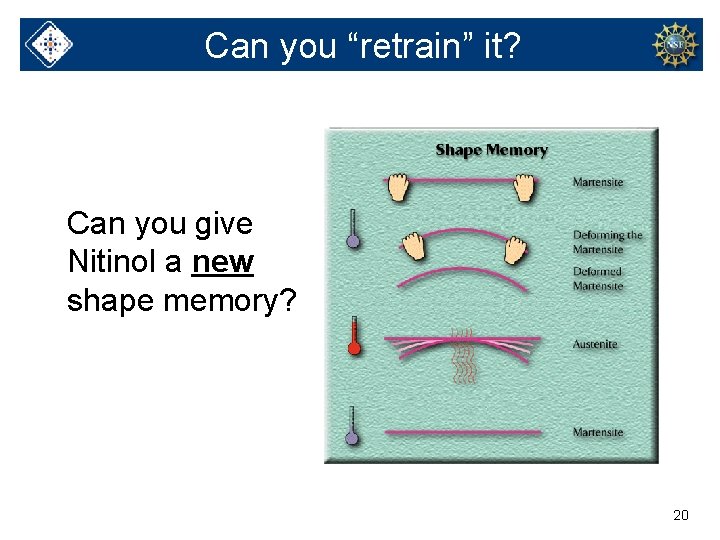
Can you “retrain” it? Can you give Nitinol a new shape memory? 20

Properties of Nitinol • Shape memory alloy • Elastic • Does not react with water or oxygen • Can be used in the body 21

Austenite temperature • Slight difference in ratio of Ni to Ti can change the temperature from -50°C to +95°C • Engineered for different functions 22

Think! • How do you think Nitinol might be useful? 23
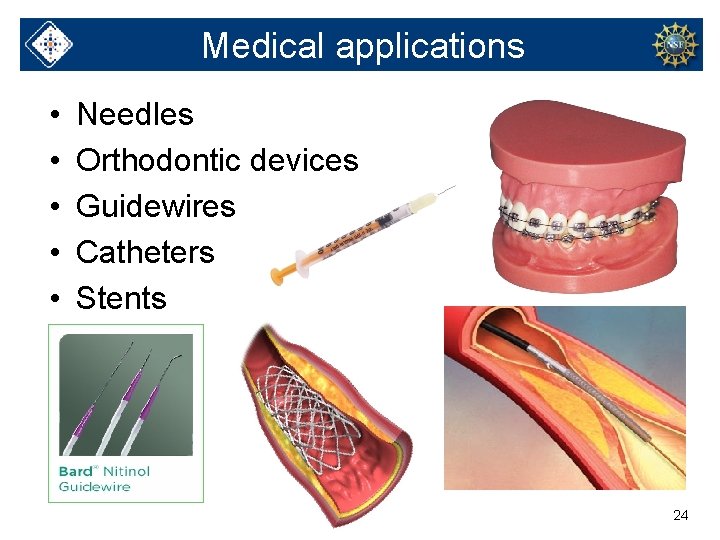
Medical applications • • • Needles Orthodontic devices Guidewires Catheters Stents 24

Other applications 25

Summary 1. Learned about Nitinol’s properties 2. Heated it 3. Found other ways of heating it 4. Retrained it 5. Learned about its applications 26
- Slides: 26