NIHR focus on Health Technology Assessment HTA Portfolio
































- Slides: 35

NIHR – focus on Health Technology Assessment (HTA) Portfolio www. nihr. ac. uk Dottie Goble Senior Research Manager, Identification HTA NIHR Evaluation, Trials and Studies Coordinating Centre

Introduction • • HTA remit and outline Overview of application assessment process What makes a good application Tips for success www. nihr. ac. uk

A FEW FACTS Ø The largest of the NIHR research programmes. Ø Research about the effectiveness, costs and impact of healthcare treatments and tests. Ø Who are the programme’s evidence users: § § Policy decision-makers locally and nationally across health and care Members of the public Patients NHS health professionals www. nihr. ac. uk

A FEW FACTS 2015/2016 440 250 67 99 131 43, 432 110, 781 1, 801 Live projects Active trials New projects Final reports Articles in peer reviewed journals Participants were recruited Participants in RCTs Peer reviewers www. nihr. ac. uk

Health Technology Assessment Programme (HTA) • Research evidence on the effectiveness, costs and broader impact of healthcare treatments and tests • For those who plan, provide or receive care in the NHS and community • “Health technology” is any intervention used to promote health, prevent and treat disease and improve rehabilitation and long term care, including drugs, devices, procedures, settings of care and screening • Primary research and evidence synthesis • Evidence of efficacy but uncertainty exists • Programme Director – Professor Hywel Williams www. nihr. ac. uk

Typical HTA study Multidisciplinary and multi-centre Effectiveness and cost-effectiveness (usually estimate £/QALY) Pragmatic and externally valid Median number of patients = 700 (Range of 15 to 75, 000 across current projects) o Average duration ~4 years and cost £~1. 5/2 m o Protocols available on web site Types of studies funded: o randomised controlled trials and non-randomised trials o stand-alone pilot and feasibility studies (where there is evidence they will lead to a full trial) or embedded pilot/ feasibility o cohort studies (retrospective or prospective) o adaptive and efficient study designs, methodological studies o evidence synthesis and modelling studies o o www. nihr. ac. uk

HTA Project Examples Gastric Bypass, adjustable gastric Banding or Sleeve gastrectomy surgery to treat severe and complex obesity: a multi-centre randomised controlled trial (The By-Band-Sleeve Study) Professor Jane Blazeby, University of Bristol, £ 3, 939, 934 (119 months) Add-Aspirin Trial: A phase III double-blind placebo-controlled randomized trial assessing the addition of aspirin after standard primary therapy in early stage common solid tumours Professor Ruth Langley, University College London £ 3. 1 M (144 months) A systematic review of psychological interventions to improve motivation for self-management in people with type 1 and type 2 diabetes Kirsty Winkley, King’s College London £ 180 K (18 months) www. nihr. ac. uk

HTA programme applications and assessment

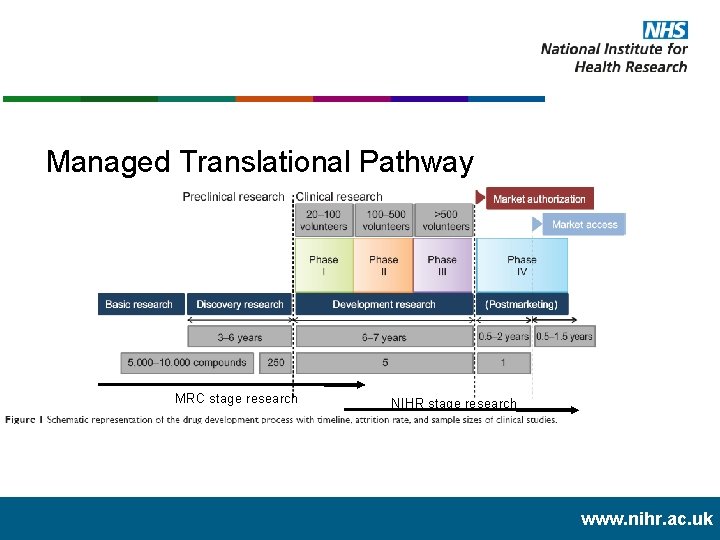
Managed Translational Pathway MRC stage research NIHR stage research www. nihr. ac. uk

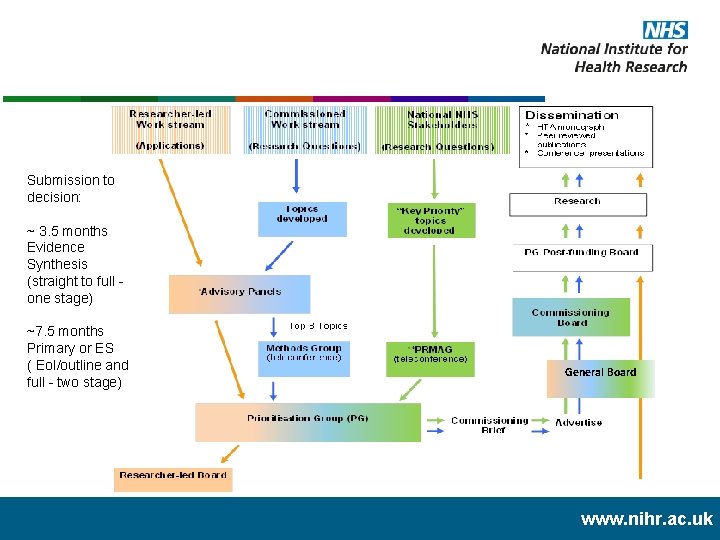
Submission to decision: ~ 3. 5 months Evidence Synthesis (straight to full - one stage) ~7. 5 months Primary or ES ( Eo. I/outline and full - two stage) General Board www. nihr. ac. uk

Commissioned work stream • Designed to meet the needs of decision makers, typically but not exclusively within NHS. • Topics prioritised by expert panels and commissioning briefs advertised to address identified evidence need. • Regular calls - up to three times year • Single stage or two stage process dependant on programme and call. • Primary research and evidence synthesis. www. nihr. ac. uk

Researcher-led work stream • Calls for applications on research topics/questions directly proposed by researchers. • Highlight notices/ themed calls in areas of specific need. • Applications prioritised on NHS or other information need by advisory panels/boards. Dependant on programme and call: • Regular calls - up to three times year • Single stage or two stage process. • Primary research and evidence synthesis. www. nihr. ac. uk

Commissioned vs Researcher-led? Commissioned • Importance for research identified by programme • Read the commissioning brief • Check the supporting specification document and remit info • Ensure your project fits – tailor if needed Researcher-led • Check the remit of the programme • Search for funded projects (website) for examples info on costs, duration, methodology, team etc • Justify importance of your research Contact the programmes in case of query – email in summary for remit advice www. nihr. ac. uk

NIHR Themed calls • NIHR themed calls are issued to meet an identified health challenge or government priority • Calls issued once a year • Cross NIHR initiative with programme fit / flex • Different timings across programmes • Previous calls have focused on: Obesity, Dementia , Surgery, Primary Care Interventions, Very Rare Diseases, Long term conditions in childhood, Multi morbidities in older people, Antimicrobial resistance • Applications in all previous themed call areas continue to be welcomed by NIHR via researcher-led calls • Recent calls -Prevention and treatment of obesity - Antimicrobial resistance www. themedcalls. nihr. ac. uk www. nihr. ac. uk

Board membership Approximately 20 members • All experts in their respective fields • Eclectic mix • Clinical, statistical, health economics, quantitative, qualitative, organisational expertise, trialist… • Expertise relevant to programme remit At least Two public or patient members www. nihr. ac. uk

Expert Review ~1, 000+ pa Commissioning briefs – expert views sought Outline proposals – option for 1 clinical /professional review Full Primary research proposals - 5 to 6 reviewers Reviews generally comprise: • 2 -3 Clinical or professional experts • 2 methodologists (statistical/health economics/ trialist) • 1 public or patient review + method/International review on higher cost studies > £ 2 m Applicants invited to respond to reviewer comments www. nihr. ac. uk

What happens at the board All members receive outlines and full application papers • Applications • Reviewers comments • Applicants response to referees comments • Approx 35 -40 outlines & 20 full proposals are considered per Board meeting • Three Designated Board Members per application (one lead DBM + 2 supporting DBMs of any discipline) • Specific comments on Public and patient involvement Outcome and board feedback www. nihr. ac. uk

Applying for research funding - Tips for success Know your target • • • Make sure you check aims / programme remit How and when will the application be assessed? What is the scale of a typical award? Who has received previous awards? What research is already in the portfolio e. g. systematic reviews, ongoing research? • How does the proposals relate to NICE guidance? www. nihr. ac. uk

• ‘Importance’: The need for research • How much will patients and the NHS benefit from the proposed research? • How might the results help decision making? Need = frequency x severity x impact of technology x evidence deficit Discounted for time to produce evidence IMPORTANT QUESTION ON AN IMPORTANT TOPIC www. nihr. ac. uk

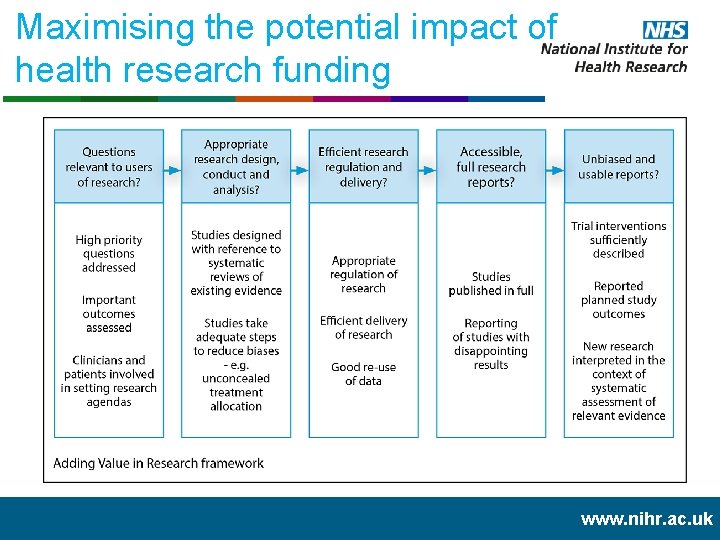
Maximising the potential impact of health research funding www. nihr. ac. uk

What do the panels expect? Clear HTA research question Based on clear PICO Summary of the burden of disease Incidence or prevalence Mortality and quality of life Place of the technology in the care pathway Thorough summary of current research Patient centred outcomes www. nihr. ac. uk

The ‘science’ bit Population: Sample size - Effect size Intervention – clear description of the treatment Comparators Best alternative, treatment as usual, placebos Outcomes Not composite, except by convention eg qalys Not surrogates, except established Systematic reviews – clear parameters Developed technologies – Esp complex interventions Team expertise Value for money – Cost / participant Board consider carefully. But what is VFM? www. nihr. ac. uk

Use available support in designing the right project – be innovative • Our Research Design Service (RDS) provides design and methodological support to health and social care researchers across England • INVOLVE is our national advisory group supporting active public involvement in NHS, public health and social care research • Our Clinical Trials Units (CTUs) provide specialist expert statistical, epidemiological and other advice and coordination to undertake successful clinical trials • Clinical Research Networks (CRN) across the UK to support development and delivery of clinical studies www. nihr. ac. uk

Ensure the proposal is feasible • • Design fits NHS Context Timing and timeline Evidence of feasibility If unclear consider a feasibility study – with pre-defined feasibility criteria – standalone / internal – clear route to main trial • Recruitment plan: Study sites, eligible patients, patient consents, follow-up, adequate retention of participants, expectations of service providers/patients – consider contingency arrangements • Leave enough time for study set up: – ethics and research governance approvals (and costs) www. nihr. ac. uk

Include public and patients (and pay them) Public and Patient involvement (PPI) matters to the NIHR - consider it carefully and justify your decisions Were patients and public actively involved in : • • Identifying the research topic or prioritising the research questions? If not why not? Preparing the application? If yes, give details. How PPI informed and/or influenced the development of the application and how patients and the public have been involved. If no, please explain why patient and public involvement was not thought necessary. Please indicate the ways in which patients and the public will be actively involved in the proposed research. If active involvement is planned, please give more details, including how it will benefit the research, the reasons for taking this approach and arrangements for training and support. If there are no plans for active involvement, please explain why it is not thought necessary or possible www. nihr. ac. uk

Produce a carefully costed proposal • Sorting out the budget takes time! • Is cost to address the health issue justified ? • Does the topic account for costs requested and provide VFM • Be realistic in costing & use plausible % FTE rates • Full applications scrutinised by accountants - check and challenge • Total costs should be identified at the outline stage • Account appropriately for – research costs, – NHS treatment costs (excess CRN) – NHS support costs www. nihr. ac. uk

Think about target audience/ presentation • Simple English for mixed audience – – – Clinical, methodologists, reviewer, panel /board Patient and public Tell the story well for the non-expert Clear plain English summary Follow the writing guides/ Newspaper style • Visible headings – (e. g. sample size, outcomes, technologies) • Create white space – use paragraphs • Flow diagrams • PROOF-READ SUBMISSIONS CAREFULLY! www. nihr. ac. uk

Common Pitfalls • • Contradictory accounts of planned research Incorrect or inconsistent numbers e. g. sample size Awareness of major or related work in train Too many acronyms Insufficient detail, or muddled detail – esp. methods Gaps in expertise on the research team Not enough detail on: – The health issue – The intervention – The importance to evidence users www. nihr. ac. uk

Tips for applicants • Identify the problem clearly, review relevant literature • Identify all possible data sources, carry out preliminary research if necessary • Clearly define your research questions • Justify methodology • Project management essential • Early work with a CTU • Maintaining equipoise – clarity / non-bias www. nihr. ac. uk

Consider pathways to impact • Who are the main stakeholders • How can learning can be shared rapidly and effectively with them – Commissioners – Providers – Health care professionals – Patients and public • Consider working with stakeholders to develop your dissemination plan • Be creative and plan to target all audiences (not just publications - movie, website, social media ) www. nihr. ac. uk

Tips for success • Check application fits within the programme remit www. nets. nihr. ac. uk/faqs/remit & www. ccf. nihr. ac. uk • Check the existing portfolio of research http: //www. nihr. ac. uk/research-and-impact/nihr-clinical-researchnetwork-portfolio/ • Is it a logical proposal addressing clear evidence need • Research findings timely • Does it matter to decision makers and the public? • Demonstrated PPI and good quality plain language summary • Don’t leave it to the last minute www. nihr. ac. uk

Get help • Use web resources – Guidance notes, FAQs, remits Podcasts and resources for applicants http: //www. nets. nihr. ac. uk/funding www. ccf. nihr. ac. uk • Contact us – we’re friendly and helpful www. nihr. ac. uk

Get involved • Suggest future commissioned research topics http: //www. nets. nihr. ac. uk/identifying-research/make-asuggestion • Trainee reviewer development scheme • Professional and public contributors welcome - develop your skills and knowledge base http: //www. nets. nihr. ac. uk/become-a-reviewer/reviewer-form • Apply for Panel member/Board member roles - advertised regularly on our website • Join our mailing lists for programme and funding bulletins http: //www. nets. nihr. ac. uk/news/bulletins ; www. nihr. ac. uk

GET IN TOUCH WITH THE PROGRAMME’S SECRETARIAT Htaoas@southampton. ac. uk The HTA website: http: //www. nets. nihr. ac. uk/programmes/hta www. nihr. ac. uk

Any Questions? www. nihr. ac. uk