NIH Common Data Elements Repository Systems Demonstration Systems















- Slides: 17

NIH Common Data Elements Repository Systems Demonstration - Systems for Knowledge Management Session 87 Liz Amos, Vojtech Huser, Lisa Lang, and Christophe Ludet National Library of Medicine, National Institutes of Health Twitter: #AMIA 2017 or @NLM_news

Disclosures Authors have no financial conflicts to disclose. Amos, Huser and Lang are employed by the National Library of Medicine, National Institutes of Health and are supported by the Intramural Research Program at NLM. Ludet is an employee of Medical Science & Computing, LLC (MSC), and is a contractor to NLM; his work is supported by NLM and the Office of the Secretary (HHS) Patient-Centered Outcomes Research Trust Fund for Data Infrastructure. AMIA 2017 | amia. org 2

Policies to increase access to data from NIHfunded scientific research Notice of Proposed Rulemaking on Clinical Trial Registration and Results Submission Proposed NIH Policy on Dissemination of NIH-Funded Clinical Trial Information NIH Plan for Increasing Access to Scientific Publications and Digital Scientific Data from NIH Funded Scientific Research Genomic Sharing Data Policy https: //grants. nih. gov/policy/sharing. htm

Example: accessing clinical trials data and metadata • Clinical. Trials. gov contains information about medical studies in human volunteers • It is a registry and results database • 257, 336 registered trials as of November 7, 2017 • Of those, 28, 853 trials have reported with summary results • Many ways of collecting similar data, e. g. depression • Increasing granularity in outcome measurement reporting

A Common Data Element (CDE) is a data element common to multiple sets across different studies Fixed representation of a variable to be collected within a particular analytic of clinical domain Consists of a precisely defined question and a specified format or set of permissible values for responses (answers) Defined unambiguously in human and machine-computable terms Sets of CDEs can be combined into more complex questionnaires, survey instruments, and case report forms

Examples of Common Data Element Initiatives at NIH https: //www. nlm. nih. gov/cde/

NIH CDE Repository Promote the use of standards-based data elements and forms in support of electronic data capture and exchange for clinical research • Centralized library of data elements and standardized instruments recommended or required by NIH Institutes and Centers, and others • Facilitate use of machine-readable definitions through standardsbased export capabilities https: //cde. nlm. nih. gov/ AMIA 2017 | amia. org 7

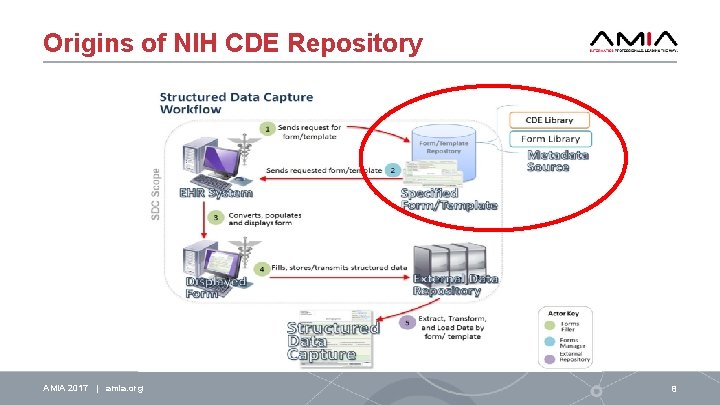
Origins of NIH CDE Repository AMIA 2017 | amia. org 8

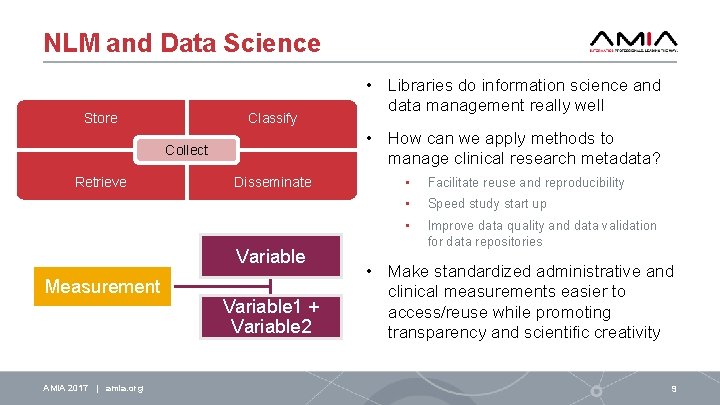
NLM and Data Science Store Classify • How can we apply methods to manage clinical research metadata? Collect Retrieve Disseminate Variable Measurement AMIA 2017 | amia. org • Libraries do information science and data management really well Variable 1 + Variable 2 • Facilitate reuse and reproducibility • Speed study start up • Improve data quality and data validation for data repositories • Make standardized administrative and clinical measurements easier to access/reuse while promoting transparency and scientific creativity 9

Key functionalities of the NIH Common Data Element Repository Develop new CDE and Form collections for clinical research Identify/discover CDEs and Forms recommended or required by NIH Institutes and Centers, and others Facilitate harmonization of data elements and forms, as well as specification using EHR-relevant health data standards Provide a platform for management and maintenance of CDE and Form collections Export authoritative versions of requested CDEs and Forms AMIA 2017 | amia. org 10

NIH CDE Repository features Today’s Use Case Demos: Today’s Highlighted Capabilities: NINR Symptom Science P 20 Centers Search recommended or required measures; Browse by domain • Find and reuse elements • Data elements and groupings • Collaborate with peers • Standardized assessment instruments, surveys, Case Report Forms • Draft and publish recommendations LASIK Quality of Life Collaboration Project (FDA, NEI, and DOD) • Create electronic form • Export Create and Collaborate Export for use in electronic data capture systems • JSON, XML/ODM, XML/SDC, REDCAP • Published form AMIA 2017 | amia. org 11

NINR Symptom Science NINR first developed a set of CDEs commonly collected in all symptom studies -- Sleep, Fatigue, Pain, and Cognitive/Affect -- regardless of diagnosis. Along with symptoms, also included are demographic information (e. g. , date of birth/ age, race, ethnicity); on participants throughout a study • Search • Create a Board • Compare two data elements, forms • Add comment and reviewer Required in specific P 20 FOAs (NINR) NINR Common Data Element Portal

LASIK Quality of Life Collaboration Project FDA, NEI and DOD project to better understand potential risk of severe problems that can result from LASIK. • Patient-Reported Outcomes with LASIK (PROWL) • • Create a CDE • Create a Form • Add a Section Pre-Op and Post-Op Questionnaires NLM asked to create CDEs and electronic form More information on scales used in PROWL Questionnaires LASIK Quality of Life Collaboration Project (FDA site)

Standards Terminology Export and Exchange • • JSON – NIH/CDE Schema • Structured Data Capture - XML with XSL transform • CDISC Operational Data Model – XML • Lister Hill National Center for Biomedical Communications (LHNCBC) Forms Widget • REDCAP zipped CSV LOINC – pull panels from LOINC as they are requested for collection • Newborn Screening • Social, psychological and behavioral observations - 2015 Edition Health IT Certification Criteria set • eye. GENE® (NEI) • Phen. X (hybrid LOINC/RTI curated) • SNOMED CT – annotation of data elements • Me. SH – NLM indexers evaluating use of Me. SH to aid discoverability AMIA 2017 | amia. org 14

Challenges, Opportunities and Future Directions • Curation, duplication, quality assurance and harmonization • • Siloed development/initial curation of metadata Proliferation of many standards • Copyright analysis and display • • Environmental scan of standardized assessment instruments to determine copyright status/terms of use Development of workflow for contacting copyright holder, with intention to represent in Repository • Continued development of service model to support various use cases and stakeholders with limited resources • Annotation(s) of data elements and forms to support discoverability • • SNOMED CT (Poster Session 11/7 @ 5 pm) Me. SH (collaboration with NLM Index Section) • Move to Amazon Web Services AMIA 2017 | amia. org 15

Challenges, Opportunities and Future Directions (Continued) • Enhanced collaboration with other public repositories (both data and data element/form) • Enhanced discovery and authoring capabilities to support measure and registry authoring projects, including enhanced coordination with the NLM Value Set Authority Center (VSAC), particularly for authoring using EHRrelevant health data standards • FHIR (SMART on FHIR) AMIA 2017 | amia. org 16

Thank you! cde. nlm. nih. gov