Niels Bohr 1885 1962 Nobel Prize 1922 l

Niels Bohr (1885 -1962) (Nobel Prize, 1922) l Based his studies on Hydrogen l Proposed energy levels l Planetary model of the Atom l We still use this today to estimate place of electron
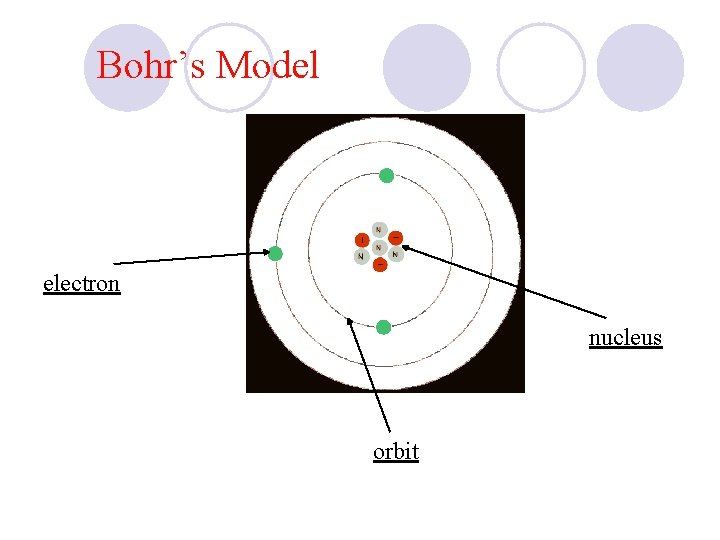
Bohr’s Model electron nucleus orbit

Bohr Analogy üAn Onion If you cut an onion in half you’ll see rings. Bohr’s model has “rings” called energy levels in which the electrons are located outside of the nucleus.
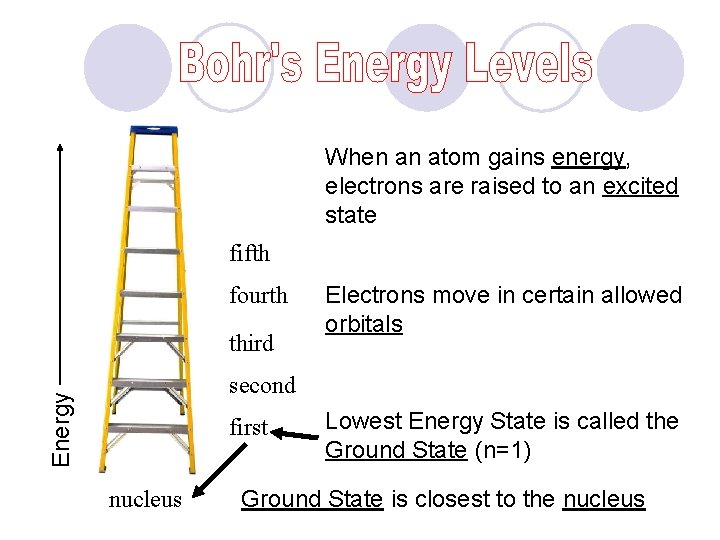
When an atom gains energy, electrons are raised to an excited state fifth fourth third Electrons move in certain allowed orbitals Energy second first nucleus Lowest Energy State is called the Ground State (n=1) Ground State is closest to the nucleus

Bohr to quanta Bohr’s theory was a great accomplishment and radically changed our view of matter. But problems existed with Bohr theory — ¡The theory only successful for the H atom. l So, we go on to Quantum Mechanical Model of the Atom was developed

Quantization of Energy was started by Max Planck l Planck’s hypothesis: Matter can only gain or lose energy in small specific amounts called a quantum. (1858 -1947)

Louis de Broglie 1892 -1987 l Proposed the wave particle duality of nature l Wave Particle duality of nature says Waves can act like particles or waves l Compared electron orbitals to waves l Related Planck’s quantum idea to electron orbitals to state that orbitals must be in WHOLE numbers.

Werner Heisenberg l Proposed the Uncertainty Principle ¡ Said it is impossible to know both the exact position and velocity of an electron at the same time Make a prediction but not for sure Einstein did not like it. He said “God Doesn’t Play Dice”

Erwin Schrodinger l Used de Broglie and Planck’s work to propose a new model of the atom, in which electrons are treated as waves l Described three dimension area around nucleus called the atomic orbital to describe the probability of the electrons’ location l Electron cloud model of an atom 1887 -1961
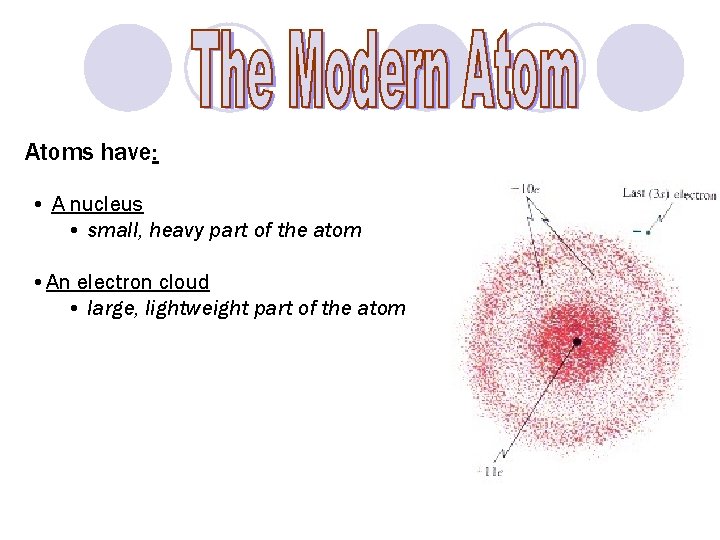
Atoms have: • A nucleus • small, heavy part of the atom • An electron cloud • large, lightweight part of the atom Nucleus Electron Cloud

Don’t forget tonight’s homework: Update the Scientist Timeline
- Slides: 11