NIE for Strong vs Weak Acids HCl Na
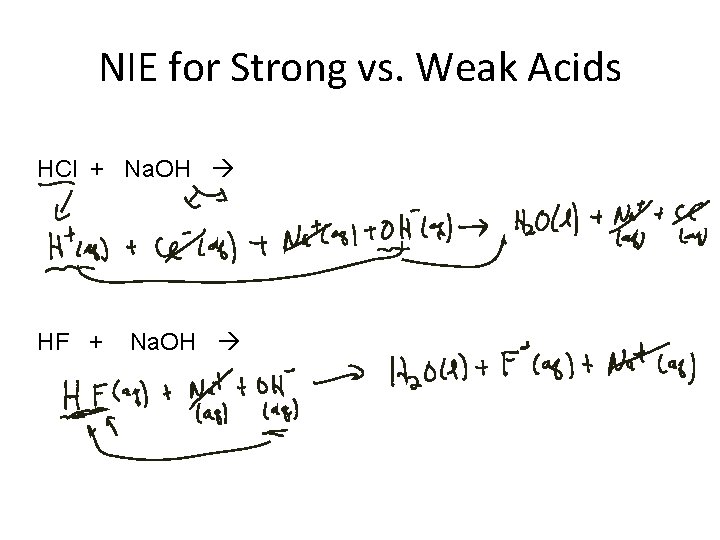





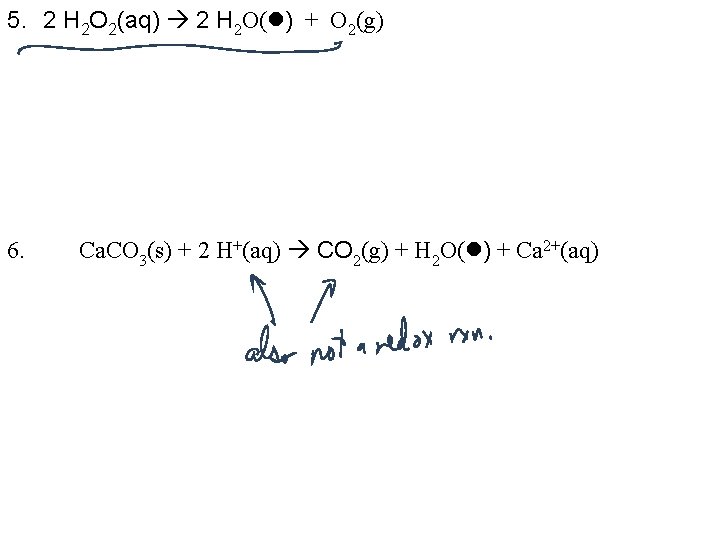

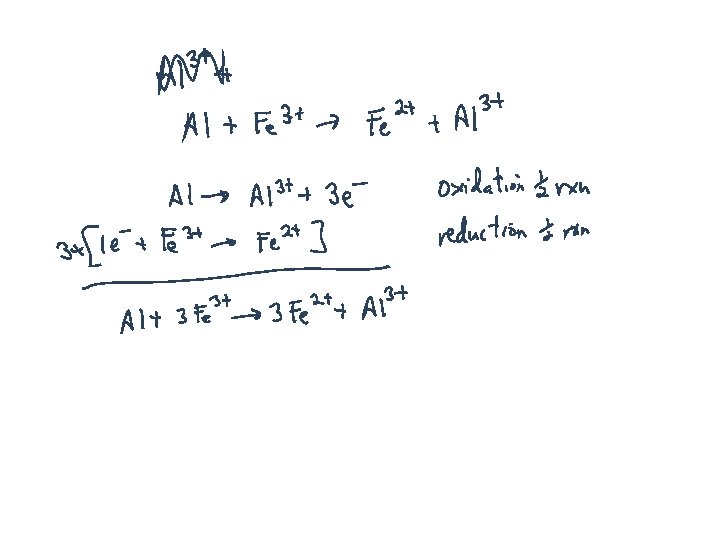


- Slides: 21
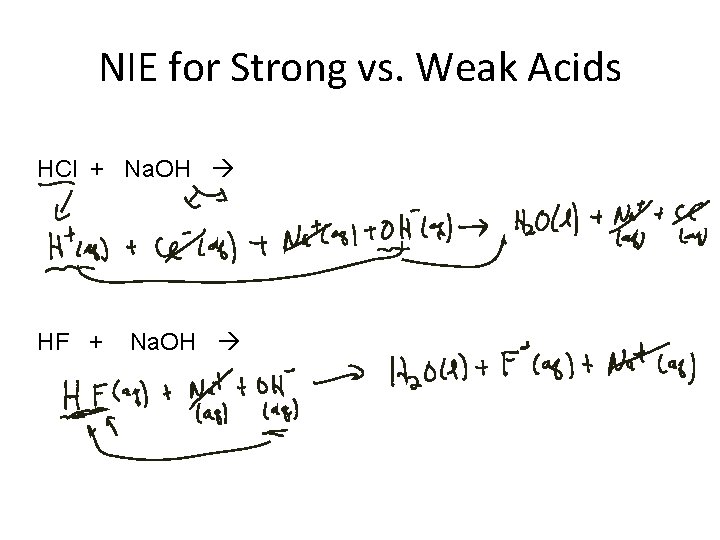
NIE for Strong vs. Weak Acids HCl + Na. OH HF + Na. OH

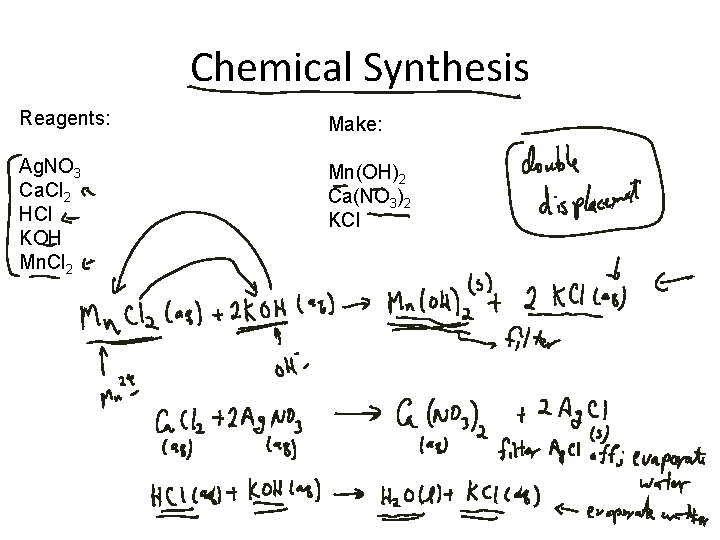
Chemical Synthesis Reagents: Make: Ag. NO 3 Ca. Cl 2 HCl KOH Mn. Cl 2 Mn(OH)2 Ca(NO 3)2 KCl

Oxidation Reduction Reactions Redox Reactions Electron-Transfer Reactions


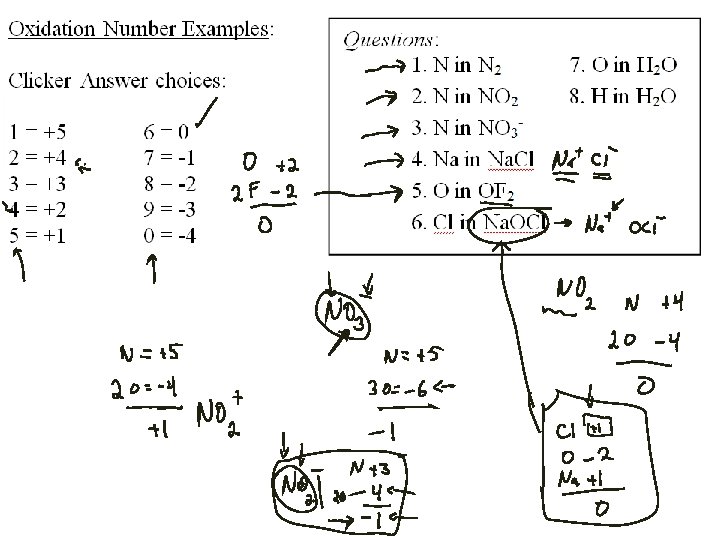
Oxidation Numbers: A set of rules for tracking charges for atoms in covalent compounds 1. Each atom in a pure element has an oxidation number of zero. 2. For monatomic ions, the oxidation number is equal to the charge on the ion. 3. Fluorine always has an oxidation number of – 1 in compounds with all other elements. 4. Cl, Br, and I always have oxidation numbers of – 1 in compounds, except when combined with oxygen or fluorine. 5. The oxidation number of H is +1 and of O is – 2 in most compounds. • Exception for H: In compounds with metals, H is – 1. • Exception for O: In peroxides, O 22 - ion gives O an oxidation number of – 1. 6. The algebraic sum of the oxidation numbers for the atoms in a neutral compound must be zero; in a polyatomic ion, the sum must be equal to the ion charge.


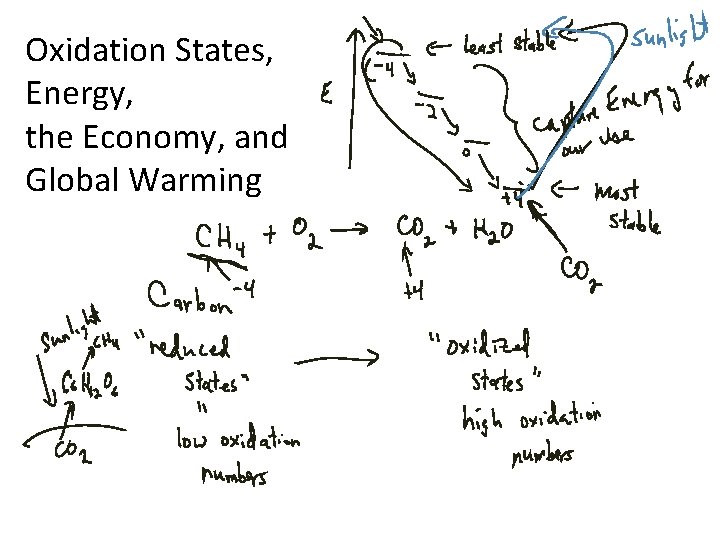
Oxidation States, Energy, the Economy, and Global Warming

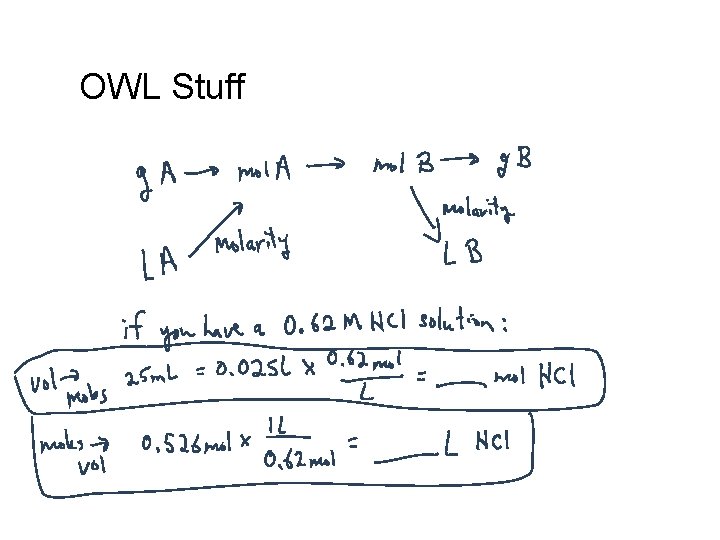
OWL Stuff

Oxidation-Reduction Reactions Oxidation = loss of electrons = increase in ox # Reduction = gain of electrons = decrease in ox # 4 Fe + 3 O 2 2 Fe 2 O 3 Oxidizing agent = oxidant = gains electrons Reducing agent = reductant = loses electrons


Recognizing Redox Reactions: Always involves changes in oxidation numbers Metals reacting with Nonmetals: 2 Al + 3 Br 2 Al 2 Br 6 Almost anything with Oxygen: 2 Mg + O 2 2 Mg. O Organic Compounds: CH 4 + 2 O 2 CO 2 + H 2 O Others, more difficult to detect: 5 Fe 2+ + Mn. O 4 - + 8 H+ 5 Fe 3+ + Mn 2+ + 4 H 2 O S + O 2 SO 2

1. 4 Al(s) + 3 O 2(g) 2 Al 2 O 3(s) 2. Cu. O(s) + H 2(g) Cu(s) + H 2 O( )

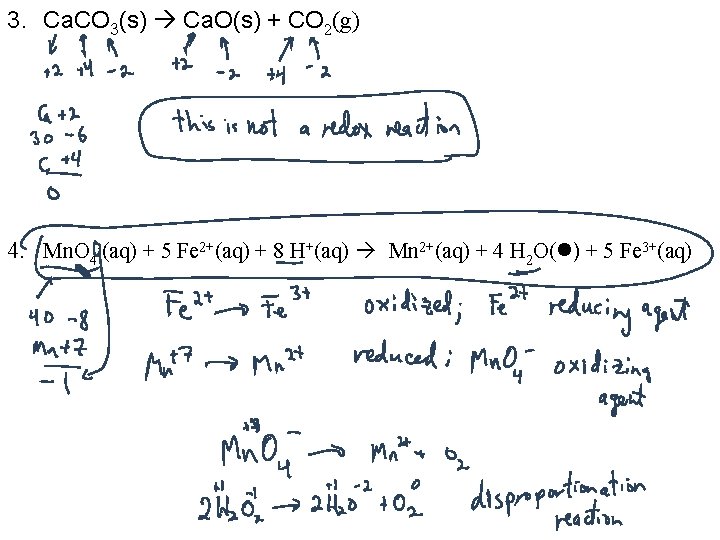
3. Ca. CO 3(s) Ca. O(s) + CO 2(g) 4. Mn. O 4 -(aq) + 5 Fe 2+(aq) + 8 H+(aq) Mn 2+(aq) + 4 H 2 O( ) + 5 Fe 3+(aq)
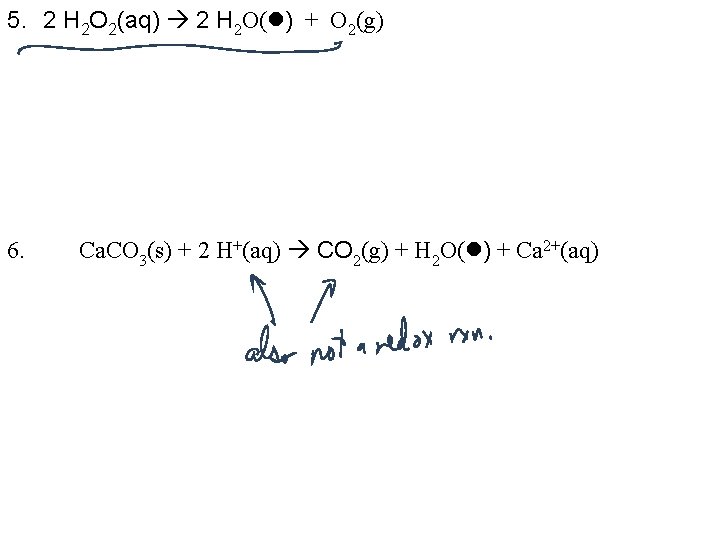
5. 2 H 2 O 2(aq) 2 H 2 O( ) + O 2(g) 6. Ca. CO 3(s) + 2 H+(aq) CO 2(g) + H 2 O( ) + Ca 2+(aq)

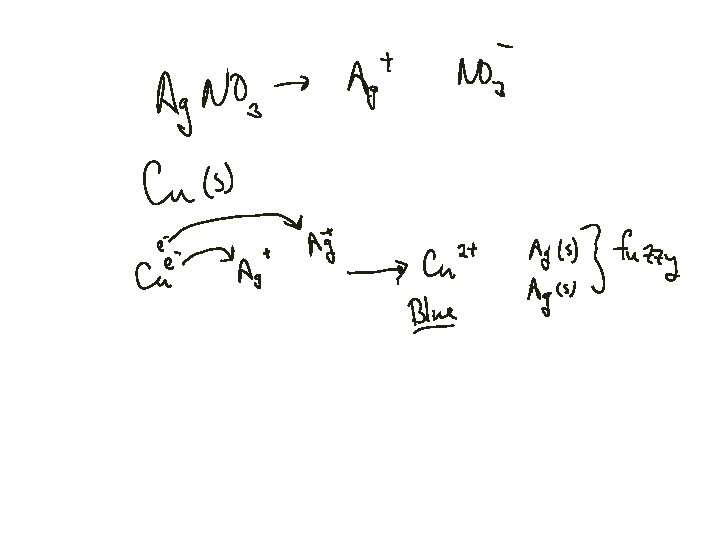
Balancing Redox Reactions Cu(s) + Ag+(aq) Cu 2+(aq) + Use Half-Reactions Ag(s)
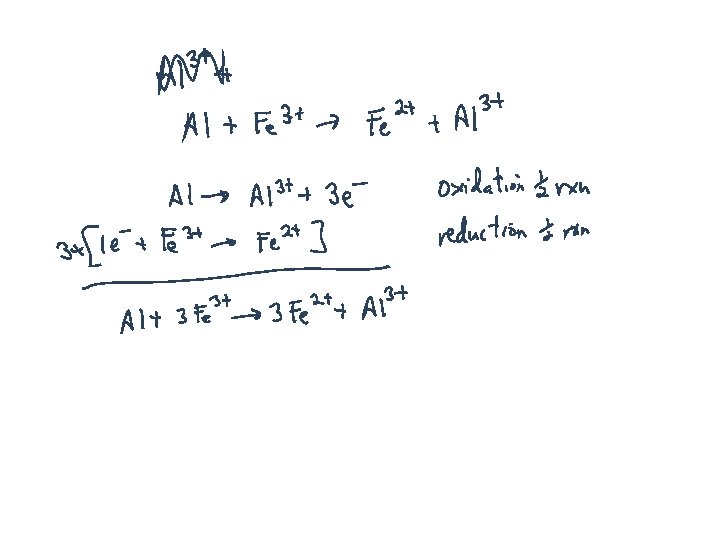


Combination and Decomposition Reactions Thermal decomposition of metal carbonates: MCO 3(s) MO(s) + CO 2(g) Combination of two elements to form a compound Zn + S Zn. S

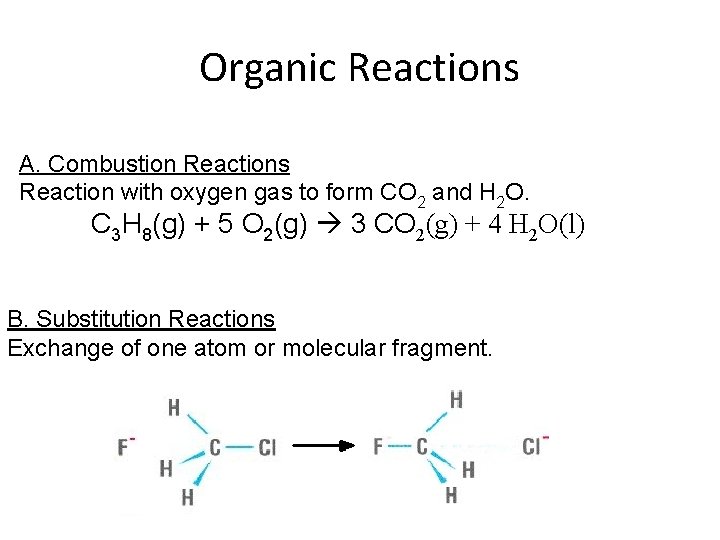
Organic Reactions A. Combustion Reactions Reaction with oxygen gas to form CO 2 and H 2 O. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) B. Substitution Reactions Exchange of one atom or molecular fragment.

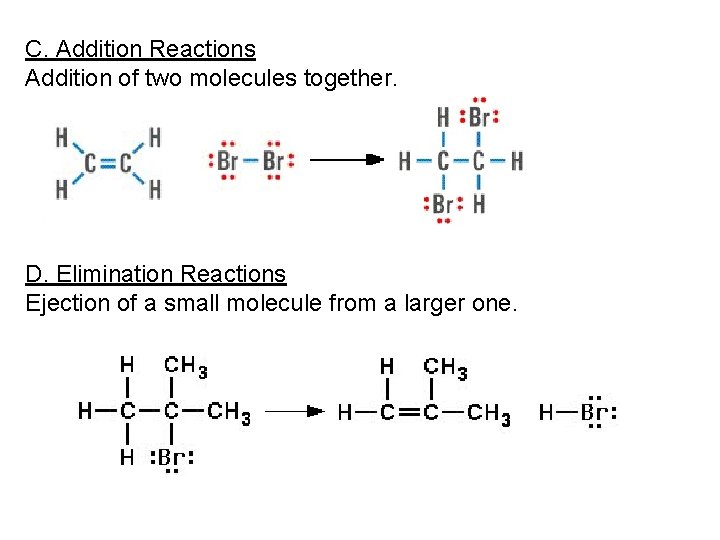
C. Addition Reactions Addition of two molecules together. D. Elimination Reactions Ejection of a small molecule from a larger one.

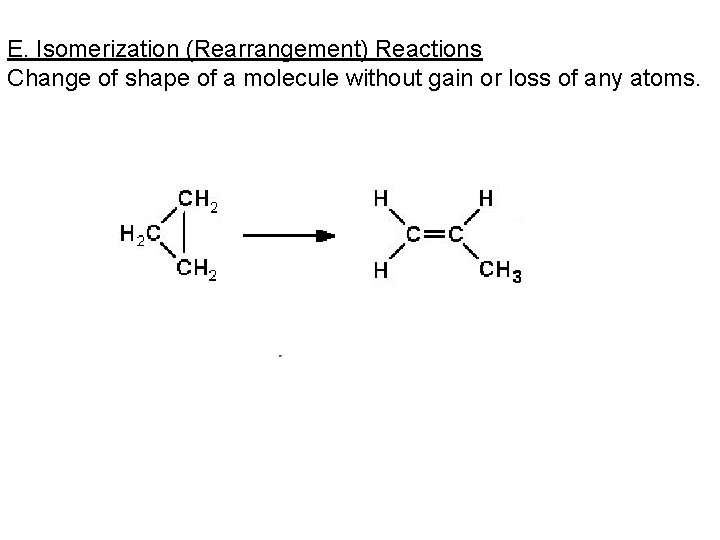
E. Isomerization (Rearrangement) Reactions Change of shape of a molecule without gain or loss of any atoms.