Nickel Institute Corrosion by Process Waters R W

Nickel Institute

Corrosion by Process Waters R. W. Ross Consultant Nickel Institute

Summary h Scaling and Corrosion h Effects of Velocity h Biological Effects h Chlorides h Rouging of SS

Water Chemistry Effects Corrosion • Dissolved Oxygen • Chlorides • p. H • Hardness • Temperature Scale • Dissolved Solids • Calcium Ions • p. H • Temperature

Water Chemistry Effects
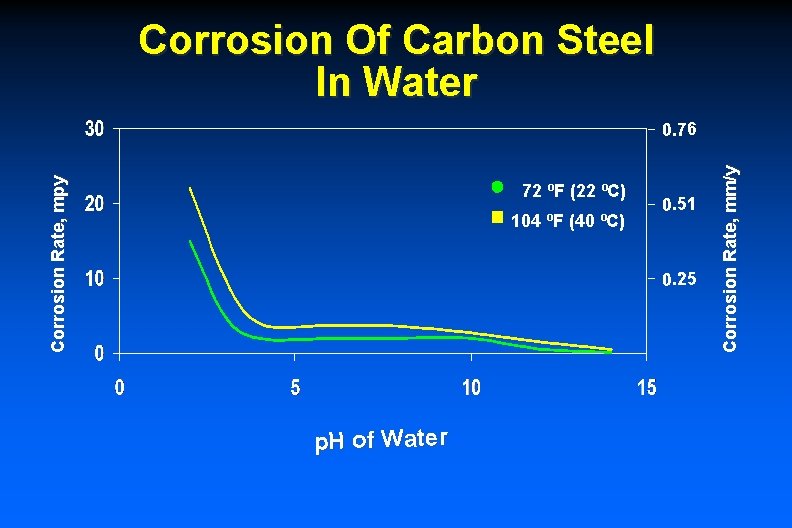
Corrosion Of Carbon Steel In Water 72 ºF (22 ºC) 104 ºF (40 ºC) 0. 51 0. 25 p. H of Water Corrosion Rate, mm/y Corrosion Rate, mpy 0. 76

Corrosion Of Carbon Steel In Low-velocity Water
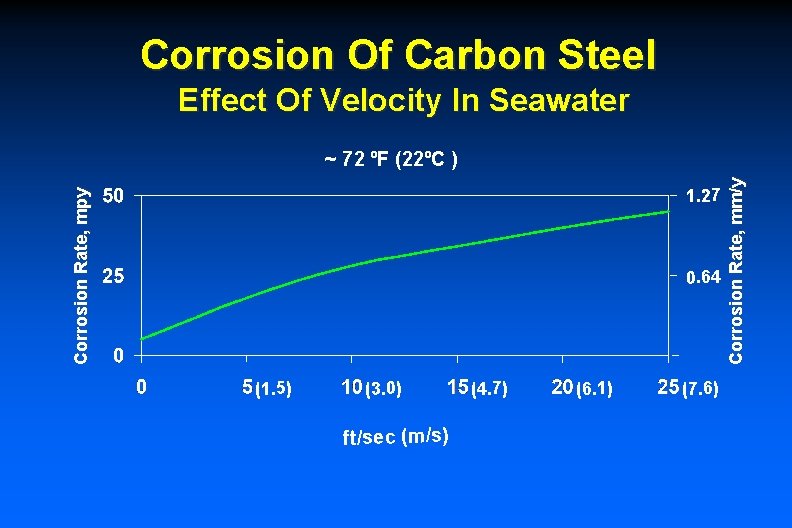
Corrosion Of Carbon Steel Effect Of Velocity In Seawater Corrosion Rate, mpy 1. 27 0. 64 (1. 5) (3. 0) ft/sec (m/s) (4. 7) (6. 1) (7. 6) Corrosion Rate, mm/y ~ 72 ºF (22ºC )

Erosion-corrosion - Inlet
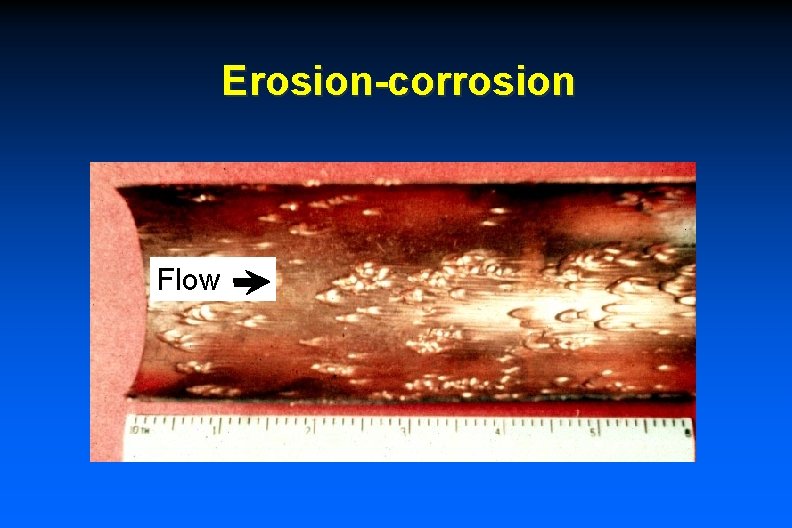
Erosion-corrosion Flow

Erosion-corrosion Tube Blockage Flow
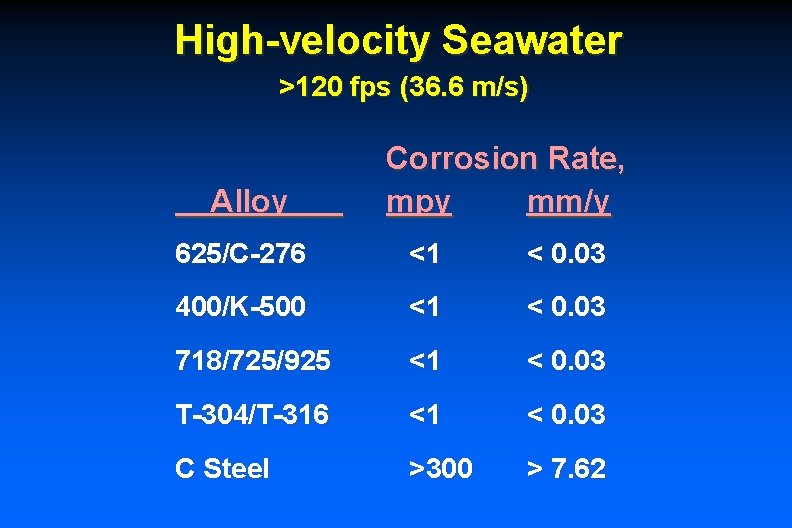
High-velocity Seawater >120 fps (36. 6 m/s) Alloy Corrosion Rate, mpy mm/y 625/C-276 <1 < 0. 03 400/K-500 <1 < 0. 03 718/725/925 <1 < 0. 03 T-304/T-316 <1 < 0. 03 C Steel >300 > 7. 62

Biological Effects Macrofouling • Mussels • Clams • Barnacles • Plant Life

Biological Effects Macrofouling

Bacteria Effects - MIC (Microbiologically Induced Corrosion) Species Oxygen Metals Corrosive Desulfovibrio No Fe, Al, Cu Sulfide Thiobacillus Yes Fe, Cu Sulfuric Acid Gallionella Yes Fe Fe++ to Fe+++ Mn++ to Mn+++
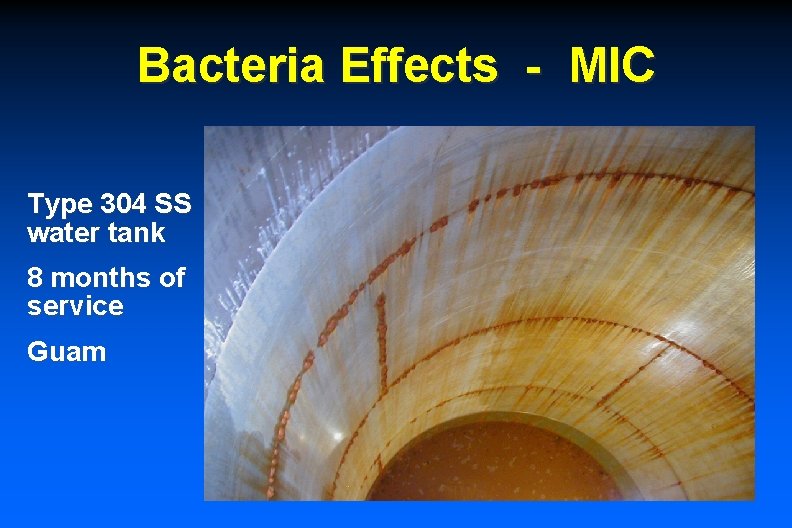
Bacteria Effects - MIC Type 304 SS water tank 8 months of service Guam
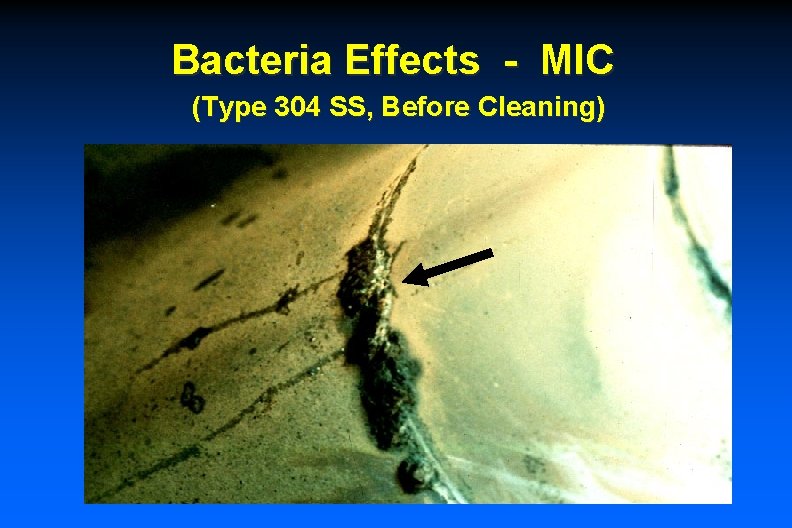
Bacteria Effects - MIC (Type 304 SS, Before Cleaning)

Bacteria Effects - MIC (After Cleaning) 0. 15 in. (3. 8) mm) Max. Attack

Bacteria Effects - MIC (After Cleaning - No Attack)

Prevention Of MIC • Keep The System Clean • Keep Water Flow > 6 fps (2 m/s) • Use Bactericide: – Chlorine Dioxide – Hypochlorite – Ozone – Non-oxidizing

Prevention Of MIC • Use Continuous Cleaning • Use High Pressure Hydrolancing • Use Stainless Steel Scrapers (Hard to Remove or Heavy Deposits) • Use Alloy Resistant to MIC

Prevention Of MIC - 6% Mo ALLOY

Effects of Chlorides

Crevice Corrosion Type 303
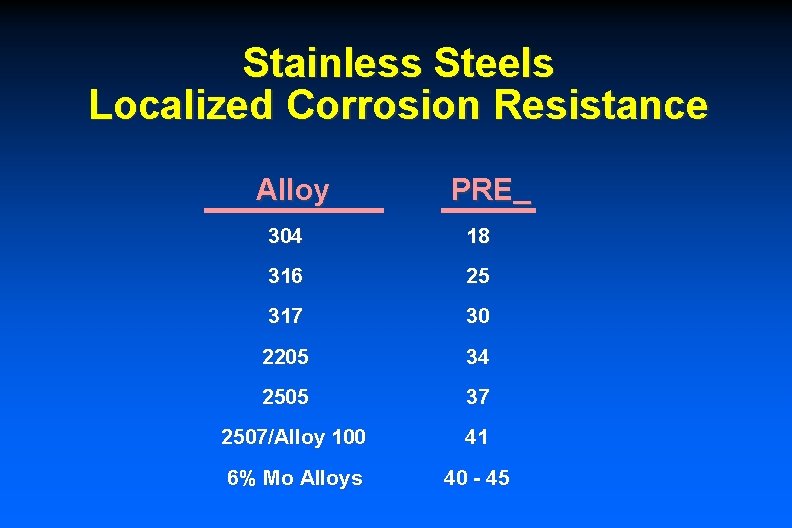
Stainless Steels Localized Corrosion Resistance Alloy PRE 304 18 316 25 317 30 2205 34 2505 37 2507/Alloy 100 41 6% Mo Alloys 40 - 45
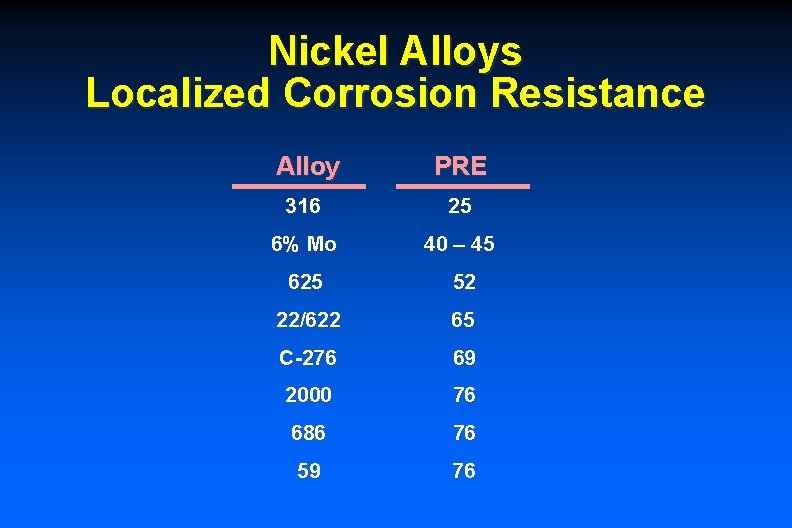
Nickel Alloys Localized Corrosion Resistance Alloy PRE 316 25 6% Mo 40 – 45 625 52 22/622 65 C-276 69 2000 76 686 76 59 76

Stainless Steels for Use in Waters Potable water Type 304 < 200 ppm chlorides Type 316 < 1000 ppm chlorides River water Risk of MIC if water is not treated Use type 316 or higher Mo grades: 2205 904 L 2507 6 Mo Well water Risk of MIC if water is not treated Use type 316 or higher Mo grades

Do not confuse Chloride Cl- and Chlorine Cl 2 Maximum Concentration (ppm) in Water to Avoid Crevice Corrosion Chloride Cl- Chlorine Cl 2 304 200 2 316 1000 4 Shock dosing, such as 25 ppm chlorine for 24 hours, is common practice and has not been found to cause problems.

Stress Corrosion Cracking (SCC) S m a te e n i L

Chloride SCC Duplex vs T-316 Stainless Steel No cracking below lines (315) Type 316 22 Cr Duplex 18 Cr Duplex (204) (93)

Effect of Nickel Content on Stress Corrosion Cracking Boiling 45% Mg. Cl 2 No SCC Ni Alloys Duplex SS SCC 6% Mo SS

HIGH CHLORIDE WATERS

HIGH CHLORIDE WATERS How does external environment affect process equipment?

Marine Corrosion of C Steel Relative Corrosion Rates* – Vary with Sea Conditions Atmospheric Splash *Protected Harbor 25 mpy (0. 64 mm/y) Tidal Submerged Subsoil 5 mpy (0. 13 mm/y)

Uniform Corrosion
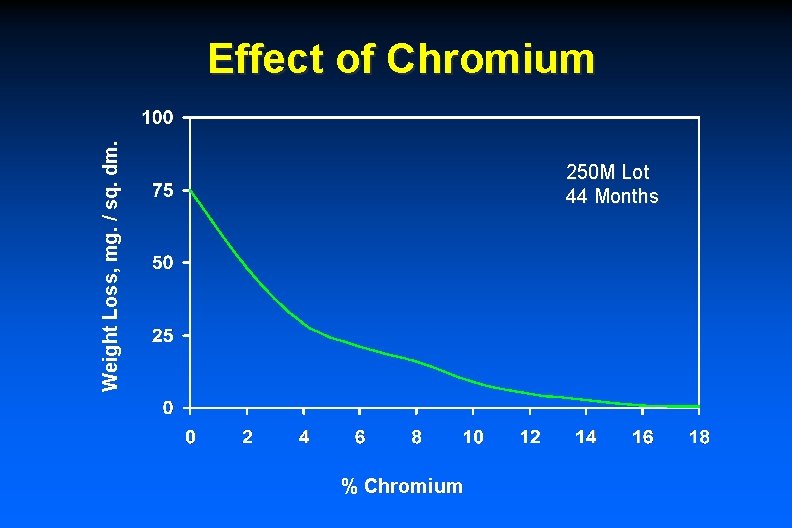
Weight Loss, mg. / sq. dm. Effect of Chromium 250 M Lot 44 Months % Chromium

Alloy C in Marine Atmosphere 56 Years of Exposure


Type 304 Fastener In Marine Tide After 6 Months

Type 304 Fastener Above Marine Tide After 6 Months

Crevice Corrosion

Crevice Corrosion of Alloy 825 Heat Exchanger Tubing – Shell Side 85° F, Aerated Seawater

Crevice Corrosion of Alloy 625 Waterbox With Deaerated, Treated Seawater 165° F 235° F 165 -235° F
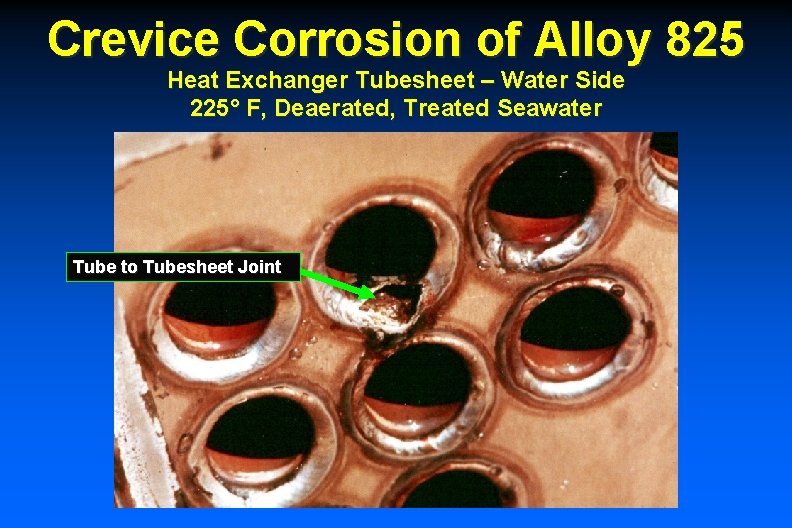
Crevice Corrosion of Alloy 825 Heat Exchanger Tubesheet – Water Side 225° F, Deaerated, Treated Seawater Tube to Tubesheet Joint
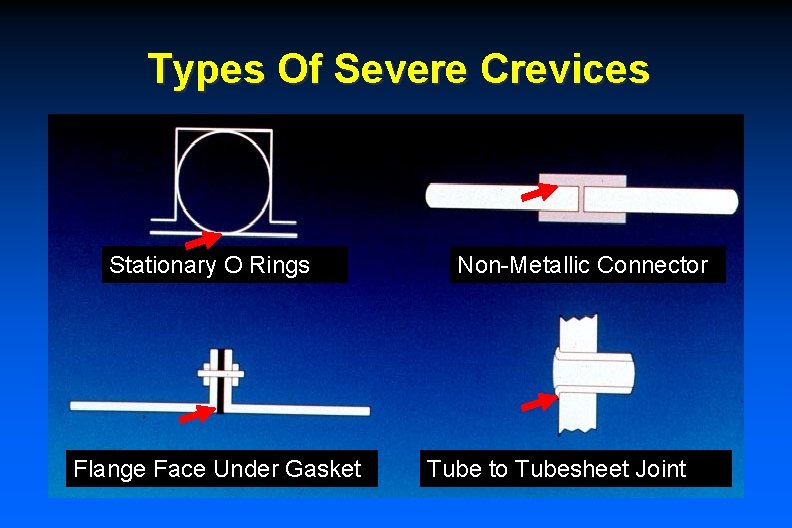
Types Of Severe Crevices Stationary O Rings Flange Face Under Gasket Non-Metallic Connector Tube to Tubesheet Joint
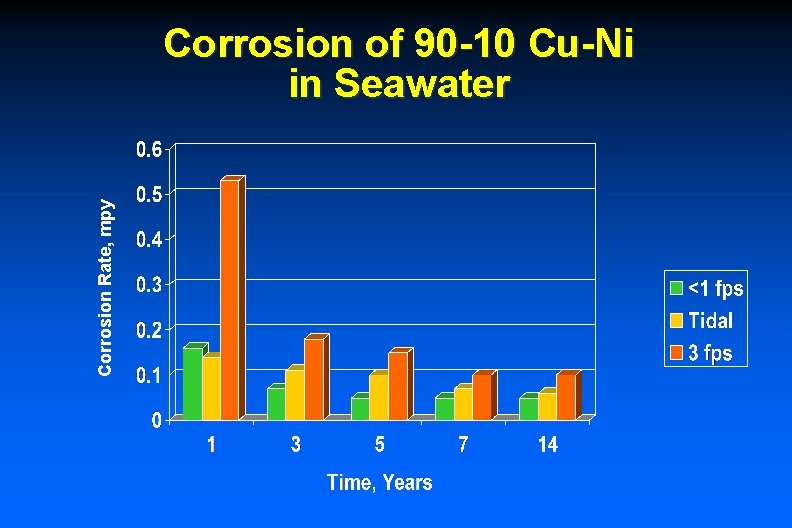
Corrosion Rate, mpy Corrosion of 90 -10 Cu-Ni in Seawater

Marine Fouling 18 Months in Quiet Seawater C Steel Aluminum

Fouling of Titanium Waterbox 3 mo. Exposure

Fouling of Titanium Waterbox 6 mo. Exposure
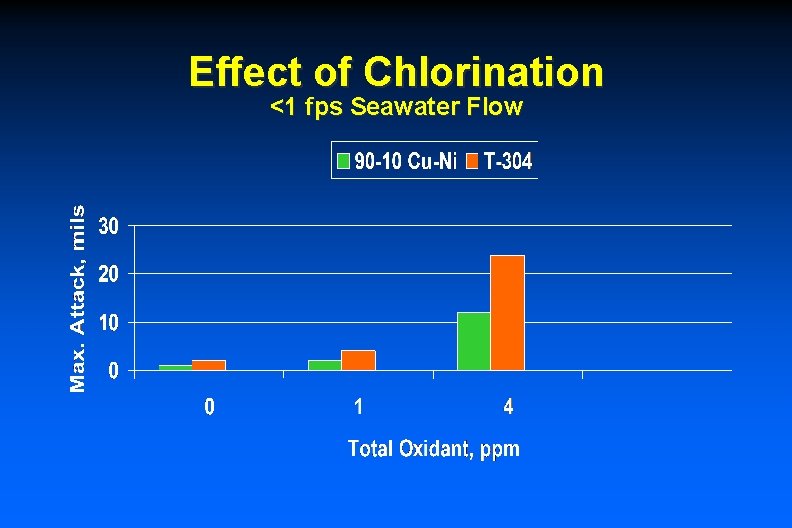
Effect of Chlorination <1 fps Seawater Flow

90 -10 Cu-Ni Alloy Fouling - Quiet Seawater 3 Mo 9 Mo 3 Yr 4 Yr 18 Mo 5 Yr

90 -10 Cu-Ni Intake Piping Desalination Plants

90 -10 Cu-Ni Alloy Seawater Piping Systems

90 -10 Cu-Ni Alloy Seawater Heat Exchangers

Pumps - Impellers

Rouging of Stainless Steels High Purity Water For Injection (WFI)

Why Use Stainless Steels (316 L) for Pharma & Biotech? Good corrosion resistance and excellent batch to batch cleanability Good structural properties for process equipment Easily formed, fabricated and welded

What about Rouging? What is Rouging? Rouging is a general term used to describe several species of predominately iron oxide deposits on the wall of piping and vessels in high purity water systems.

Rouging is not New! Rouging is not unique to the pharmaceutical and biotech industries. Was recognized over 40 years ago with rouging of SS vessels at Savannah River.

Where is Rouge often Found Water systems, usually high purity water and clean-steam systems Distillation and clean-steam generating equipment Rouge found on wall of vessels, piping and polymer gaskets (Teflon®) downstream of where originated

Is Rouge Harmful? No reports or evidence that rouging is precursor to a SS corrosion failure. We are not in a position to comment on whether rouge is harmful to the product being produced. Common practice is to remove rouge.

Rouging Generally a loose powdery deposit, but can be tightly adherent Hydrated or partially hydrated ferric oxide (Fe 2 O 3) or ferroso-ferric oxide (Fe 3 O 4) Usually occurs in high purity (0. 5 -1. 0 µS/cm), high temperature water (60 – 100 C)

Rouging Reddish brown rust color, but can range from orange to blue-black. Origin is uncertain but generally thought to be ions or colloids that are formed at one location and transported in the solution to another where they are precipitated. Removed by acid cleaning in nitric, phosphoric, citric, or oxalic acid.

Rouging - Types Type 1 – Corrosion of Steel, Deposits Downstream Pumps prime suspects – cavitation or erosion when velocity over ~ 100 fps and higher temperatures Delta ferrite in cast impellers may contribute by eroding easier and higher iron content Type 2 – Corrosion Product of Stainless Steel Type 3 – Corrosion Product of Stainless Steel in Steam Systems > 100 C

Rouging of Stainless Steels

Rouging over 4 years inside electropolished Type 316 L - column still used to produce ultrapure water for pharmaceutical use

Rouging of Stainless Steels

De-rouging & Passivation 3 Steps Cleaning – detergent wash followed by thorough water rinse De-rouging chemical treatment Passivation followed by thorough water rinse

Electrochemical Coloring Proprietary electrochemical processes – invented in 1972 by Inco, further developed in Japan • Interference between the light beams refracted from the substrate and the surface of the oxide film creates color • Appearance and color vary with immersion time and surface finish Incident light Color Oxide

Experience Music Project

Summary h Discussed Scaling and Corrosion h Described Effects of Velocity h Reviewed Biological Effects h Discussed Chlorides h Summarized Rouging of SS

Questions ?
- Slides: 72