NGIS Data Quality Report Guidance Version Number v

NGIS Data Quality Report Guidance Version Number: v 1. 0

NGIS DQ Report purpose The DQ Report spreadsheet provides: • information relating to data issues submitted via TOMS that affect the submission of the case to bioinformatics • data issue warnings about the test order • discrepancies between clinical data submitted via the test request and sample data submitted via CSV • Information on test orders where sample data submitted via CSV has not been received It does not contain information on whether the CSV submission has passed the validation rules outlined in the sample specification. This is shown in the MI portal in real time. v 1. 0 15 Nov 2019 2

NGIS DQ Reporting Output An Excel spreadsheet is produced and sent 3 x per day. It will contain four tabs: • Summary: provides a count of each of the rules that have failed for the organisation. • Full output: a full listing of all failures for each test order, complete with all the patient’s identifiers to aid access to the test order. • Streamline Output: a listing of failures that can be actioned immediately. • Appendix – all rules: information about all the current active rules in the DQ Report. v 1. 0 15 Nov 2019 3
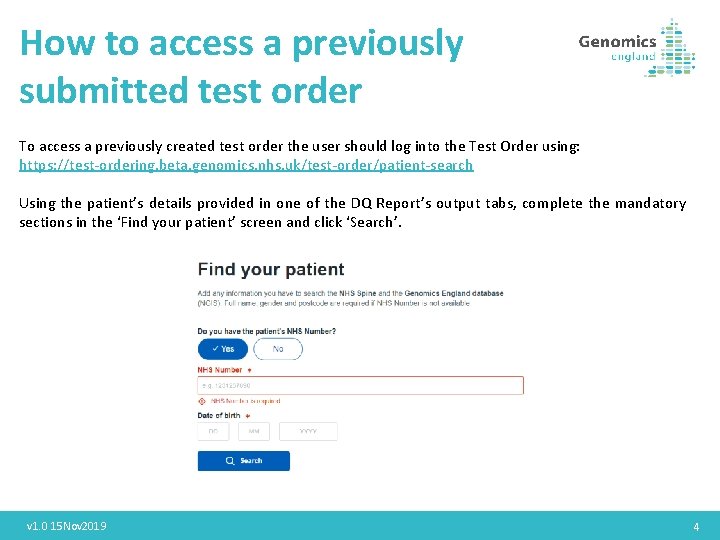
How to access a previously submitted test order To access a previously created test order the user should log into the Test Order using: https: //test-ordering. beta. genomics. nhs. uk/test-order/patient-search Using the patient’s details provided in one of the DQ Report’s output tabs, complete the mandatory sections in the ‘Find your patient’ screen and click ‘Search’. v 1. 0 15 Nov 2019 4
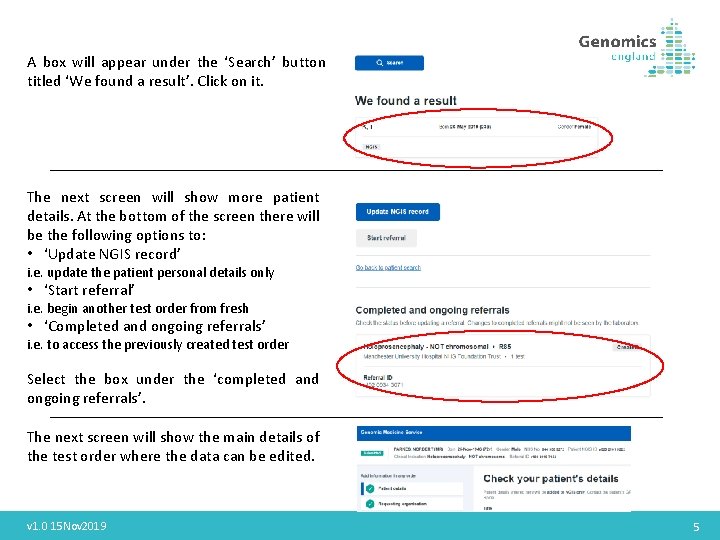
A box will appear under the ‘Search’ button titled ‘We found a result’. Click on it. The next screen will show more patient details. At the bottom of the screen there will be the following options to: • ‘Update NGIS record’ i. e. update the patient personal details only • ‘Start referral’ i. e. begin another test order from fresh • ‘Completed and ongoing referrals’ i. e. to access the previously created test order Select the box under the ‘completed and ongoing referrals’. The next screen will show the main details of the test order where the data can be edited. v 1. 0 15 Nov 2019 5

Guidance for resolving rule failures The detail of Rule ID, rule description and resolution guidance for each rule follows. Please note: If the original data submitted is correct, please inform Service Desk to mute this row from future DQ reports. v 1. 0 15 Nov 2019 6
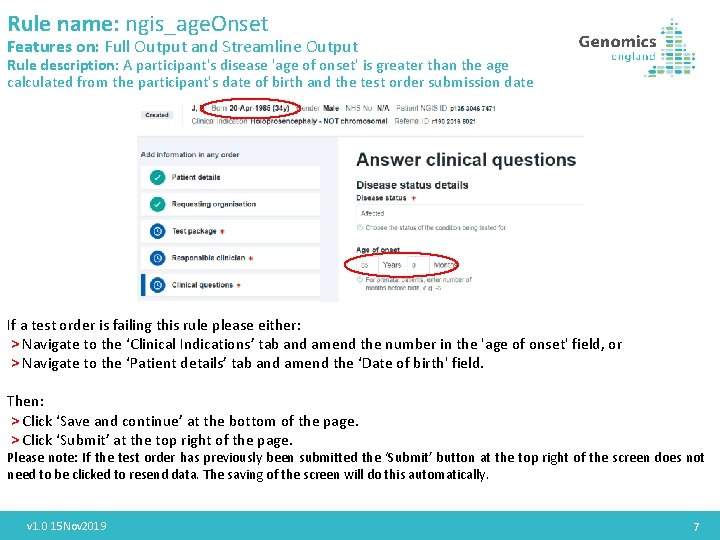
Rule name: ngis_age. Onset Features on: Full Output and Streamline Output Rule description: A participant's disease 'age of onset' is greater than the age calculated from the participant's date of birth and the test order submission date If a test order is failing this rule please either: > Navigate to the ‘Clinical Indications’ tab and amend the number in the 'age of onset' field, or > Navigate to the ‘Patient details’ tab and amend the ‘Date of birth' field. Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 7
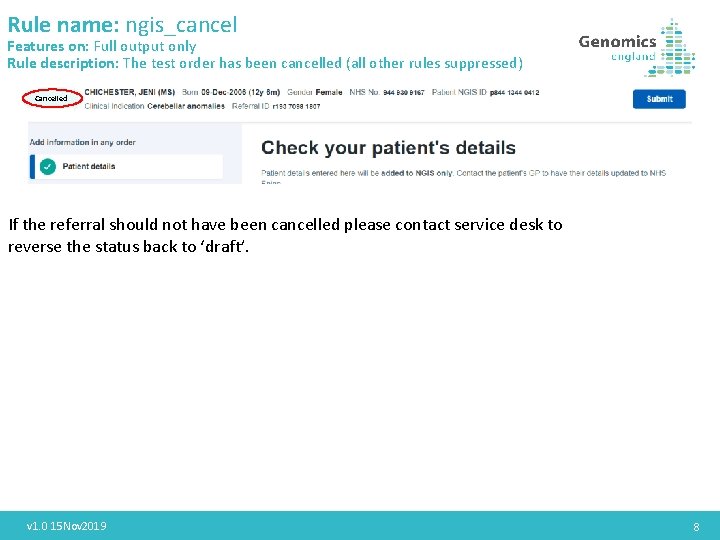
Rule name: ngis_cancel Features on: Full output only Rule description: The test order has been cancelled (all other rules suppressed) Cancelled If the referral should not have been cancelled please contact service desk to reverse the status back to ‘draft’. v 1. 0 15 Nov 2019 8

Rule name: ngis_col. Date; ngis_morph; ngis_sam. State; ngis_sam. Type; ngis_topo Features on: Both outputs Rule description: Sample data provided in TOMS does not match sample data provided in GEL 1001 (only if both the fields have been provided) If a test order fails one of these rules, please either: > Amend the data in TOMS by navigating to the ‘Samples’, selecting the appropriate sample by pressing the “>” button, then select ‘Save and continue’ until the page with the data point in question is presented. > Enter the correct data into the appropriate field. > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically OR > Amend the data in GEL 1001 and resubmitting to the SFTP folder. v 1. 0 15 Nov 2019 9

Rule name: ngis_dob Features on: Both outputs Rule description: The proband's date of birth is less than 12 years later than that of their parents If a test order is failing this rule please either: > Navigate to the ‘Family members’ tab to amend the ‘Date of birth' field for the parents, or > Navigate to the ‘Patient details’ tab to amend the ‘Date of birth' field for the proband, or > Ensure the correct patient has been added to the test order. - Family members can be removed by selecting the dustbin icon - Probands cannot be removed. Contact the service desk to cancel the test order and begin a new test order. Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 10

Rule name: ngis_germline Features on: Full Output only Rule description: Germline sample data has not been submitted via the GEL 1001 csv file for every participant in the test order If a test order is failing this rule please: > Submit a GEL 1001 via SFTP, ensuring that there is a germline record for each of the test order’s participants. Rule name: ngis_germline 2 Features on: Streamline Output only Rule description: Germline sample data has not been submitted via the GEL 1001 csv file for every participant in the test order, despite there being a submission of a valid GEL 1001 file from the GLH since the test order submission date. If a test order is failing this rule please: > Submit a GEL 1001 via SFTP, ensuring that there is a germline record for each of the test order’s participants. v 1. 0 15 Nov 2019 11
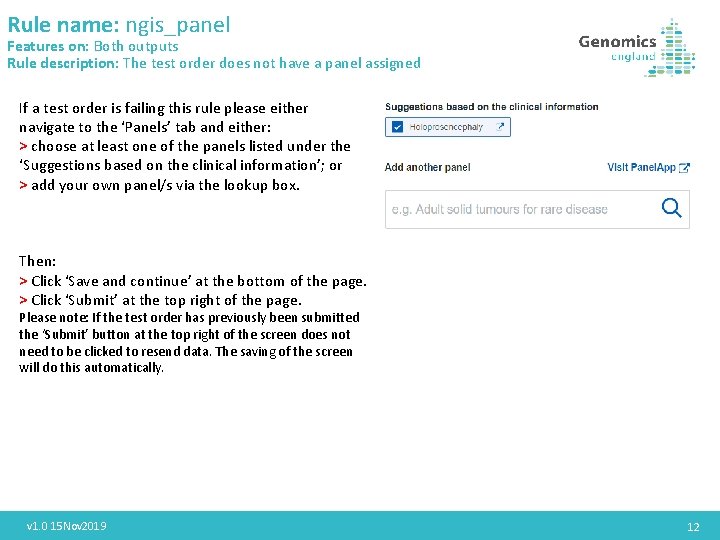
Rule name: ngis_panel Features on: Both outputs Rule description: The test order does not have a panel assigned If a test order is failing this rule please either navigate to the ‘Panels’ tab and either: > choose at least one of the panels listed under the ‘Suggestions based on the clinical information’; or > add your own panel/s via the lookup box. Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 12
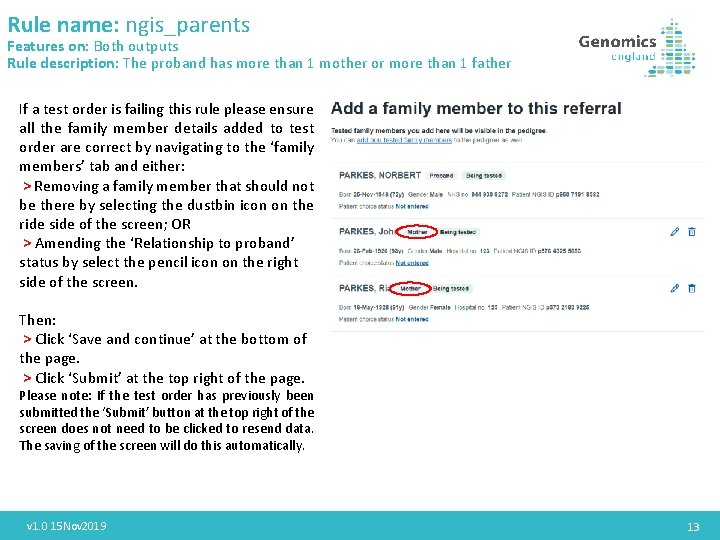
Rule name: ngis_parents Features on: Both outputs Rule description: The proband has more than 1 mother or more than 1 father If a test order is failing this rule please ensure all the family member details added to test order are correct by navigating to the ‘family members’ tab and either: > Removing a family member that should not be there by selecting the dustbin icon on the ride side of the screen; OR > Amending the ‘Relationship to proband’ status by select the pencil icon on the right side of the screen. Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 13
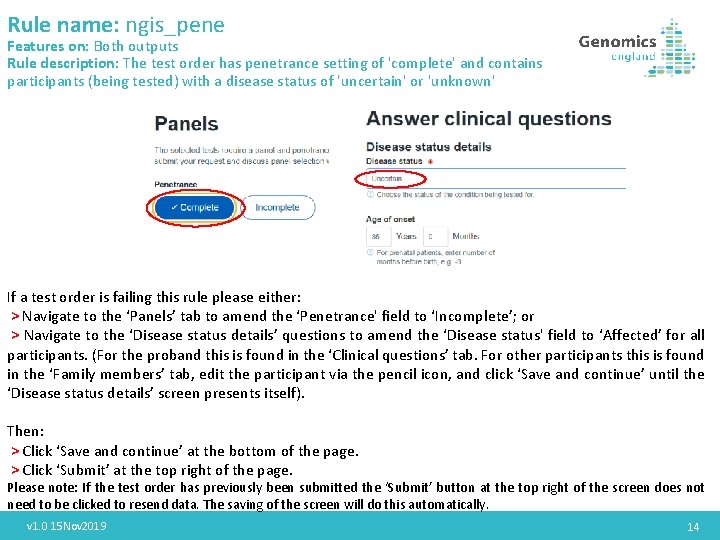
Rule name: ngis_pene Features on: Both outputs Rule description: The test order has penetrance setting of 'complete' and contains participants (being tested) with a disease status of 'uncertain' or 'unknown' If a test order is failing this rule please either: > Navigate to the ‘Panels’ tab to amend the ‘Penetrance' field to ‘Incomplete’; or > Navigate to the ‘Disease status details’ questions to amend the ‘Disease status' field to ‘Affected’ for all participants. (For the proband this is found in the ‘Clinical questions’ tab. For other participants this is found in the ‘Family members’ tab, edit the participant via the pencil icon, and click ‘Save and continue’ until the ‘Disease status details’ screen presents itself). Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 14

Rule name: ngis_relation Features on: Both outputs Rule description: The phenotypic sex / gender provided for the family member does not match the gender inferred from the relationship to the proband value. If a test order is failing this rule please: > Navigate to the ‘Family members’ tab and ensure each family member has a ‘Relationship to proband’ value that corresponds with the patient’s phenotypic sex or gender. > Amend and incorrect relationship values. This can be achieved by clicking on the pencil icon next to each family member and cycling through the screens by pressing ‘Save and continue’. Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 15
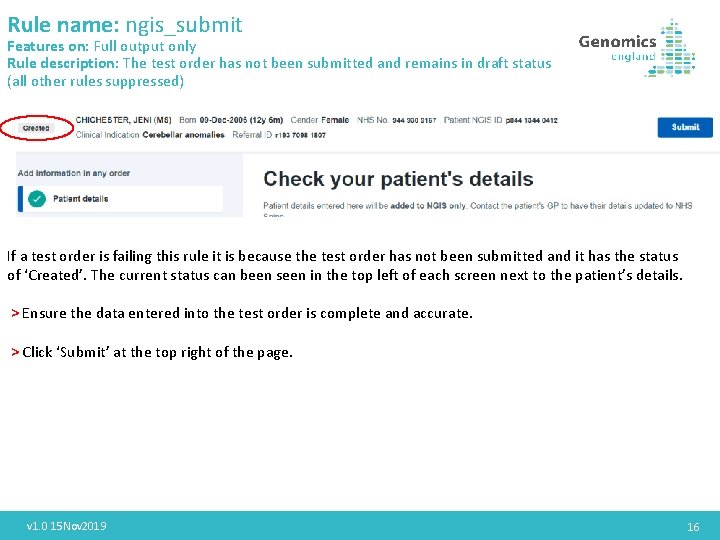
Rule name: ngis_submit Features on: Full output only Rule description: The test order has not been submitted and remains in draft status (all other rules suppressed) If a test order is failing this rule it is because the test order has not been submitted and it has the status of ‘Created’. The current status can been seen in the top left of each screen next to the patient’s details. > Ensure the data entered into the test order is complete and accurate. > Click ‘Submit’ at the top right of the page. v 1. 0 15 Nov 2019 16

Rule name: ngis_submit 2_ca Features on: Streamline output only Rule description: The test order has not been submitted after 4 weeks from the test order creation and both the germline and tumour sample data have not been added to the test order (all other rules suppressed) If a test order is failing this rule because the test order has not been submitted; 4 weeks have passed since the referral creation date; and at least one germline and at least one tumour sample record has not been added to the test order (as shown in the diagram above), please: > Add at least one tumour and one germline sample to the test order via the ‘Samples’ tab. Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 17

Rule name: ngis_submit 2_rd Features on: Streamline output only Rule description: The test order has not been submitted after 4 weeks from the test order creation and not all the family members have been added to the test order (all other rules suppressed) If a test order is failing this rule it is because 4 weeks have passed since the referral creation date, and the number of family members being tested (added via the ‘Family members’ tab) does not match the value selected at the ‘Total number of participants’ question on the ‘Test package’ tab, please either: > Add all family members to be tested to the test order; OR > Consider amending the number of participants for this tab. Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 18
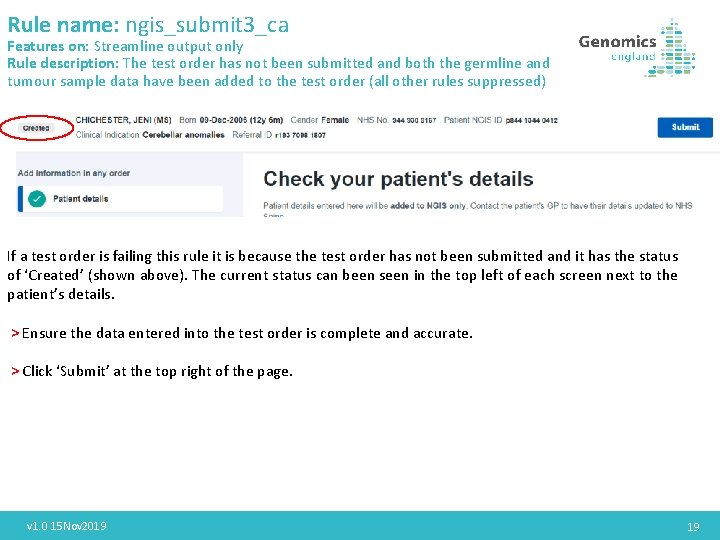
Rule name: ngis_submit 3_ca Features on: Streamline output only Rule description: The test order has not been submitted and both the germline and tumour sample data have been added to the test order (all other rules suppressed) If a test order is failing this rule it is because the test order has not been submitted and it has the status of ‘Created’ (shown above). The current status can been seen in the top left of each screen next to the patient’s details. > Ensure the data entered into the test order is complete and accurate. > Click ‘Submit’ at the top right of the page. v 1. 0 15 Nov 2019 19
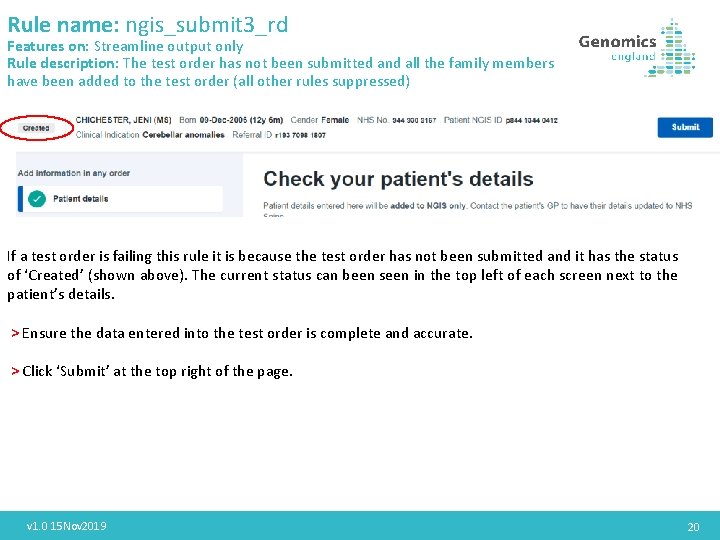
Rule name: ngis_submit 3_rd Features on: Streamline output only Rule description: The test order has not been submitted and all the family members have been added to the test order (all other rules suppressed) If a test order is failing this rule it is because the test order has not been submitted and it has the status of ‘Created’ (shown above). The current status can been seen in the top left of each screen next to the patient’s details. > Ensure the data entered into the test order is complete and accurate. > Click ‘Submit’ at the top right of the page. v 1. 0 15 Nov 2019 20

Rule name: ngis_tum. Con Features on: Both outputs Rule description: A tumour content value has not been provided for all tumour samples associated with the referral If a test order is failing this rule, please: > Add the tumour content value by navigating to the ‘Samples’ tab, selecting the tumour sample by pressing the “>” button, then select ‘Save and continue’ to reach the page with the ‘tumour content’ value > Enter the tumour content of the submitted tumour sample Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. v 1. 0 15 Nov 2019 21

Rule name: ngis_tum. Con. Low Features on: Both outputs Rule description: 'A 'Tumour content' value submitted in the TOMS referral is less than 30 percent for at least one solid tumour sample If a test order is failing this rule, please: > Review the value provided by navigating to the ‘Samples’ tab, selecting the tumour sample by pressing the “>” button, then select ‘Save and continue’ to reach the page with the ‘tumour content’ value > If the value isn’t correct, please amend. Then: > Click ‘Save and continue’ at the bottom of the page. > Click ‘Submit’ at the top right of the page. Please note: If the test order has previously been submitted the ‘Submit’ button at the top right of the screen does not need to be clicked to resend data. The saving of the screen will do this automatically. Otherwise: > If the value is correct, no further action is required unless informed otherwise. v 1. 0 15 Nov 2019 22

Rule name: ngis_tumour Features on: Full Output only Rule description: Tumour sample data has not been submitted via the GEL 1001 csv file If a test order is failing this rule please: > Submit a GEL 1001 via SFTP, ensuring that there is a tumour record for the cancer referral Rule name: ngis_tumour 2 Features on: Streamline Output only Rule description: Tumour sample data has not been submitted via the GEL 1001 csv file for the test order, despite there being a submission of a valid GEL 1001 file from the GLH since the test order submission date. If a test order is failing this rule please: > Submit a GEL 1001 via SFTP, ensuring that there is a tumour record for the cancer referral v 1. 0 15 Nov 2019 23
- Slides: 23