Newonset Diabetes After Transplantation NODAT 1 Dr Ruba

New-onset Diabetes After Transplantation (NODAT) 1 Dr. Ruba Nashawati

Incidence § 5 -20% 1 st year § Up to 30% later 2

NODAT Risk for Graft Survival 3
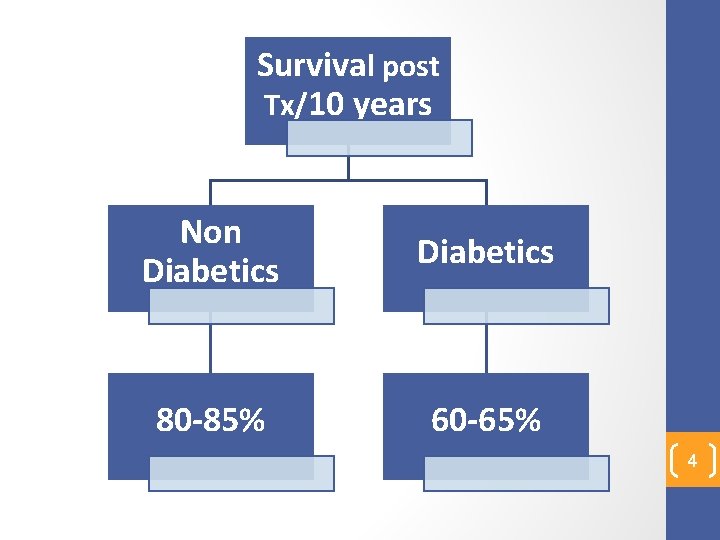
Survival post Tx/10 years Non Diabetics 80 -85% 60 -65% 4
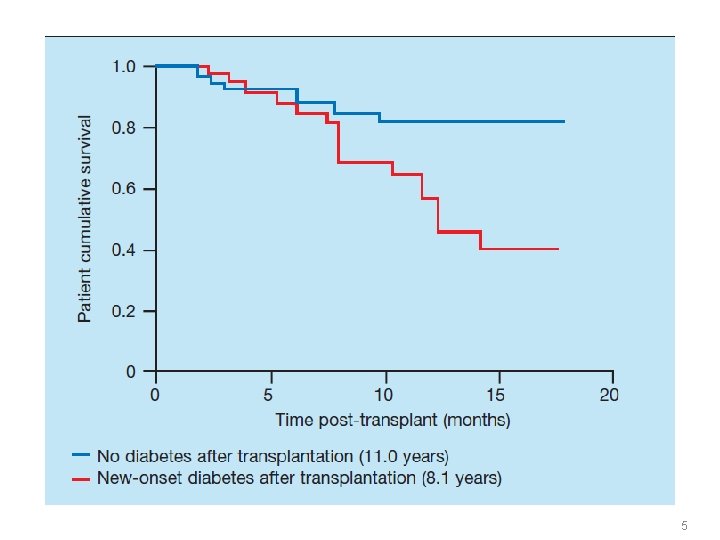
5

NODAT � Cardiovascular death + Infections 6
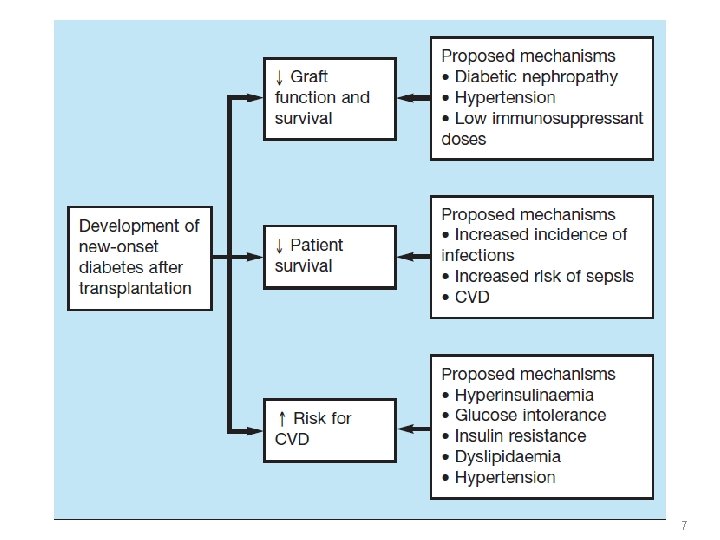
7

Definition 8

WHO world health organization criteria 9

WHO world health organization criteria 10

WHO world health organization criteria 11

Risk factor 12

13

14

OBESITY : BMI >30 15

Azathioprine + MMF are Non Diabetogenic MMF may mitigate Diabetes 16

Prevention 17

• Screen All patients Before Tx to Before Tx establish their risk of developing NODAT • Monitor using Fasting Plasma Glucose • Thereafter Glucose measurements at 3, 6, 12 months then annually 3, 6, 12 18

• Screening (CASUAL) Blood Glucose CASUAL measurements useful but Cannot identify Glucose Intolerance • Consider 2 hour 75 g Glucose tolerance test with Impaired FBS and High-Risk 19

For High Risk • Lower BMI < 25 • Steroid free IS • Cs. A opposite to TAC • MMF 20

SCREENING FOR NEW-ONSET DIABETES AFTER TRANSPLANTATION: 21 KDIGO

v. Screening all Nondiabetic KTRs with 1. Fasting Plasma Glucose 2. Oral Glucose Tolerance Testing 3. and/or Hb. A 1 c 22 KDIGO

at least: • Weekly For 4 Weeks • Every 3 Months For 1 Year • Annually, Thereafter 23 KDIGO

v. Screening for NODAT with 1. Fasting Glucose 2. Oral Glucose Tolerance Testing 3. and/or Hb. A 1 c 24 KDIGO

After Starting or Starting Increasing the dose, of CNIs, m. TORi, or Corticosteroids 25 KDIGO

HBA 1 c Don’t rely on HBA 1 c � takers weeks No useful if recent blood transfution (somebody's else HBA 1 c) Used only for monitoring treatment 26

Other modifiable cardiovascular risk factors : • Hypertension • Hyperlipidemia • Smoking should be identified and treated 27

Diagnosis 28

Symptoms • Polyuria • Polydipsia • Weight Loos • Fatigue 29

Blood Glucose 1 st Visit WHO Criteria 30

Established NODAT 31

1. Diet + Weight Reduction 2. Immunosuppressive 3. Blood Glucose 4. Withdraw B-blocker + Thiazide 32

Diet + Weight Control 33

Lifestyle Modifications • NA < 2400 mg /day • Saturated Fats < 7 % calories • Trans-fatty Acids 2 -3 % of calories 34

Two servings of Fish per week > than 25 g/day of Dietary Fiber Regular Aerobic Exercise Weight loss of 5% to 10% 35

Immunosuppressive Medications 36

Changes that may Reverse Or Ameliorate NODAT include: 37
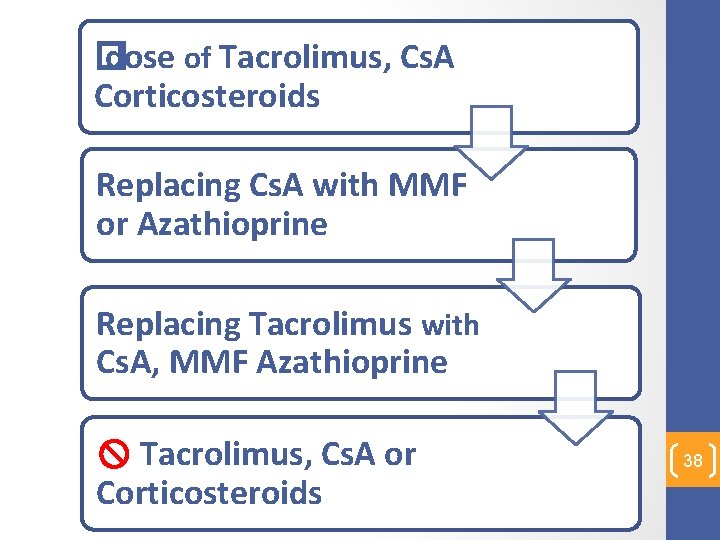
� dose of Tacrolimus, Cs. A Corticosteroids Replacing Cs. A with MMF or Azathioprine Replacing Tacrolimus with Cs. A, MMF Azathioprine Tacrolimus, Cs. A or Corticosteroids 38

If NODAT develops + after Weighing the Risk Of Rejection and other potential adverse effects. 39 KDIGO

consider Modifying the Modifying immunosuppressive drug to Reverse or Ameliorate Diabetes or Ameliorate 40 KDIGO

Glycemic control 41

Similar to Non-transplant patient 42

Recommended • HBA 1 c level < 7. 5% • FPG <100 mg/dl • 2 -hour postprandial PG < 140 mg/dl 43

HBA 1 c < 6. 0% � Risks Hypoglycemia � Increased Mortality. 44

Consider 1. Targeting HBA 1 c 7. 0– 7. 5 % 2. Avoid targeting Hb. A 1 c ≤ 6. 0% 3. especially if hypoglycemic reactions are common 45 KDIGO
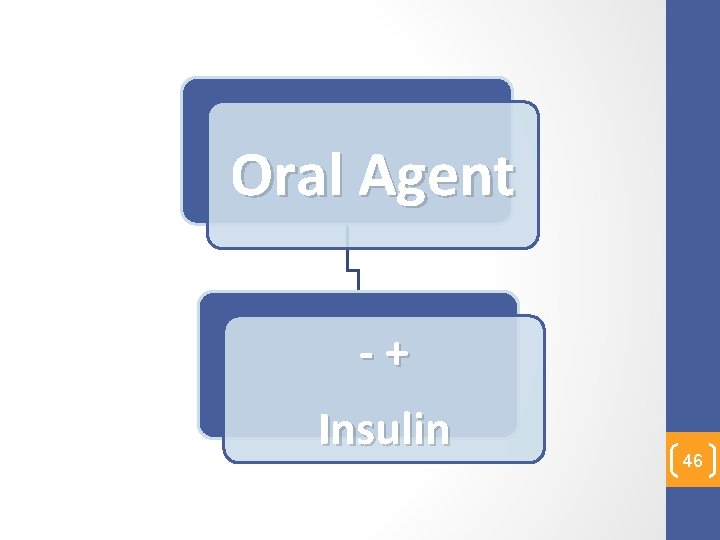
Oral Agent - + Insulin 46

• Hyperglycemia soon after transplantation� Insulin • Followed by withdrawal of Insulin and replacement with Oral Ant. Diabetic agents 47

Step 1 48
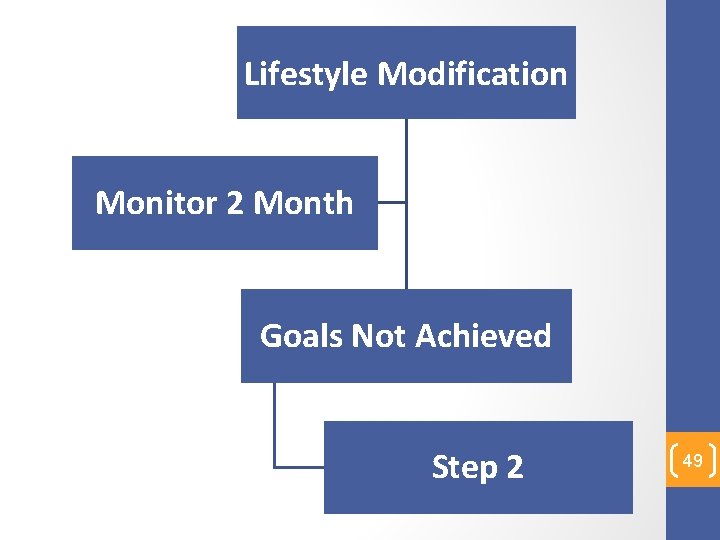
Lifestyle Modification Monitor 2 Month Goals Not Achieved Step 2 49

Step 2 50
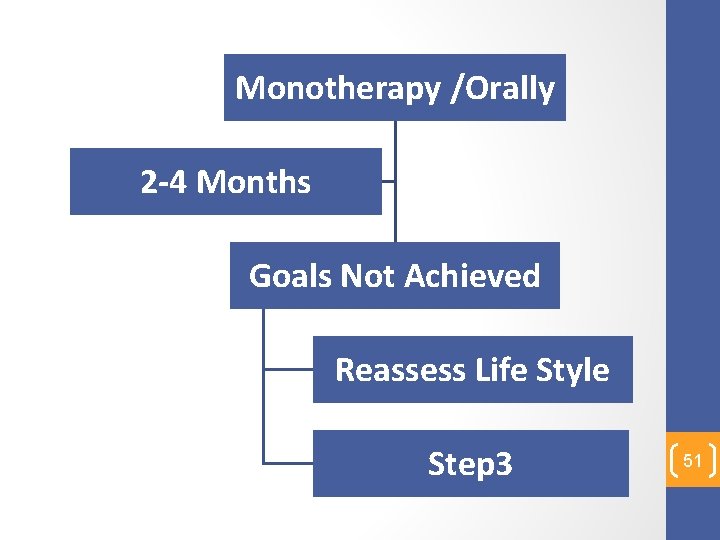
Monotherapy /Orally 2 -4 Months Goals Not Achieved Reassess Life Style Step 3 51

Step 3 52
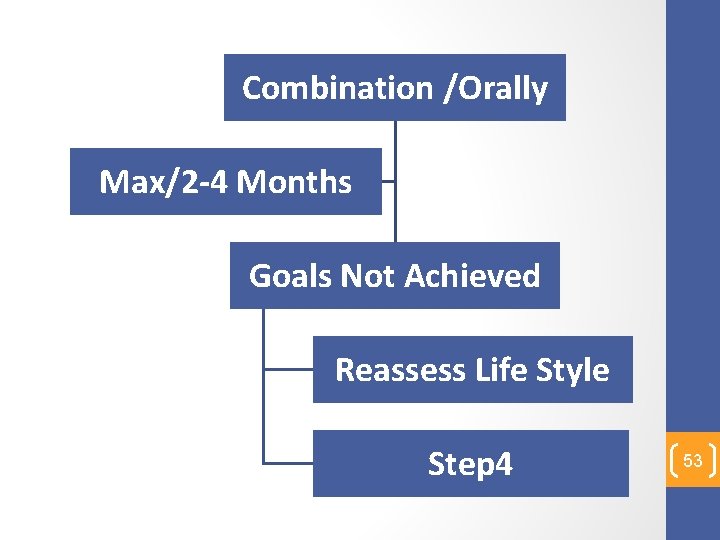
Combination /Orally Max/2 -4 Months Goals Not Achieved Reassess Life Style Step 4 53

Step 4 54
Insulin + Oral Agent Symptomatic Hypoglycemia Ketosis Step 5 55

Step 5 56
Insulin Achieve Target Later May Add Oral Agent 57

Oral Anti Diabetic 58

First-generation Sulfonylureas 1. ↑ Cs. A levels 2. Adjust according to GFR 3. Avoid Acetohexamide Tolazamide 59

Second-generation Sulfonylureas 1. ↑ Cs. A levels 2. No dose adjustment 3. except Glibenclamide Glisentideb 60

Alpha-glucosidase inhibitors Check GFR Biguanides 1. Phenformin Contraindicated 2. Metformin Contraindicated if SCr ≥ 1. 5 Meglitinides Check GFR ↑ Repaglinide with Cs. A 61

Thiazolidinedione No dose adjustment Incretin mimetic Exenatide: Avoid if GFR <30 62

Amylin Analog Pramlintide No dose adjustment if GFR >20 DDP-4 Inhibitor Sitagliptin ↓ 50% if GFR 30– 50 ↓ 75% if GFR <30 Vildagliptine Avoid CKD/Hemodialysis 63

Other Withdraw B-Blocker + Thiazide Aspirin? ! 64

in patients with Diabetes: Aspirin (65– 100 mg/day) for the primary prevention of CVD BASED ON Balancing the risk for Ischemic events /Bleeding 65 KDIGO

66

Approach to the Assessment and Management of Diabetes in Organ Transplant Recipients 67

Pre-transplant Baseline Evaluation 1. Screen for asymptomatic Diabetes or glucose intolerance 2. Identify Metabolic Syndrome + CVD 3. Identify High-Risk characteristics 4. Individualized Counseling 68

Individualization Of Immunosuppressive Therapy Consider Risk of NODAT in the Selection of Immunosuppression 69

Individualization Of Immunosuppressive Therapy 1. Consider avoiding /rapidly tapering Corticosteroids post-transplantation 2. Consider Risks + benefits with Calcineurin Inhibitors 70

Monitor For Diabetes Post transplantation Measure blood Glucose in clinic Weekly � 4 weeks then at 3 months 6 months thereafter Annually 71

Assess Response to Treatment in patients who develop Diabetes 1. Measure pre + Post-prandial Blood Glucose 2. self-monitoring of capillary glucose in patients on insulin + some with Oral antidiabetic Agents 72

Assess Response to Treatment in patients who develop Diabetes 1. Measurement of HBA 1 c / 3 months 2. Assessment of blood Lipid Profile 3. Rigorous control of Blood Pressure 4. Consideration of Antiplatelet Therapy for high-risk patients 73

Surveillance For Long-term Complications Of Diabetes • Retinopathy • Neuropathy • Nephropathy 74

75
- Slides: 75