Newish Psychotropics NICOLE L CUPPLES PHARMD BCPP BCPS

Newish Psychotropics NICOLE L. CUPPLES PHARMD, BCPP, BCPS CLINICAL PHARMACY SPECIALIST- AUDIE L MURPHY VA ADJUNCT FACULTY-UNIVERSITY OF TEXAS HEALTH SCIENCE SAN ANTONIO SEPTEMBER 16, 2017

OBJECTIVES • Review indications for 3 new psychotropic medications • Identify advantages and disadvantages to agents’ use • Describe new mechanism of action of pimavanserin • Explain the restrictions and training required for prescribing buprenorphine subdermal implant
PRE-TEST QUESTION # 1 • The only FDA-approved treatment option for Parkinson’s Disease Psychosis is an atypical antipsychotic. • A. True • B. False

PRE-TEST QUESTION # 2 • Pimavanserin acts as a/an: • A. Agonist • B. Antagonist • C. Inverse agonist • D. Pacifist

PRE-TEST QUESTION # 3 • Which of these medications is/are FDA approved for tardive dyskinesia? A. Tetrabenazine B. Deutetrabenazine C. Valbenazine D. Both A and C

PRE-TEST QUESTON # 4 • Buprenorphine implant can be used as a treatment method for how long? • A. 3 months • B. 6 months • C. 12 months • D. Indefinitely

Newish Psychotropics • Pimavanserin (Nuplazid®) • • Valbenazine (Ingrezza®) • • FDA approved 4/2016 for Parkinson’s Disease Psychosis FDA approved 4/2017 for tardive dyskinesia treatment Buprenorphine implant (Probuphine®) • FDA approved 5/2016 for maintenance treatment of opioid dependence

PARKINSON’S DISEASE PSYCHOSIS DIAGNOSIS • Presence of one or more • Hallucinations • Delusions • Illusions • False sense of presence • Primary diagnosis of Parkinson’s Disease • Symptoms of psychosis ≥ 1 month • Other possible causes excluded • Specifiers

PARKINSON’S DISEASE PSYCHOSIS DELUSION S HALLUCINATIONS FALSE SENSE OF PRESENC E ILLUSION S

CURRENT TREATMENT OPTIONS • • • Only pimavanserin is FDA approved for Parkinson’s Disease Psychosis • Atypical antipsychotic • Inverse agonist and antagonist activity at serotonin 5 HT 2 A , less at 5 HT 2 C Quetiapine and clozapine • Atypical antipsychotic • Antagonist at dopamine D 2 and serotonin 5 HT 2 A Rivastigmine and donepezil • • Anticholinesterase inhibitors Electroconvulsive therapy
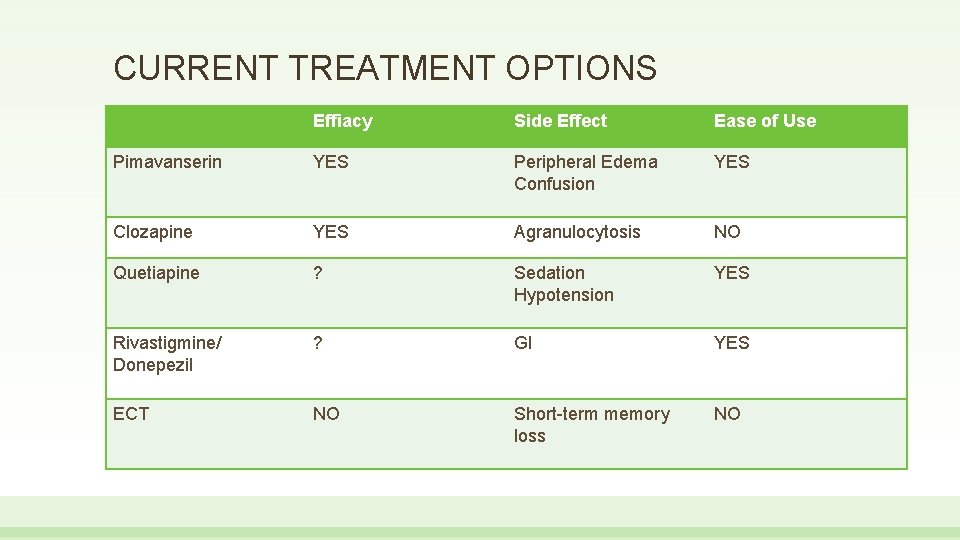
CURRENT TREATMENT OPTIONS Effiacy Side Effect Ease of Use Pimavanserin YES Peripheral Edema Confusion YES Clozapine YES Agranulocytosis NO Quetiapine ? Sedation Hypotension YES Rivastigmine/ Donepezil ? GI YES ECT NO Short-term memory loss NO
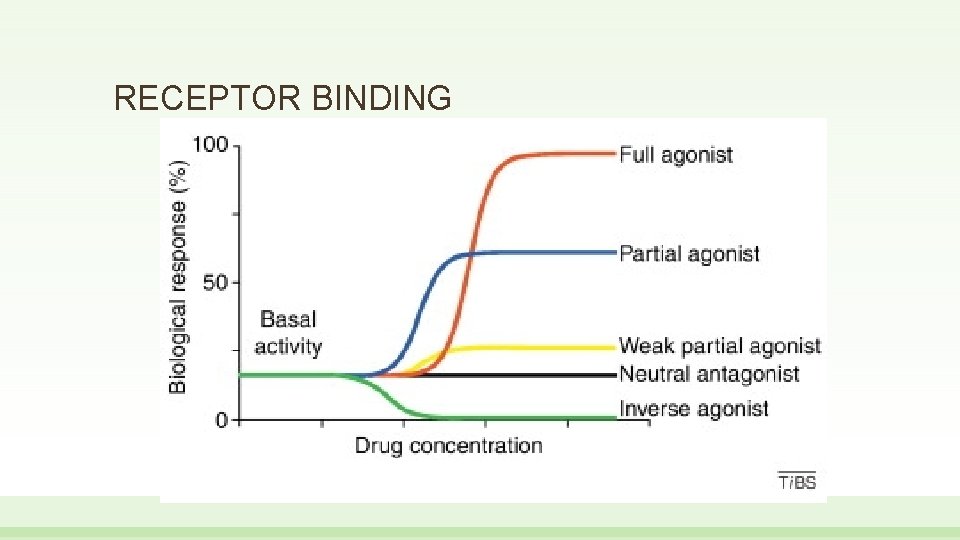
RECEPTOR BINDING
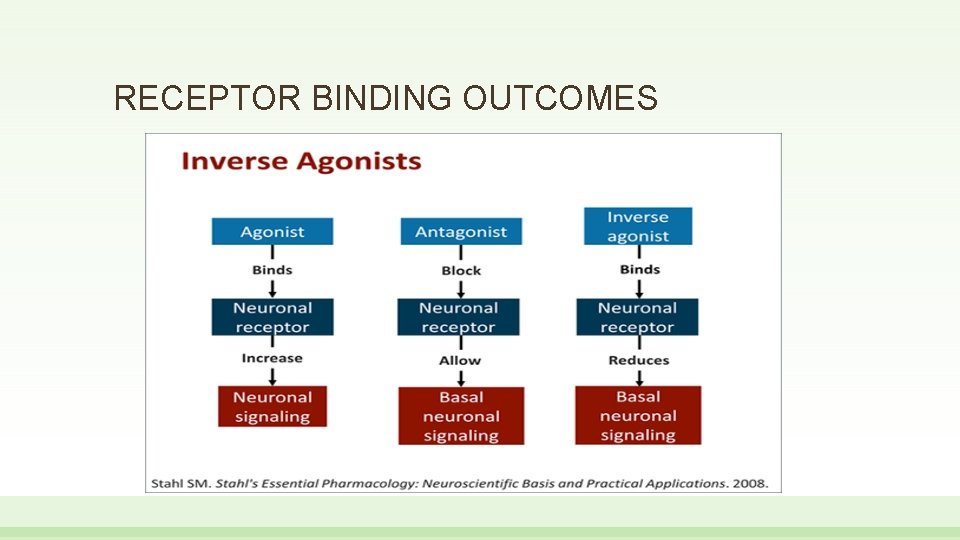
RECEPTOR BINDING OUTCOMES

PIMAVANSERIN DOSING • Daily 34 mg dose as two 17 mg tablets • May be taken with or without food • Titration is unnecessary • Dose change/Withhold for • Coadministration with strong CYP 3 A 4 inhibitors (e. g. , ketoconazole) • • 17 mg daily Coadministration with strong CYP 3 A 4 inducers • May need to increase dose, no specific dose guidance given • Renal Cr. CL ≤ 30 m. L/min –not recommended • Hepatic impairment-not defined and not recommended

PIMAVANSERIN SIDE EFFECTS • Prolongation of the QT interval • Mean increases of ~5 -8 msec • Concentration dependent • Avoid use with other QT prolonging medications, those with history of arrhythmias • Boxed warning for increased mortality in dementia-related psychosis • Peripheral edema and confusional state occurred ≥ 2 x placebo • Discontinuation rate of 10/95 (9. 5%) treated vs 2/90 (2. 1%) placebo
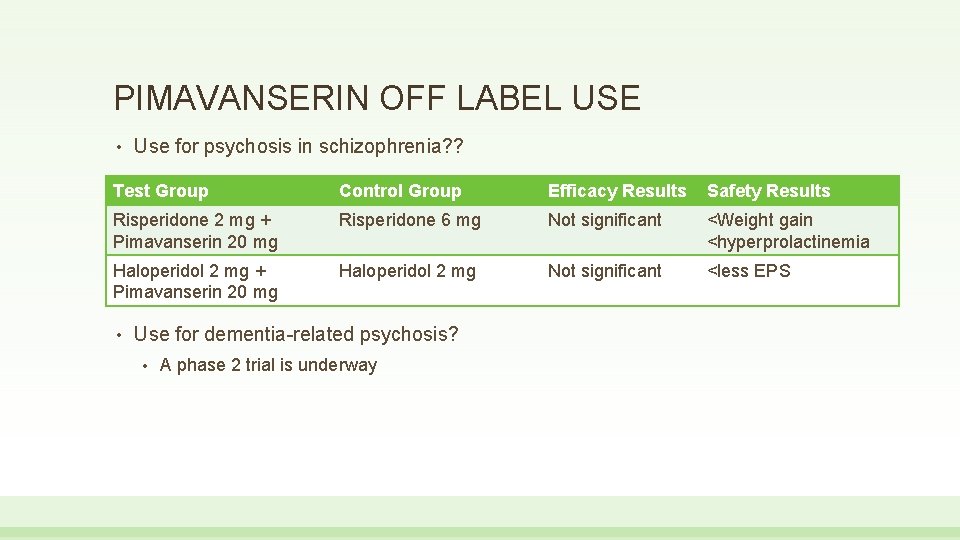
PIMAVANSERIN OFF LABEL USE • Use for psychosis in schizophrenia? ? Test Group Control Group Efficacy Results Safety Results Risperidone 2 mg + Pimavanserin 20 mg Risperidone 6 mg Not significant <Weight gain <hyperprolactinemia Haloperidol 2 mg + Pimavanserin 20 mg Haloperidol 2 mg Not significant <less EPS • Use for dementia-related psychosis? • A phase 2 trial is underway

PIMAVANSERIN PK Parameter Result Metabolized CYP 3 A 4/5 TMAX 6 hours T½ 57 hours 200 hours for active metabolite Elimination Fecal-1. 53 % Renal-0. 55 %

Tardive Dyskinesia • • Debilitating side effect from use of psychotropic medications • Dyskinesia lasting for at least a few months • Tongue, lower face and jaw • Athetoid/Choreiform • Extremities Neuroleptic withdrawal-emergent dyskinesia • <4 -8 weeks duration
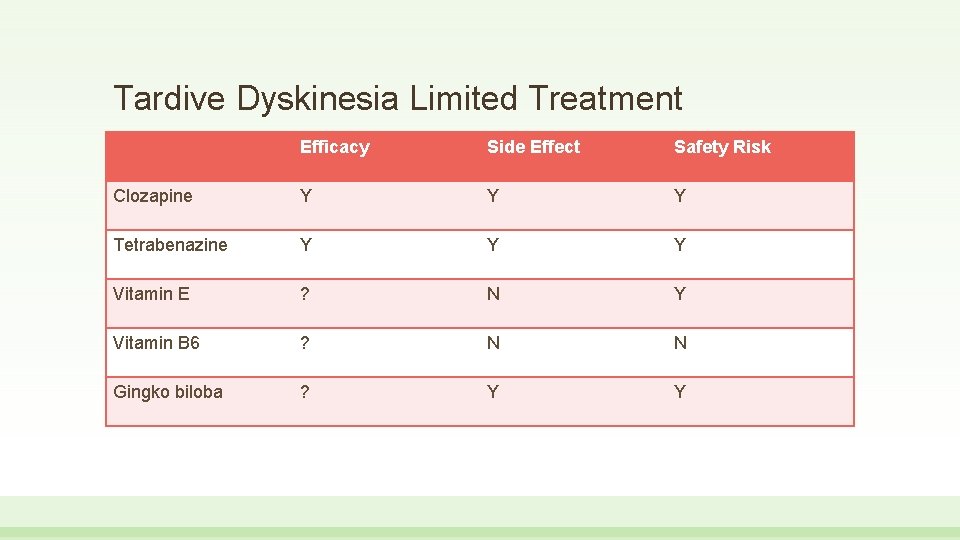
Tardive Dyskinesia Limited Treatment Efficacy Side Effect Safety Risk Clozapine Y Y Y Tetrabenazine Y Y Y Vitamin E ? N Y Vitamin B 6 ? N N Gingko biloba ? Y Y
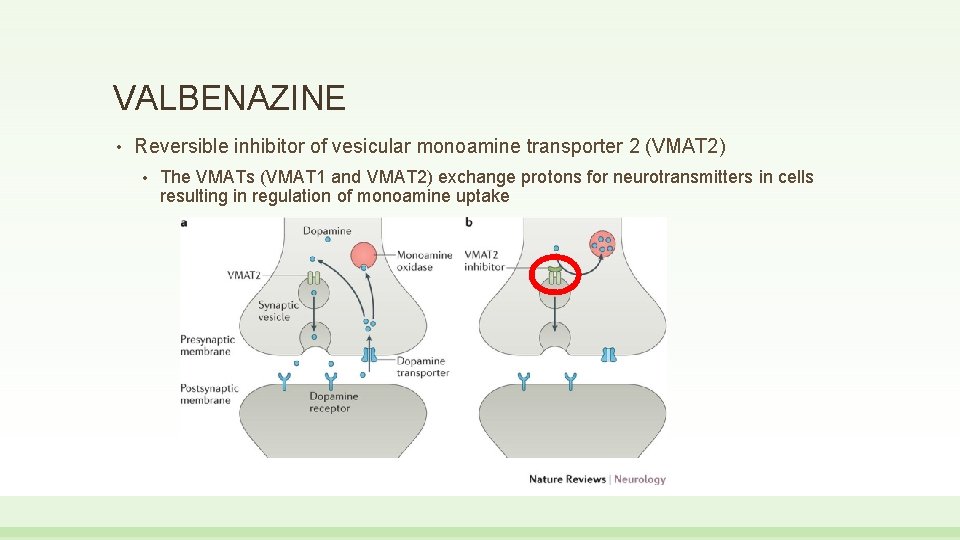
VALBENAZINE • Reversible inhibitor of vesicular monoamine transporter 2 (VMAT 2) • The VMATs (VMAT 1 and VMAT 2) exchange protons for neurotransmitters in cells resulting in regulation of monoamine uptake

VMAT 2 INHIBATORS VMAT 1 Found in PNS VMAT 2 Found in PNS/CNS • No inhibitors known • • reserpine tetrabenazine deutetrabenazine (Austedo®) valbenazine
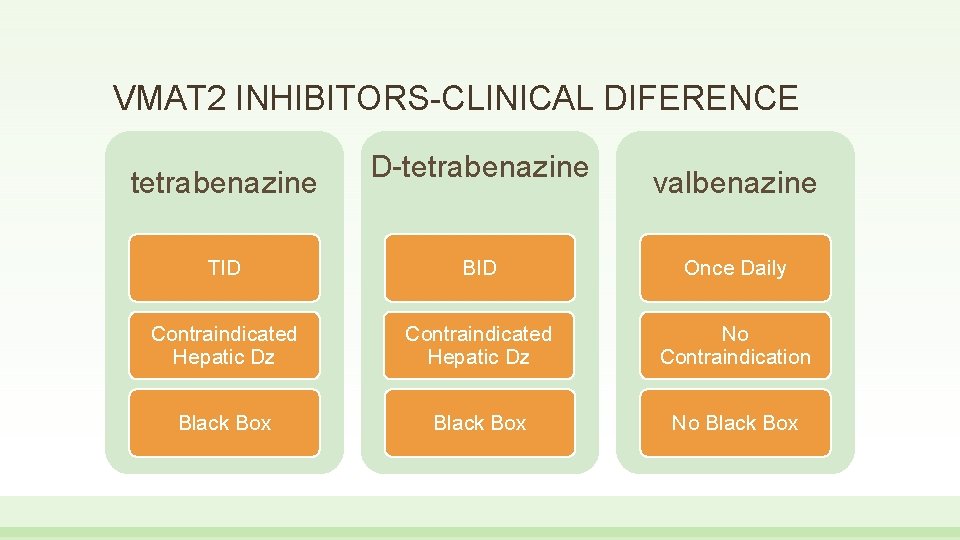
VMAT 2 INHIBITORS-CLINICAL DIFERENCE tetrabenazine D-tetrabenazine valbenazine TID BID Once Daily Contraindicated Hepatic Dz No Contraindication Black Box No Black Box

VALBENAZINE DOSING • FDA approved for tardive dyskinesia • Initial dose 40 mg x 1 week • Daily dose of 80 mg • Hepatic impairment • • • Child-Pugh score 7 -15 • 40 mg daily Renal impairment • Cr. Cl < 30 ml/min • Not recommended Use of strong CYP 3 A 4 inhibitor • 40 mg daily
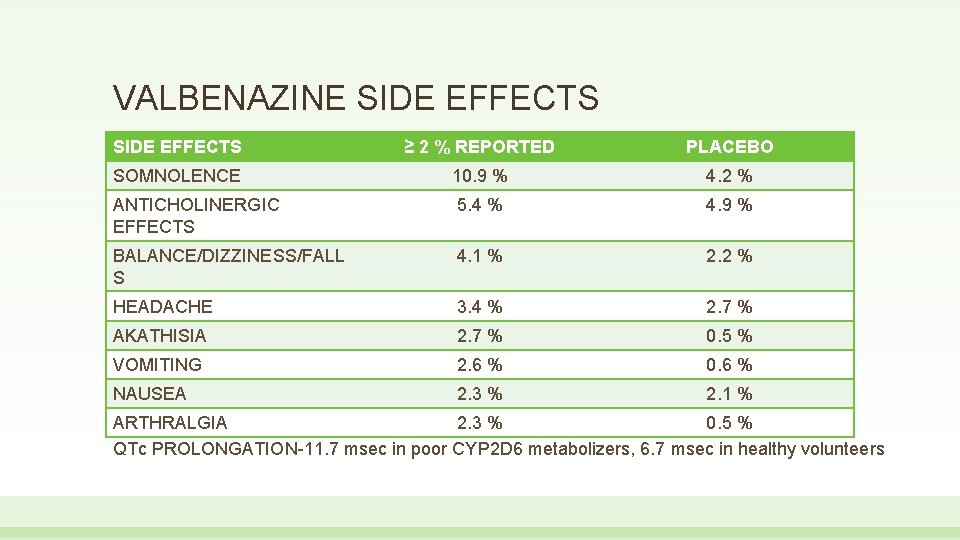
VALBENAZINE SIDE EFFECTS ≥ 2 % REPORTED PLACEBO SOMNOLENCE 10. 9 % 4. 2 % ANTICHOLINERGIC EFFECTS 5. 4 % 4. 9 % BALANCE/DIZZINESS/FALL S 4. 1 % 2. 2 % HEADACHE 3. 4 % 2. 7 % AKATHISIA 2. 7 % 0. 5 % VOMITING 2. 6 % 0. 6 % NAUSEA 2. 3 % 2. 1 % ARTHRALGIA 2. 3 % 0. 5 % QTc PROLONGATION-11. 7 msec in poor CYP 2 D 6 metabolizers, 6. 7 msec in healthy volunteers

SPECIAL POPULATIONS • • Pregnancy • May cause fetal harm • Advise not to breastfeed during treatment and for 5 days after last dose Pediatric • • Geriatric • • Not established No dose adjustment, 16 % of patients ≥ 65 years in 3 trials CYP 2 D 6 poor metabolizers • Consider reducing dose if intolerable side-effects
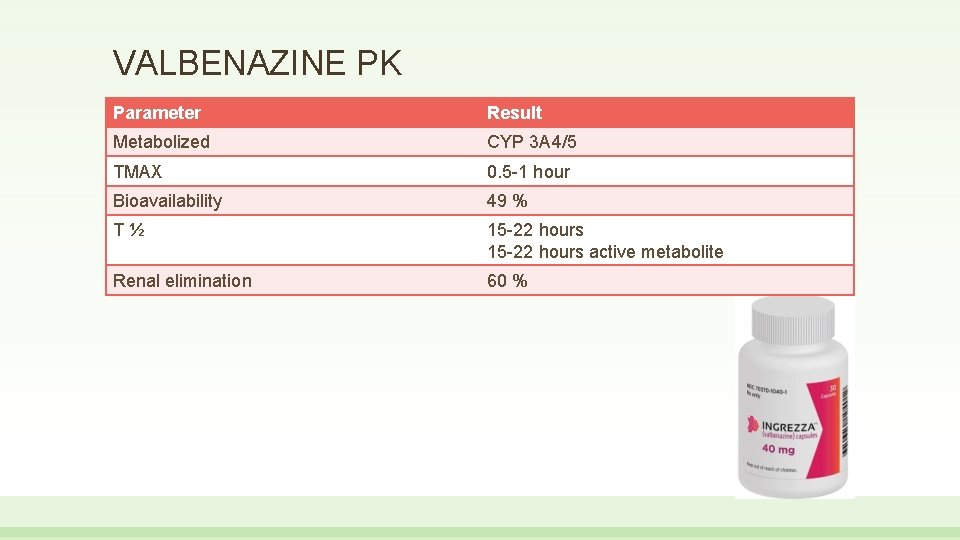
VALBENAZINE PK Parameter Result Metabolized CYP 3 A 4/5 TMAX 0. 5 -1 hour Bioavailability 49 % T½ 15 -22 hours active metabolite Renal elimination 60 %

BUPRENORPHINE IMPLANT “Indicated for the maintenance treatment of opioid dependence in patients who have achieved and sustained prolonged clinical stability on low-to-moderate doses… (i. e. , doses of no more than 8 mg per day 1 of Subutex or Suboxone sublingual tablet or generic equivalent). ” 1. 2. Bold type presenter’s emphasis Package insert citation

BUPRENORPHINE IMPLANT • • Failed FDA approval 2013 • Safety established with implant • Approximately 39. 5 % needed buprenorphine supplement with implant • Majority of patients were heroin abusers FDA approval achieved May 2016 • Approved for patients with ≥ 90 days stability on 8 mg/day buprenorphine or less • 17. 9 % treatment vs 14. 6 % placebo received oral rescue buprenorphine p =. 53 • Majority of patients were white males with prescription drug abuse history

BUPRENORPHINE IMPLANT • Four, 1 inch long Pro. Neura. TM rods inserted in inner upper arm • Oral overlap should not be necessary • Medication is released for 6 months • Prescribers must have buprenorphine prescribing authority • Those prescribing must complete a live REMS training session for use • Insertion and removal requires a live training program and prior procedure within 3 months surgical
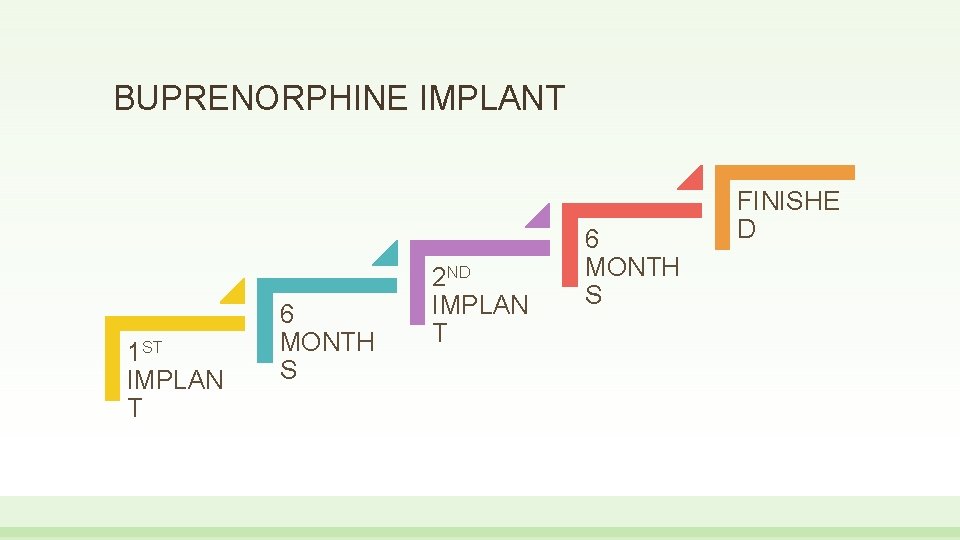
BUPRENORPHINE IMPLANT 1 ST IMPLAN T 6 MONTH S 2 ND IMPLAN T 6 MONTH S FINISHE D

BUPRENORPHINE IMPLANT • Decreases illicit use and accidental ingestion • Patient adherence is certain • Fewer follow up appointments needed • Steady release of drug

BUPRENORPHINE IMPLANT • Limited patient population • Implant may be expulsed, migrate or protrude • Opioid therapy for pain treatment is difficult to manage • One year use only • Cost?
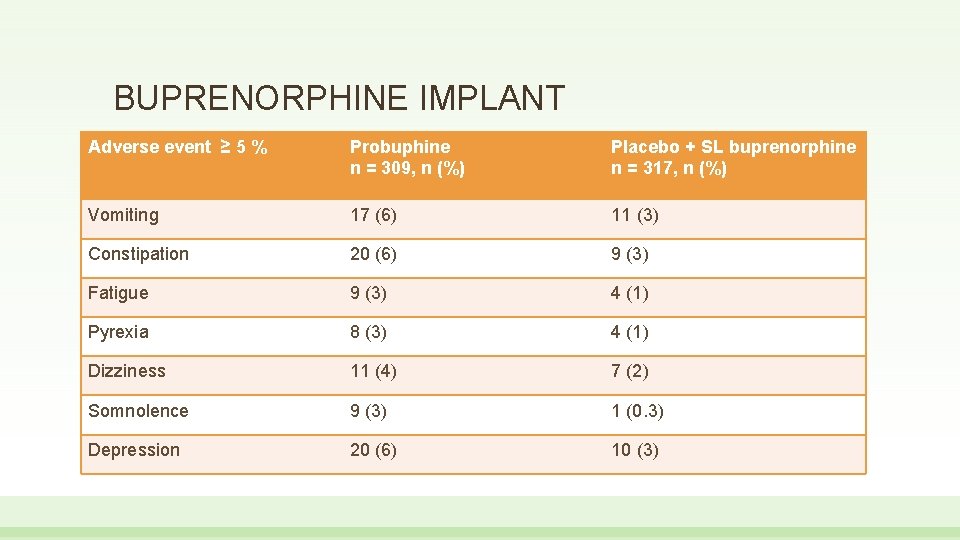
BUPRENORPHINE IMPLANT Adverse event ≥ 5 % Probuphine n = 309, n (%) Placebo + SL buprenorphine n = 317, n (%) Vomiting 17 (6) 11 (3) Constipation 20 (6) 9 (3) Fatigue 9 (3) 4 (1) Pyrexia 8 (3) 4 (1) Dizziness 11 (4) 7 (2) Somnolence 9 (3) 1 (0. 3) Depression 20 (6) 10 (3)

SPECIAL POPULATIONS • “Caution” in elderly and those with hepatic impairment • Pregnancy • • Neonatal withdrawal syndrome • Both buprenorphine and metabolite are present in breast milk and infant urine Not established for pediatric use or those < 16 years of age
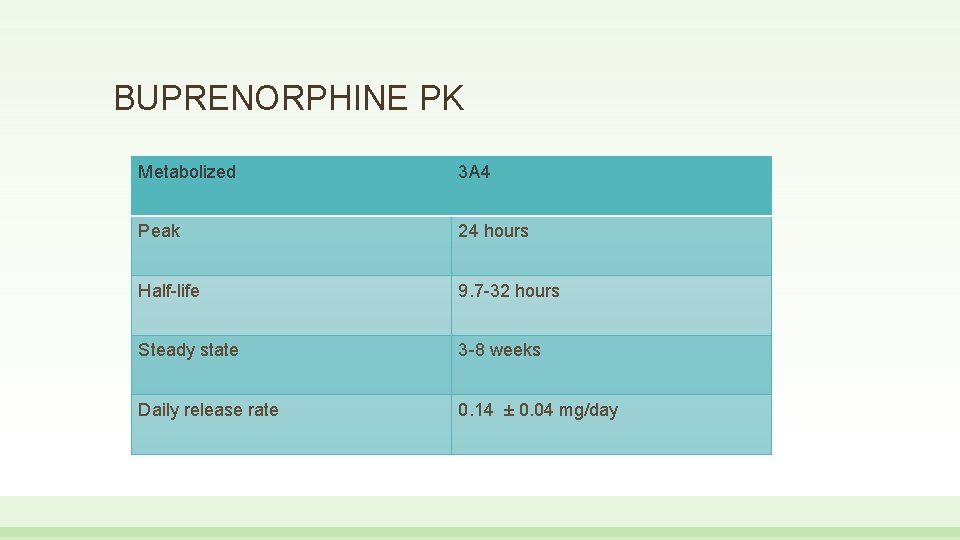
BUPRENORPHINE PK Metabolized 3 A 4 Peak 24 hours Half-life 9. 7 -32 hours Steady state 3 -8 weeks Daily release rate 0. 14 ± 0. 04 mg/day
PRE-TEST QUESTION # 1 • The only FDA-approved treatment option for Parkinson’s Disease Psychosis is an atypical antipsychotic. • A. True • B. False

PRE-TEST QUESTION # 2 • Pimavanserin acts as a/an: • A. Agonist • B. Antagonist • C. Inverse agonist • D. Pacifist

PRE-TEST QUESTION # 3 • Which of these medications is/are FDA approved for tardive dyskinesia? A. Tetrabenazine B. Deutetrabenazine C. Valbenazine D. Both A and C
PRE-TEST QUESTON # 4 • Buprenorphine implant can be used as a treatment method for how long? • A. 3 months • B. 6 months • C. 12 months • D. Indefinitely

References • Sarva H, Henchcliffe C. Evidence for the use of pimavanserin in the treatment of Parkinson’s disease psychosis. Ther Adv Neruol Disord. 2016; 9(6): 462 -73 • Meltzer H, Elkis H, Vanover K, et al. Pimavanserin, a selective serotonin (5 -HT)2 A-invers agonist, enhances the efficacy and safet of risperidone, 2 mg/day, but does not enhance efficacy of haloperidol, 2 mg/day: Comparison with reference dose risperidone, 6 mg/day. Schizophr Res. 2012; 141(2 -3): 144 -52. • Nutt D, Stahl S, Blier P et al. Inverse agonists-What do they mean for psychiatry? Eur Neruopsychopharmacol. 2017; 27: 87 -90. • Pimavanserin (NUPLAZID)National Drug Monograph. Washington, DC: Pharmacy Benefits Management Services, Medical Advisory Panel, and VISN Pharmacist Executives, Veterans Health Administration, Department of Veterans Affairs; October 2016. • Product Information: NUPLAZID(pimavanserin)tablets. Acadia Pharmaceuticals Inc. (per manufacturer), San Diego, CA, 2017.

References • Rana AQ, Chaudry ZM, Blanchet PJ. New and emerging treatments for symptomatic tardive dyskinesia. Drug Des Devel Ther. 2013 Nov 6; 7: 1329 -40. • Lerner V, Miodownik C, Kaptsan A, et al. Vitamin B 6 treatment for tardive dyskinesia: a randomized, double-blind, placebo-controlled, crossover study. J Clin Psychiatry. 2007; 68 (11): 1648– 54 • Zhang WF, Tan YL, Zhang XY, et al. Extract of Ginkgo biloba treatment for tardive dyskinesia in schizophrenia: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2011 May; 72(5): 615 -21. • Hauser RA, Factor SA, Marder SR. KINECT 3: A Phase 3 Randomized, Double-Blind, Placebo -Controlled Trial of Valbenazine for Tardive Dyskinesia. Am J Psychiatry 2017; 174: 476 -84 • Bernstein AI, Stout KA, Miller GW. The vesicular monoamine transporter 2: An underexplored pharmacological target. Neurochem Int 2014; 73: 89 -97.

References • Product Information: INGREZZA (valbenazine) capsules, for oral use. Neurocrine Biosciences, Inc. (per manufacturer) San Diego, CA, 2017. • Rosenthal RN, Lofwall MR, Kim S, et al. Effect of Buprenorphine Implants on Illicit Opioid Use Among Abstinent Adults With Opioid Dependence Treated With Sublingual Buprenorphine: A Randomized Clinical Trial. JAMA. 2016; 316(3): 282– 90. • Barnwal P, Das S, Mondal S, et al. Probuphine (buprenorphine implant): A promising candidate in opioid dependence. Ther Adv Psychopharmacol. 2017; 7(3): 119 -34 • Itzoe ML, Guarnieri M. New developments in managing opioid addiction: Impact of a subdermal buprenorphine implant. Drug Des Devel Ther 2017; 10(11): 1429 -37. • Product Information: PROBUPHINE (buprenorphine) implant for subdermal administration CIII. Braeburn Pharmaceuticals Inc. (per manufacturer) Princeton, NJ, 2016.

QUESTIONS?
- Slides: 43