New Zealand Universal List of Medicines Some history



- Slides: 5

New Zealand Universal List of Medicines

Some history • Origin – Ministry of Health initiative to standardise information about medicines • Development started in 2009, first release in 2011. • Steady uptake into clinical and administrative systems since them • Evolutionary development of resources provided • National programme to move to a fully interoperable digital health ecosystem • Becomes mandatory for all primary care clinical systems in January 2022 2

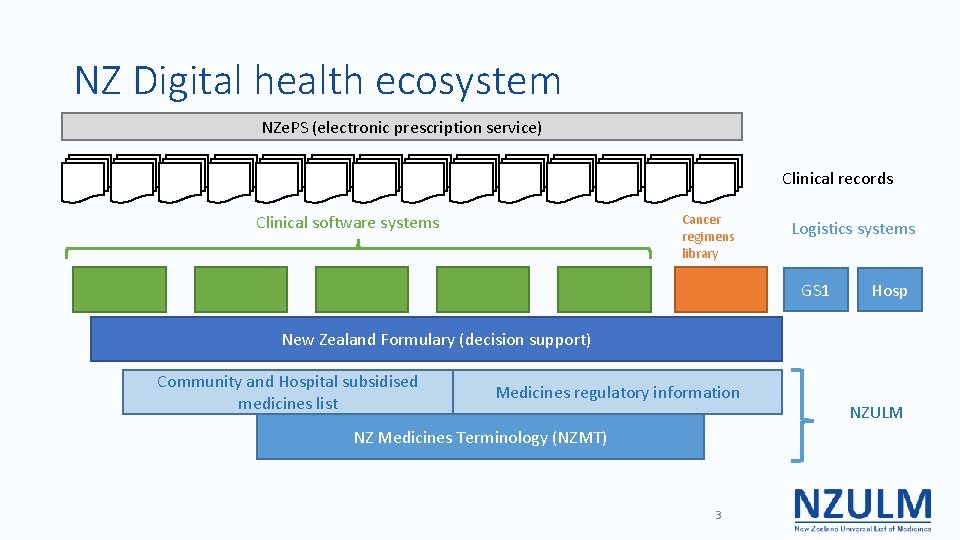
NZ Digital health ecosystem NZe. PS (electronic prescription service) Clinical records Clinical software systems Cancer regimens library Logistics systems GS 1 Hosp New Zealand Formulary (decision support) Community and Hospital subsidised medicines list Medicines regulatory information NZ Medicines Terminology (NZMT) 3 NZULM

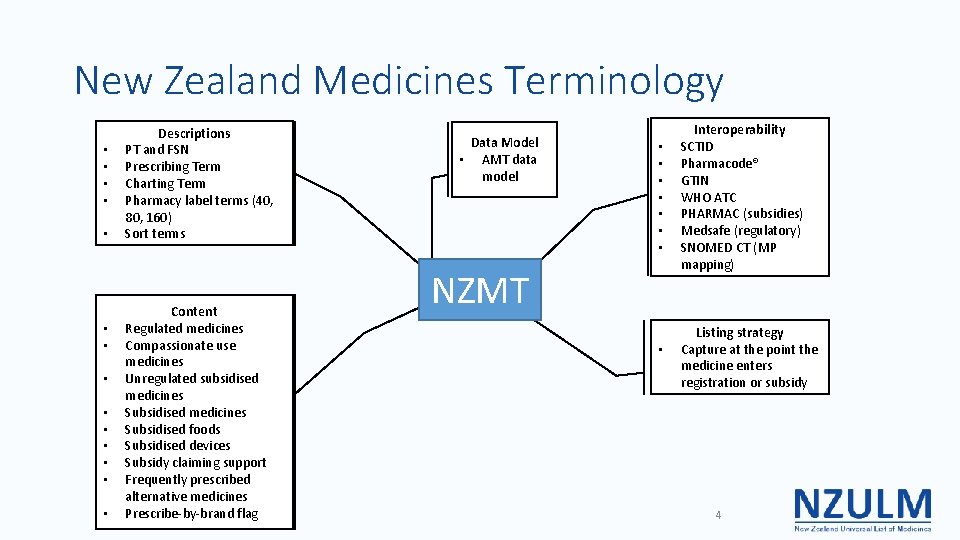
New Zealand Medicines Terminology • • • • Descriptions PT and FSN Prescribing Term Charting Term Pharmacy label terms (40, 80, 160) Sort terms Content Regulated medicines Compassionate use medicines Unregulated subsidised medicines Subsidised foods Subsidised devices Subsidy claiming support Frequently prescribed alternative medicines Prescribe-by-brand flag Data Model • AMT data model • • NZMT • Interoperability SCTID Pharmacode® GTIN WHO ATC PHARMAC (subsidies) Medsafe (regulatory) SNOMED CT (MP mapping) Listing strategy Capture at the point the medicine enters registration or subsidy 4

Issues • Few because we are a standalone system • Mapping issues • Absence of suitable SNOMED CT concepts because of our early listing strategy • Absence of suitable concepts for foods and devices • Absence of editorial guidance for some medicines • SNOMED CT Terms not suitable for clinical use in the NZ context 5