New Targets and New Treatments in Melanoma Brendan

New Targets and New Treatments in Melanoma Brendan D. Curti, MD Director, Melanoma Program Director Biotherapy Program Providence Cancer Center Earle A. Chiles Research Institute

Disclaimers • Earle A. Chiles Research Institute accepted grants of from BMS, Medarex, and Roche to cover costs of clinical trials. • I am neither employed nor do I have equity in any company or entity whose products/drugs will be discussed today. • Research Support: NIH, Prostate Cancer Foundation, Safeway Foundation, Kuni Foundation, Prometheus Pharmaceuticals • Speakers Bureau: Genentech, Prometheus • Advisory Board: BMS, Prometheus

Melanoma Statistics • More than 70, 000 people in the United States will get melanoma 2011. • About 8, 800 patients die per year from melanoma. • During the 1970 s, the rate of new cases of melanoma each year increased at about 6% per year. Since the 1980 s, the rate of increase has slowed to a little less than 3% per year. • Melanoma is more often found in whites who are about 10 X more likely to develop melanoma than African Americans. • Men are slightly more likely than women to have melanoma. • Melanoma rates are highest in older people, but occur in all ages. In fact, melanoma is one of the most common cancers in people under age 30.
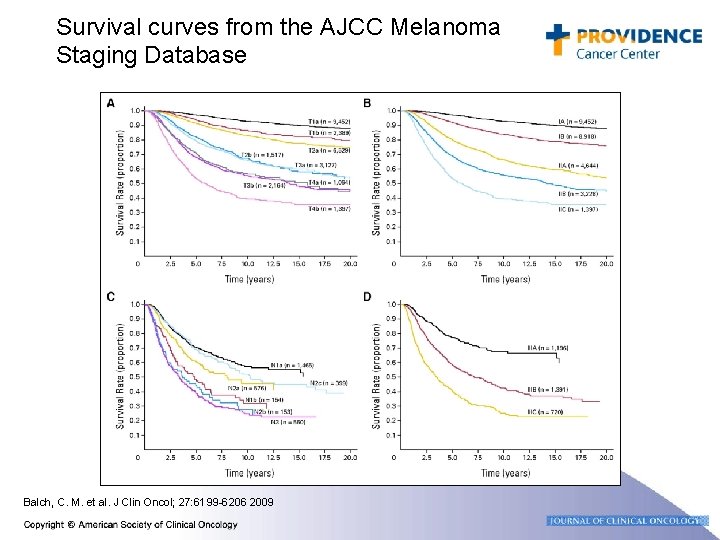
Survival curves from the AJCC Melanoma Staging Database Balch, C. M. et al. J Clin Oncol; 27: 6199 -6206 2009
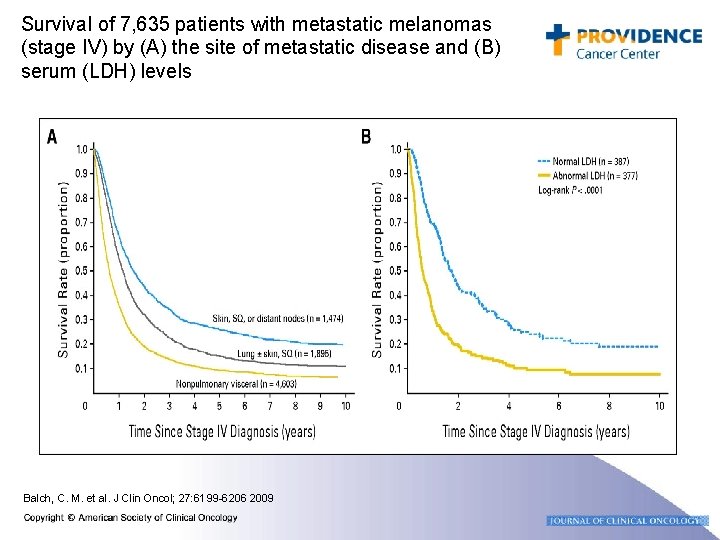
Survival of 7, 635 patients with metastatic melanomas (stage IV) by (A) the site of metastatic disease and (B) serum (LDH) levels Balch, C. M. et al. J Clin Oncol; 27: 6199 -6206 2009

TREATING METASTATIC MELANOMA The Long Dark Tunnel • No documented improvement in survival over the past 30 years • No new FDA-approved drugs for melanoma from 1992 until 2011. • Until this year, first-line therapy of questionable value over supportive care, no established second-line therapy at all, no proven value of combination regimens • Treatment of choice is a clinical trial Vern Sondak ASCO 2010
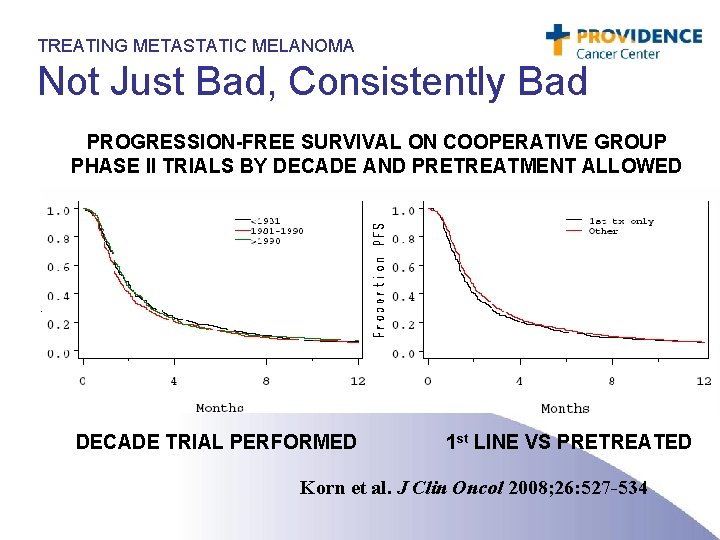
TREATING METASTATIC MELANOMA Not Just Bad, Consistently Bad PROGRESSION-FREE SURVIVAL ON COOPERATIVE GROUP PHASE II TRIALS BY DECADE AND PRETREATMENT ALLOWED DECADE TRIAL PERFORMED 1 st LINE VS PRETREATED Korn et al. J Clin Oncol 2008; 26: 527 -534
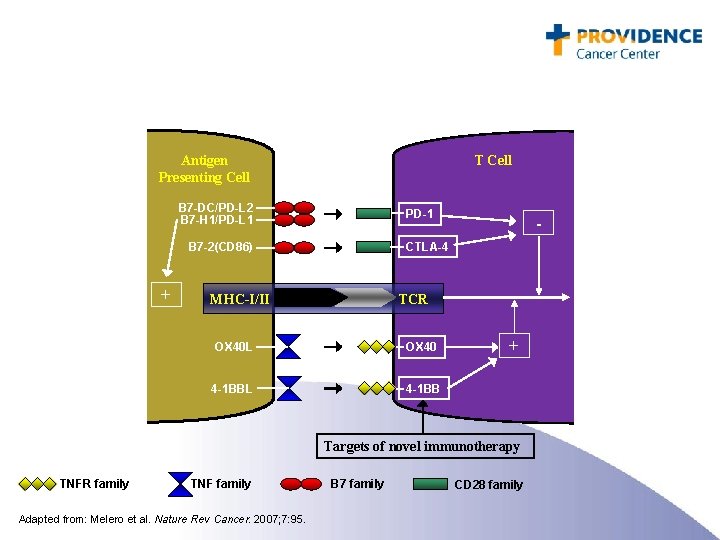
Antigen Presenting Cell T Cell B 7 -DC/PD-L 2 B 7 -H 1/PD-L 1 PD-1 CTLA-4 B 7 -2(CD 86) + - MHC-I/II TCR OX 40 L OX 40 4 -1 BBL 4 -1 BB + Targets of novel immunotherapy TNFR family TNF family Adapted from: Melero et al. Nature Rev Cancer. 2007; 7: 95. B 7 family CD 28 family
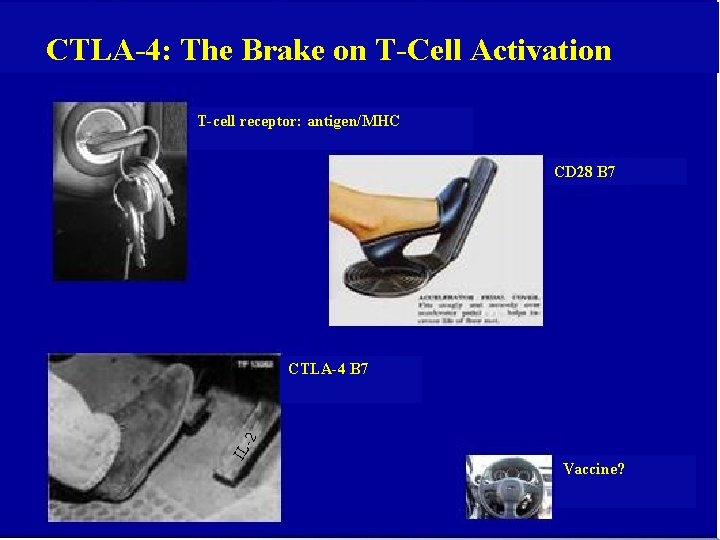
CTLA-4: The Brake on T-Cell Activation T-cell receptor: antigen/MHC CD 28 B 7 IL -2 CTLA-4 B 7 Vaccine?

Original Article Improved Survival with Ipilimumab in Patients with Metastatic Melanoma F. Stephen Hodi, M. D. , Steven J. O'Day, M. D. , David F. Mc. Dermott, M. D. , Robert W. Weber, M. D. , Jeffrey A. Sosman, M. D. , John B. Haanen, M. D. , Rene Gonzalez, M. D. , Caroline Robert, M. D. , Ph. D. , Dirk Schadendorf, M. D. , Jessica C. Hassel, M. D. , Wallace Akerley, M. D. , Alfons J. M. van den Eertwegh, M. D. , Ph. D. , Jose Lutzky, M. D. , Paul Lorigan, M. D. , Julia M. Vaubel, M. D. , Gerald P. Linette, M. D. , Ph. D. , David Hogg, M. D. , Christian H. Ottensmeier, M. D. , Ph. D. , Celeste Lebbé, M. D. , Christian Peschel, M. D. , Ian Quirt, M. D. , Joseph I. Clark, M. D. , Jedd D. Wolchok, M. D. , Ph. D. , Jeffrey S. Weber, M. D. , Ph. D. , Jason Tian, Ph. D. , Michael J. Yellin, M. D. , Geoffrey M. Nichol, M. B. , Ch. B. , Axel Hoos, M. D. , Ph. D. , and Walter J. Urba, M. D. , Ph. D. N Engl J Med Volume 363(8): 711 -723 August 19, 2010
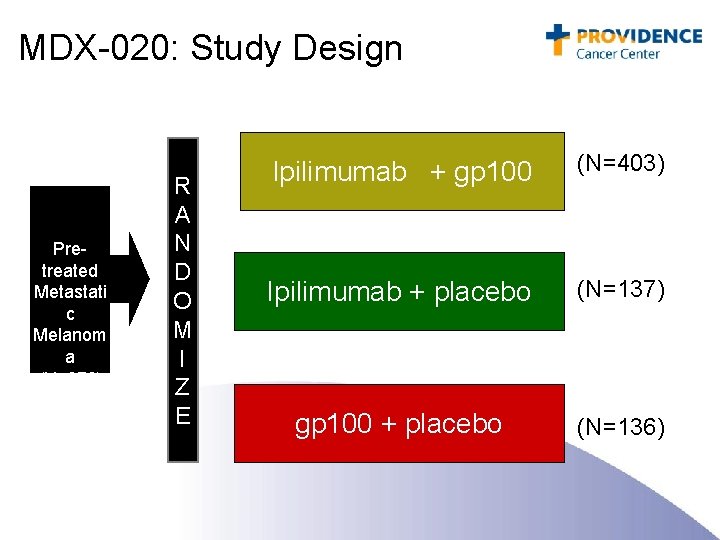
MDX-020: Study Design Pretreated Metastati c Melanom a (N=676) R A N D O M I Z E Ipilimumab + gp 100 (N=403) Ipilimumab + placebo (N=137) gp 100 + placebo (N=136)
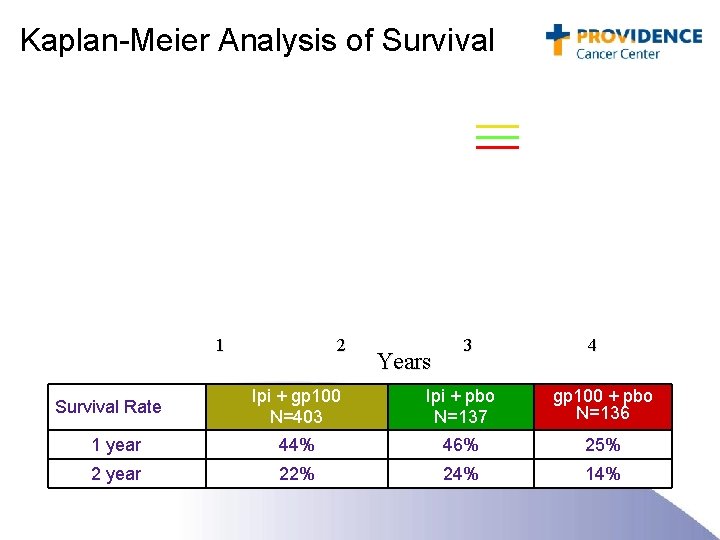
Kaplan-Meier Analysis of Survival Ipi + gp 100 Ipi alone gp 100 alone 1 2 Years 3 (A) (B) (C) 4 Ipi + gp 100 N=403 Ipi + pbo N=137 gp 100 + pbo N=136 1 year 44% 46% 25% 2 year 22% 24% 14% Survival Rate
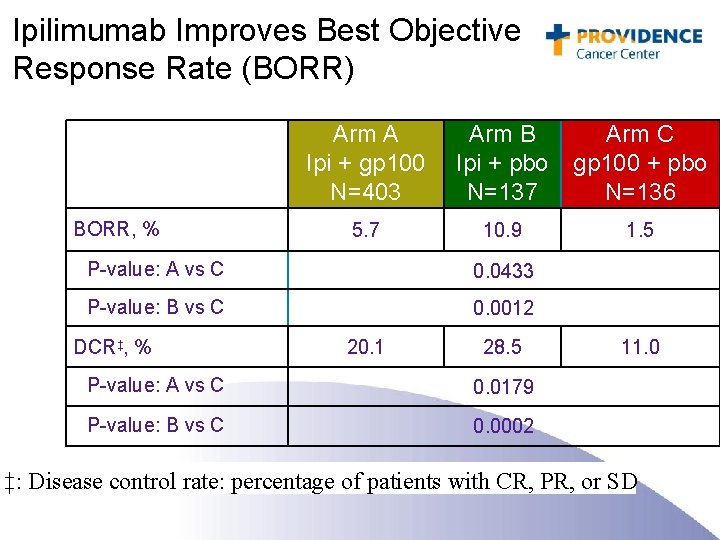
Ipilimumab Improves Best Objective Response Rate (BORR) BORR, % Arm A Ipi + gp 100 N=403 Arm B Ipi + pbo N=137 Arm C gp 100 + pbo N=136 5. 7 10. 9 1. 5 P-value: A vs C 0. 0433 P-value: B vs C 0. 0012 DCR‡, % 20. 1 28. 5 P-value: A vs C 0. 0179 P-value: B vs C 0. 0002 11. 0 ‡: Disease control rate: percentage of patients with CR, PR, or SD
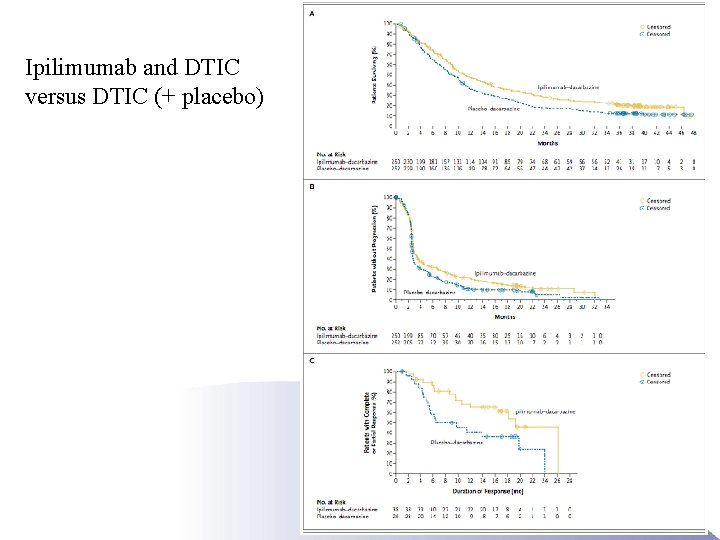
Ipilimumab and DTIC versus DTIC (+ placebo)
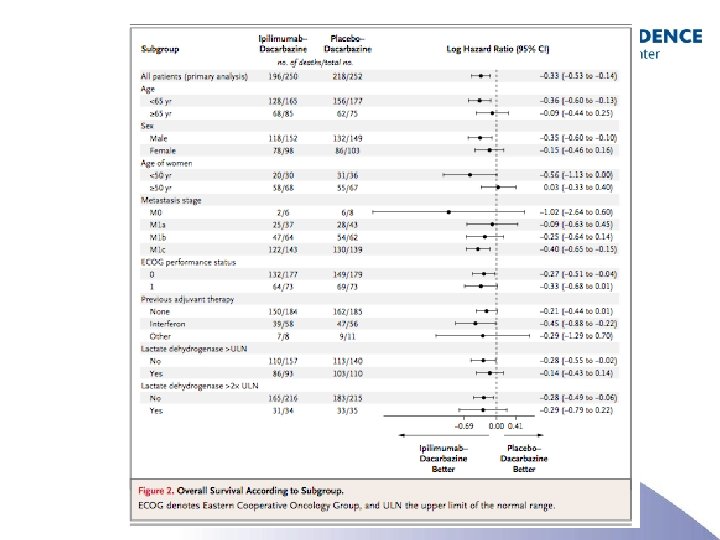

CTLA-4 Immunotherapy: Toxicity = Immune Response-related Adverse Event (IRAE) • Most common IRAEs – – Rash Diarrhea (colitis) Endocrinopathies Hepatitis • IRAEs are almost always reversible and manageable with steroids • Toxicity does not always equal response, but there does appear to be an association Weber, 2007.
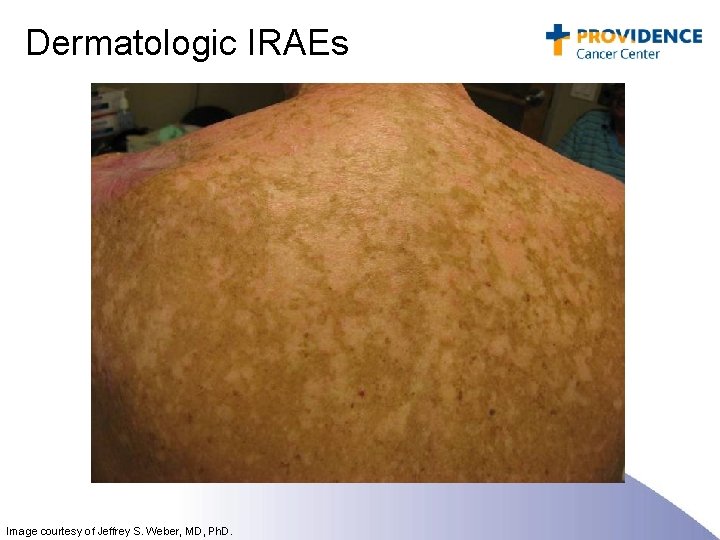
Dermatologic IRAEs Image courtesy of Jeffrey S. Weber, MD, Ph. D.
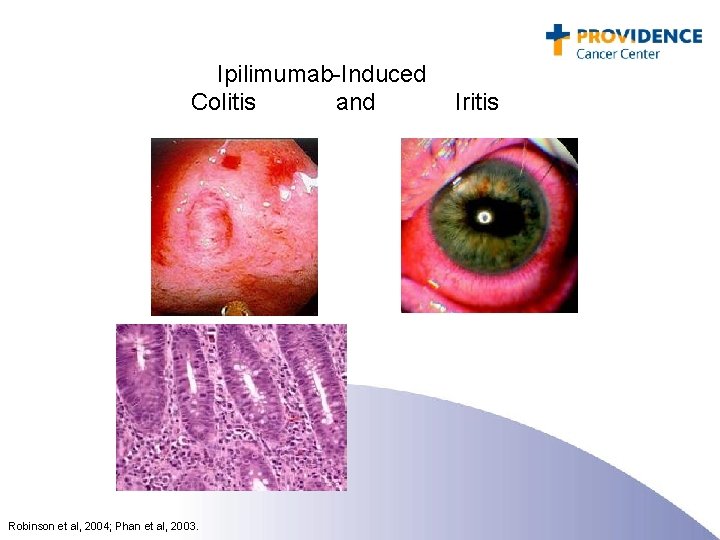
Ipilimumab-Induced Colitis and Iritis Robinson et al, 2004; Phan et al, 2003.
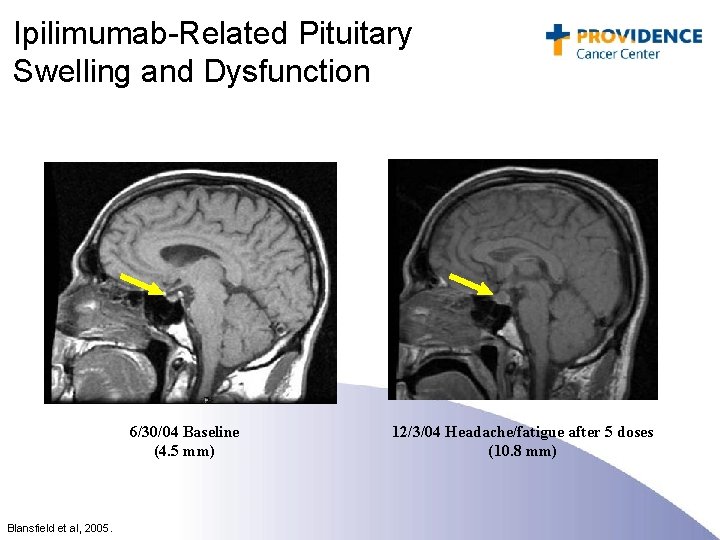
Ipilimumab-Related Pituitary Swelling and Dysfunction 6/30/04 Baseline (4. 5 mm) Blansfield et al, 2005. 12/3/04 Headache/fatigue after 5 doses (10. 8 mm)

IRAE Management • Patient education for early recognition of IRAEs • Aggressive work-up and management for moderate/severe events • Non-specific complaints may reflect endocrine (e. g. : , pituitary) toxicity • Established therapies (e. g. : , corticosteroids) are effective – Dexamethasone: 4 mg IV q 6 hrs x 7 days followed by 17 days taper. If ineffective then infliximab 5 mg IV x 1. • Algorithms established for work-up, treatment, and reporting of IRAEs • Patient wallet card and/or medical ID bracelet Beck et al, 2006.

Conclusions- Ipilimumab • Ipilimumab represents a new class of T-cell potentiators and a breakthrough for the field of immunotherapy • Further development of ipilimumab is ongoing – Treatment of a variety of cancer types – Alternative combination regimens – Refinements in dose and schedule • Approved by the FDA on March 25, 2011 – Price for 4 doses = ~$120, 000

Targeted Therapy
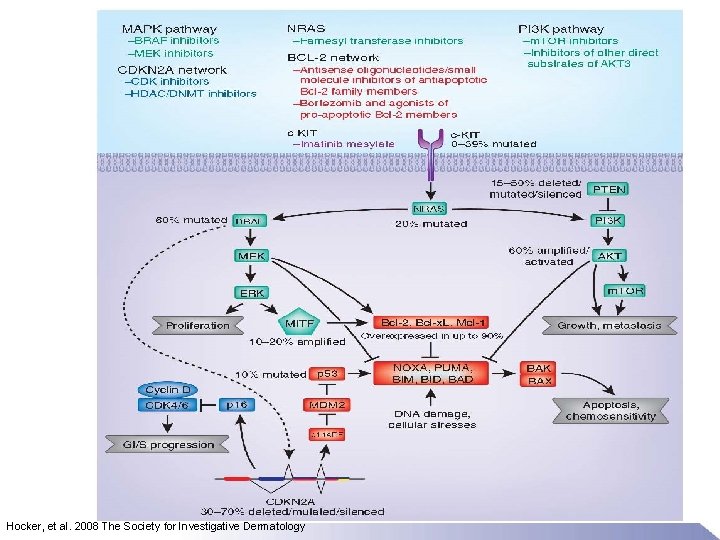
Hocker, et al. 2008 The Society for Investigative Dermatology

Relative frequency of BRAF mutations BRAF mutation location (by amino acid position and substitution) % of all BRAF mutations V 600 E 97. 3% V 600 K 1. 0% K 601 E* 0. 4% G 469 A* 0. 4% D 594 G* 0. 3% V 600 R 0. 3% L 597 V* 0. 2% *Indicates most common amino substitution, but % represents all amino acid changes reported at that position
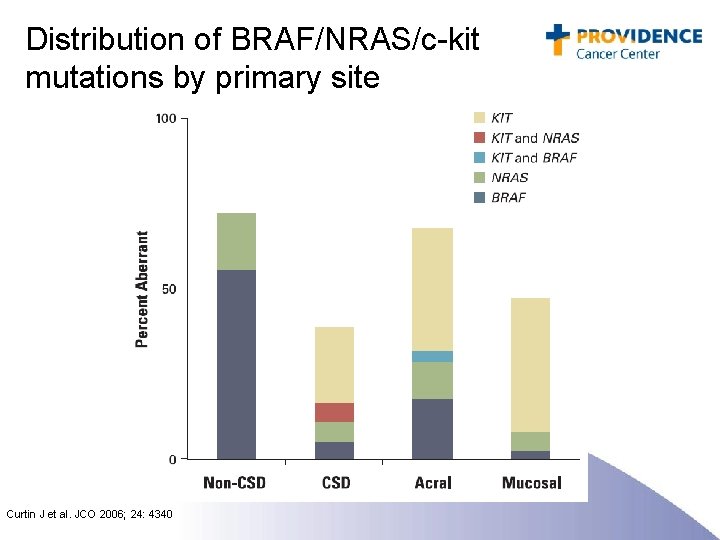
Distribution of BRAF/NRAS/c-kit mutations by primary site Curtin J et al. JCO 2006; 24: 4340

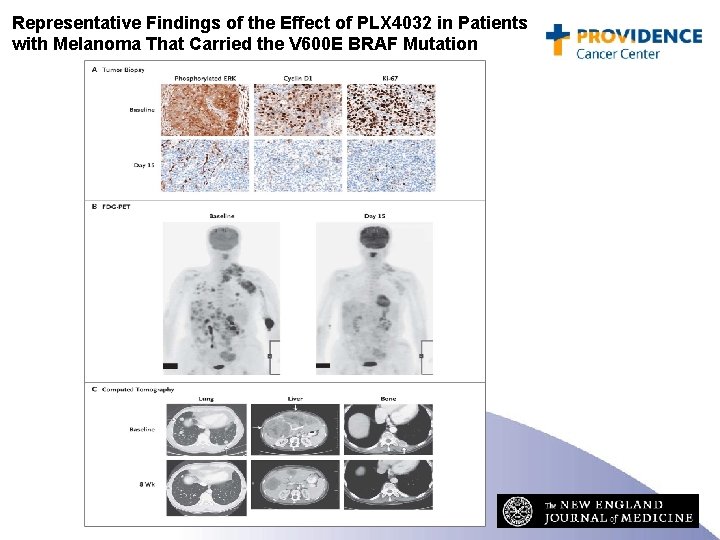
Representative Findings of the Effect of PLX 4032 in Patients with Melanoma That Carried the V 600 E BRAF Mutation Flaherty KT et al. N Engl J Med 2010; 363: 809 -819
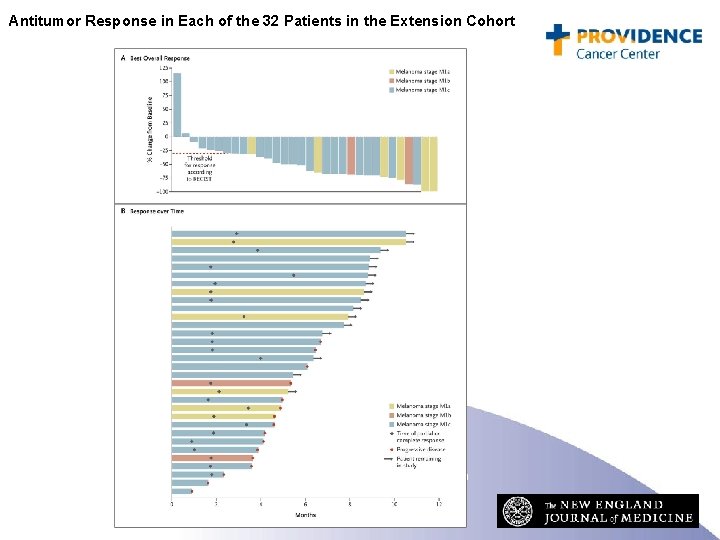
Antitumor Response in Each of the 32 Patients in the Extension Cohort Flaherty KT et al. N Engl J Med 2010; 363: 809 -819
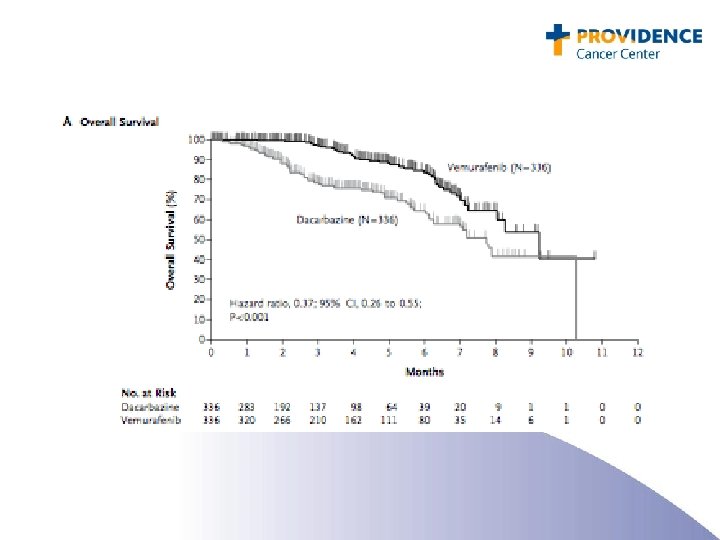
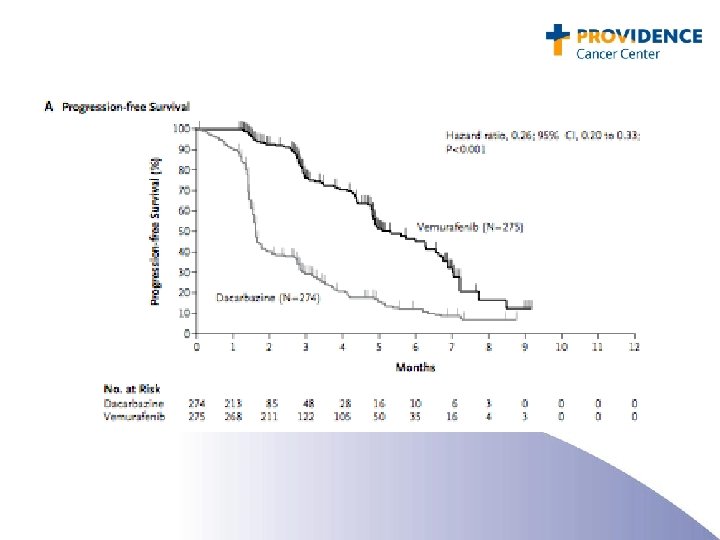

BRAF and Vemurafenib: Conclusions • Approximately 50% of melanomas contain an activating mutation in BRAF: glutamic acid for valine at amino acid 600 (the V 600 E mutation). • Side effects included rash, arthralgia, fatigue, and keratoacanthoma. • Treatment of patients with V 600 E BRAF mutated metastatic melanoma with PLX 4032 resulted in complete or partial tumor regression in the majority of patients. • Remissions do not appear to be durable. • Approved by the FDA on 8/16/2011 • Bargain price: $9800 per month ($117, 600 per year)

Patient 1 • 76 year old grandmother diagnosed in 2002 years with TXN 3 M 0 (stage 3) melanoma of left neck s/p RLND • 2004: Left axillary LN recurrence, 24/28 + on axillary dissection • May 2010: New onset dyspnea. Found to large left pleural effusion, bulky chest and abdominal adenopathy. Biopsy confirms melanoma. ECOG 2 (on a good day) • What would you do?
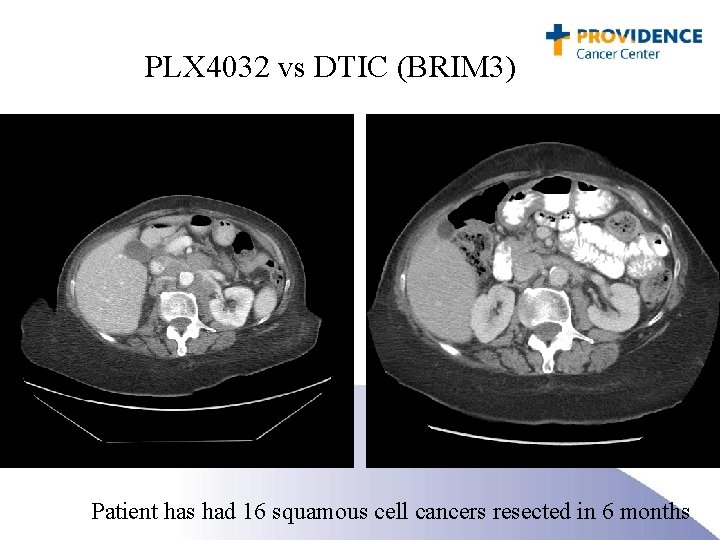
PLX 4032 vs DTIC (BRIM 3) Patient has had 16 squamous cell cancers resected in 6 months

Patient 2 • 51 year old salesman with T 2 b. N 1 M 0 melanoma on back in 2005. S/P WLE, SLN and CLND. One year “Kirkwood” IFN. • Surveillance CT shows new mediastinal and hilar adenopathy, new SQ nodule. • What would you do?
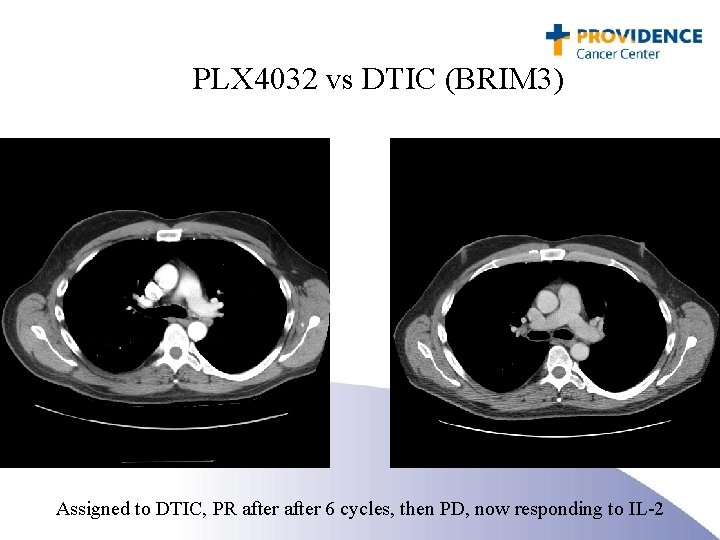
PLX 4032 vs DTIC (BRIM 3) Assigned to DTIC, PR after 6 cycles, then PD, now responding to IL-2

Patient 3 • 64 year old dentist, presents with acute abdominal pain. CT shows liver mets and adrenal tumors. Additional staging shows lung, LN, adrenal mets. LDH 8000. Biopsy confirms melanoma, PS 2 (and sliding rapidly). • What would you do?
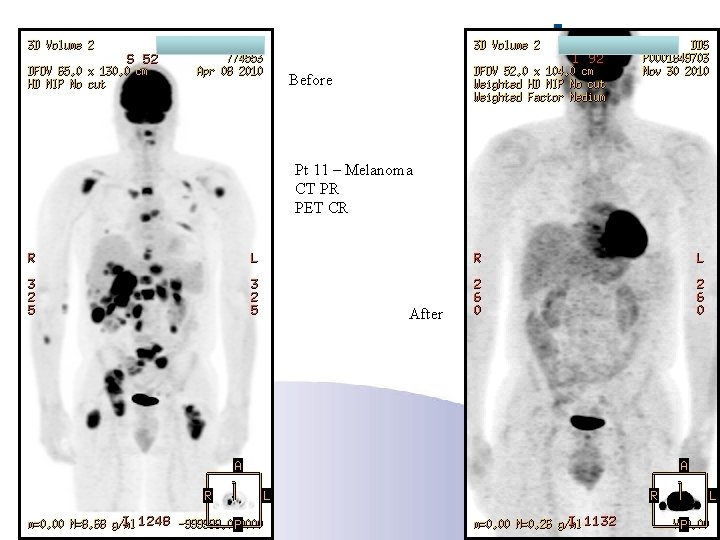
Before Pt 11 – Melanoma CT PR PET CR After
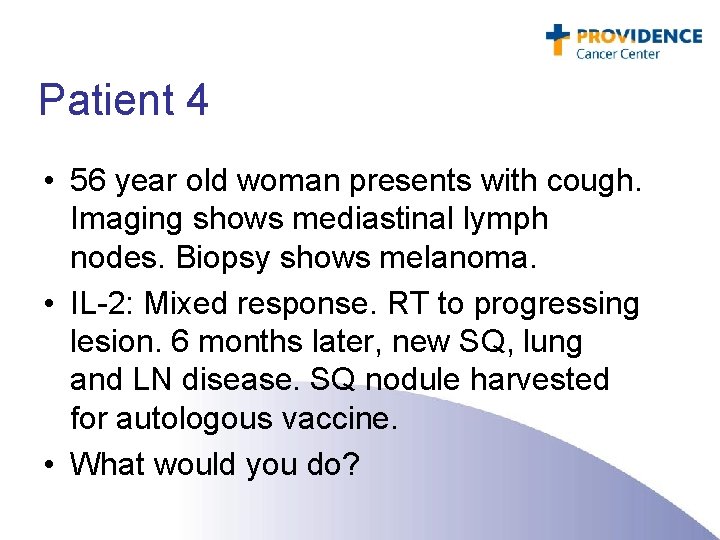
Patient 4 • 56 year old woman presents with cough. Imaging shows mediastinal lymph nodes. Biopsy shows melanoma. • IL-2: Mixed response. RT to progressing lesion. 6 months later, new SQ, lung and LN disease. SQ nodule harvested for autologous vaccine. • What would you do?
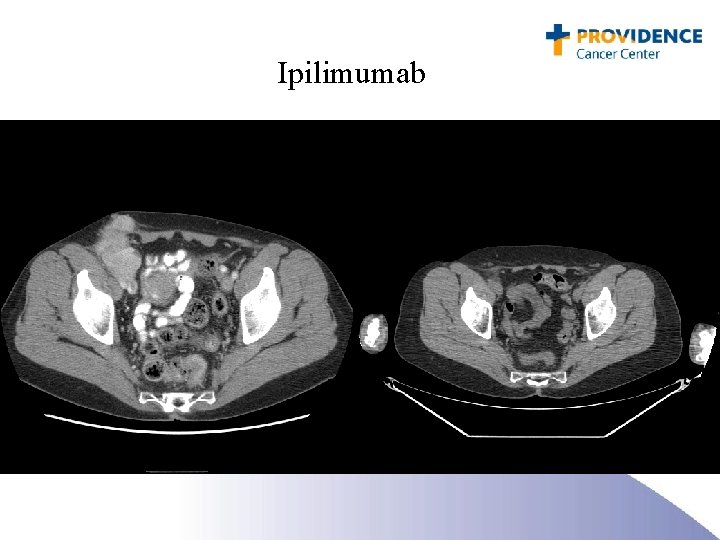
Ipilimumab

Questions for the future: • • • How excited should we be with PFS of 6 – 8 months with targeted therapies (despite the high initial response rate)? How do we sequence “targeted” therapies? We have entered the age of personalized medicine to characterize targets for melanoma treatment. Do we need to enter the age of personalized mechanisms of resistance? (and include physiologic, immunologic and other mechanisms of resistance) If BRAF resistance can (in part) be overcome with MEK inhibition, and MEK resistance can be overcome with WNT inhibition (at least in vitro), what will we need for WNT resistance? A melanoma “wiring diagram” was shown at a recent meeting with 35 nodes (connections). If we assume that there are 2 interactions from each node, and each interaction represents a path of resistance (or a path of recovery if you take the perspective of a melanoma cell), then there are 235 (= 3. 4 x 1010) potential resistance pathways. How easy will it be to address these several pathways of resistance in an individual? (and even if my estimate is wrong by 99. 99999%, there are 34 resistance pathways to manage).
- Slides: 40