New Pneumococcal Vaccines Ray Borrow Vaccine Evaluation Unit

New Pneumococcal Vaccines Ray Borrow Vaccine Evaluation Unit, Health Protection Agency, Manchester, UK ray. borrow@hpa. org. uk 21 st April 2010

Sept 2006 UK schedule 2 months DTa. P/IPV/Hib + pneumococcal vaccine 3 months DTa. P/IPV/Hib + MCC vaccine 4 months DTa. P/IPV/Hib + MCC + pneumococcal vaccine (MCC can be given at 5 months) 12 months Hib/MCC 13 months MMR + pneumococcal vaccine
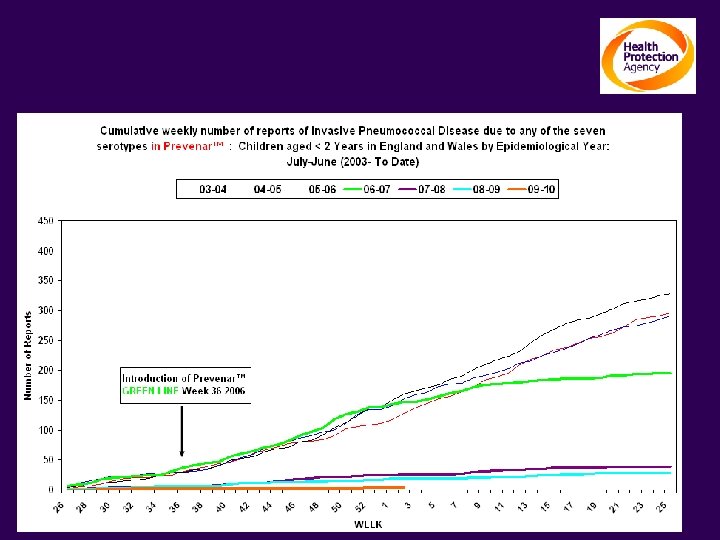
The impact on Invasive Pneumococcal Disease Children Under 2 yrs: Serotypes contained in Prevenar TM
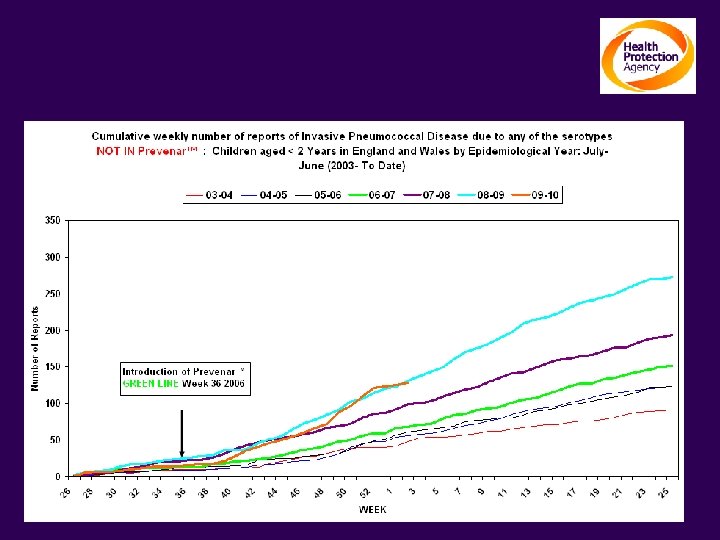
The impact on Invasive Pneumococcal Disease Children Under 2 yrs: Serotypes NOT contained in Prevenar TM
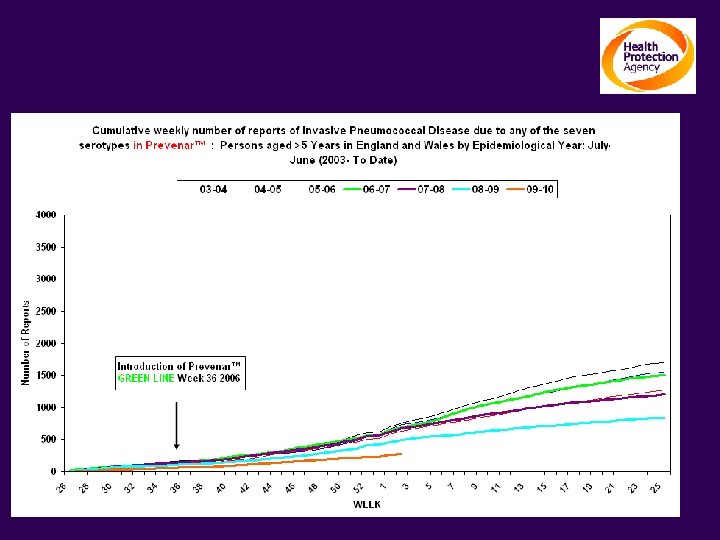
The impact on Invasive Pneumococcal Disease Persons ≥ 5 yrs: Serotypes contained in Prevenar TM
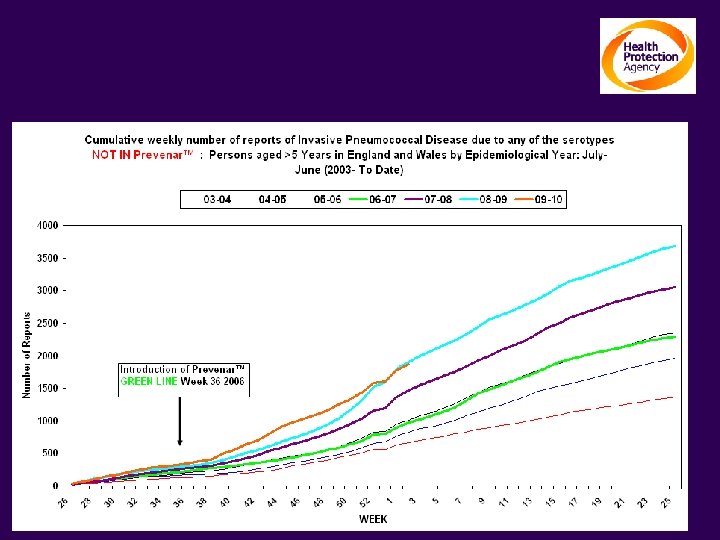
The impact on Invasive Pneumococcal Disease Persons ≥ 5 yrs: Serotypes NOT contained in Prevenar TM

IPD incidence in England Wales by serotype: children < 5 yrs old 2/4/13 month schedule Change 2008/9 vs 2005/6; All IPD -40% (-48, - 30) VT -92% (-94, -89); NVT +88% (+23, +187) Change 2008/9 vs predicted incidence in 2008/9 All IPD -51% (-60, -41) VT -93% (-95, -90); NVT +35% (-22, +134)
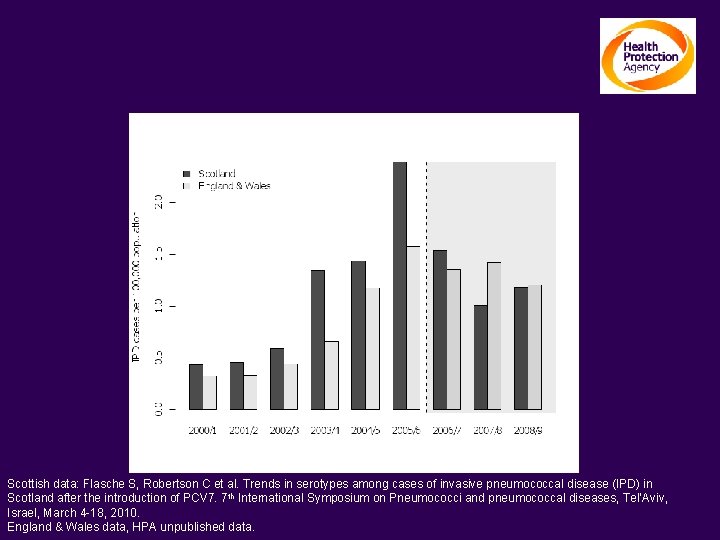
Long term trends in serotypes unrelated to PCV 7, for example ST 1 in UK, changes mainly in 5 -64 year olds Scottish data: Flasche S, Robertson C et al. Trends in serotypes among cases of invasive pneumococcal disease (IPD) in Scotland after the introduction of PCV 7. 7 th International Symposium on Pneumococci and pneumococcal diseases, Tel’Aviv, Israel, March 4 -18, 2010. England & Wales data, HPA unpublished data.

Annual number of serotypes pre PCV 7 (average 2004/5 & 2005/6) Vs post-PCV 7 year 2008/9 by vaccine category < 5 year olds: England Wales
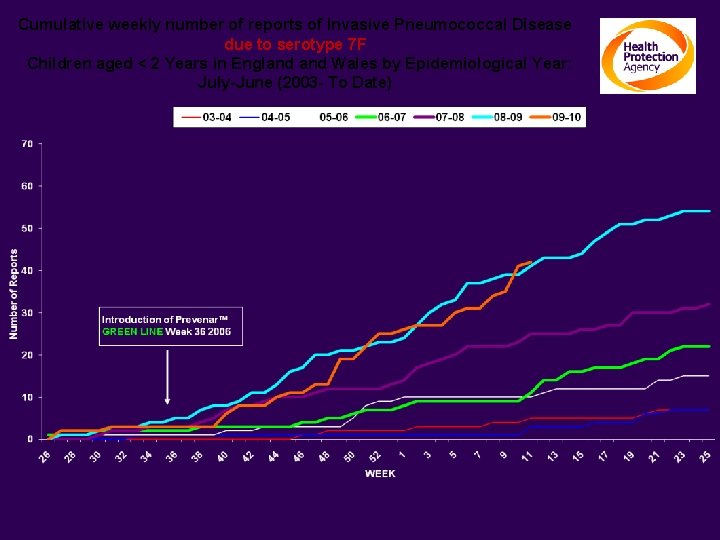
Cumulative weekly number of reports of Invasive Pneumococcal Disease due to serotype 7 F Children aged < 2 Years in England Wales by Epidemiological Year: July-June (2003 - To Date)
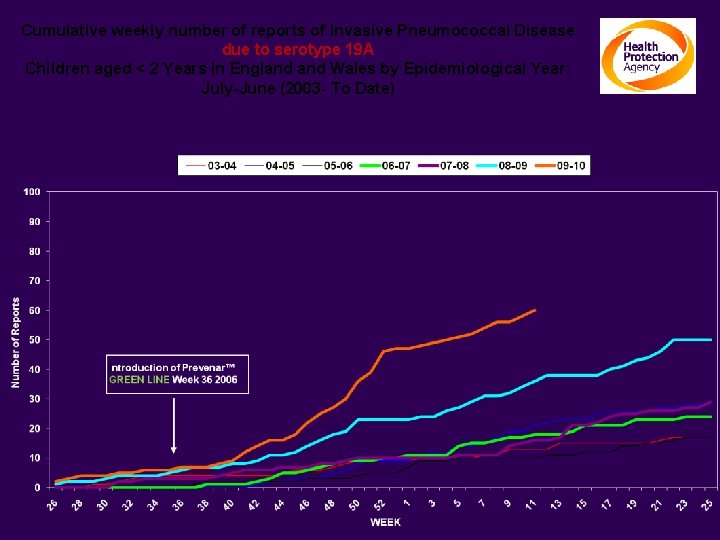
Cumulative weekly number of reports of Invasive Pneumococcal Disease due to serotype 19 A Children aged < 2 Years in England Wales by Epidemiological Year: July-June (2003 - To Date)
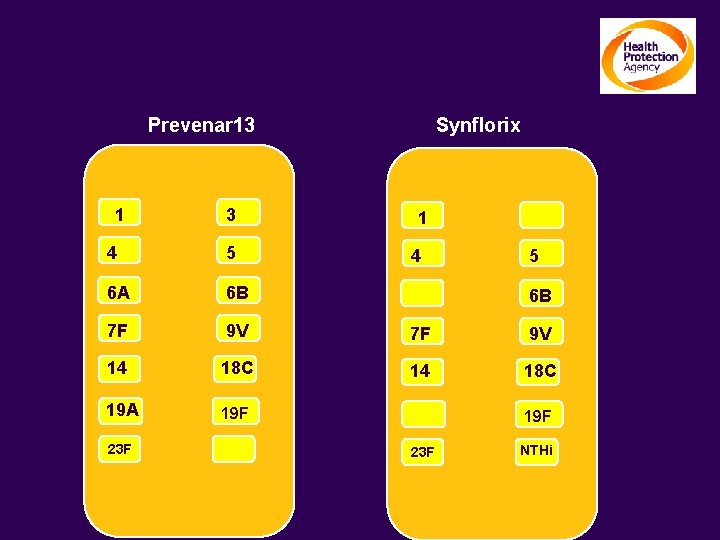
Higher valency pneumococcal conjugates Prevenar 13 1 3 Synflorix 1 4 5 6 A 6 B 7 F 9 V 14 18 C 19 A 19 F 23 F 4 5 6 B 19 F 23 F NTHi
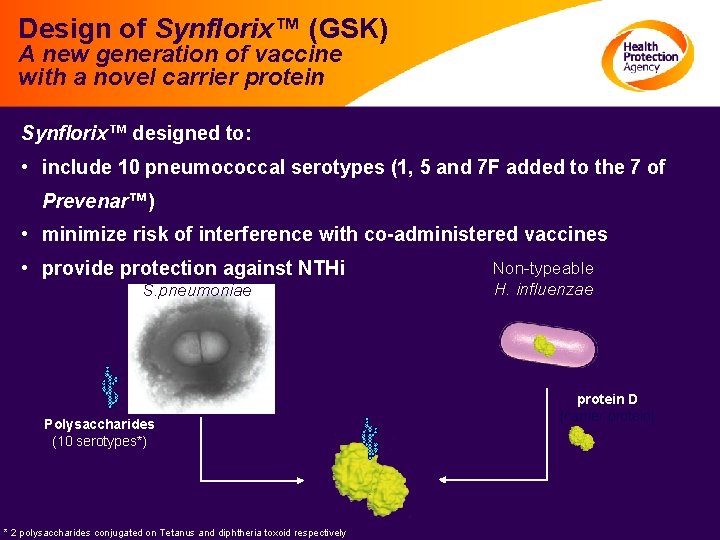
Design of Synflorix™ (GSK) A new generation of vaccine with a novel carrier protein Synflorix™ designed to: • include 10 pneumococcal serotypes (1, 5 and 7 F added to the 7 of Prevenar™) • minimize risk of interference with co-administered vaccines • provide protection against NTHi S. pneumoniae Polysaccharides (10 serotypes*) * 2 polysaccharides conjugated on Tetanus and diphtheria toxoid respectively Non-typeable H. influenzae protein D [carrier protein]

NTHi Protein D • • • Surface exposed 1 Highly Conserved Expressed in all Hi and NTHi strains tested 1 Genetically stable 2 Virulence factor Contribute to the inhibition of ciliary beating 3 Important factor in otitis media 4, 5 Anti-PD antibodies are protective in animal models 5 -7 Prevents AOM in Chinchilla model, also as carrier protein 5, 6 Increase bacterial lung clearance in Rats models 7 Immunogenic in Humans 1, 8 1. Akkoyunlu et al. 1991 ; 2. Janson, unpublished ; 3. Janson H et al. J Infect Dis. 1999; 4. Janson et al. 1994; 5. Bakaletz Infect & Imm 1999; 6. Novotny 2006; 7. Poolman Vaccine 2000; 8. Prymula et al. , Lancet 2006
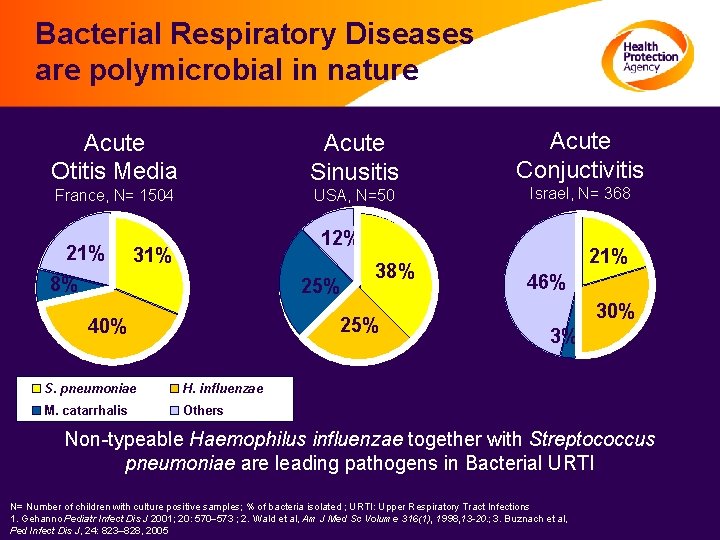
Bacterial Respiratory Diseases are polymicrobial in nature Acute Otitis Media Acute Sinusitis France, N= 1504 USA, N=50 21% Acute Conjuctivitis Israel, N= 368 12% 31% 8% 25% 38% 25% 40% S. pneumoniae H. influenzae M. catarrhalis Others 21% 46% 30% 3% Non-typeable Haemophilus influenzae together with Streptococcus pneumoniae are leading pathogens in Bacterial URTI N= Number of children with culture positive samples; % of bacteria isolated ; URTI: Upper Respiratory Tract Infections 1. Gehanno Pediatr Infect Dis J 2001; 20: 570 573 ; 2. Wald et al, Am J Med Sc Volume 316(1), 1998, 13 -20. ; 3. Buznach et al, Ped Infect Dis J, 24: 823– 828, 2005

Otitis media efficacy: POET Study Double blind, randomized (1: 1) study in Czech and Slovak Republics 11 -v Pneumococcal conjugate prototype vaccine with H. influenzae Protein D as carrier 1, 3, 4, 5, 6 B, 7 F, 9 V, 14, 18 C, 19 F, 23 F each conjugated to 1 µg of protein D
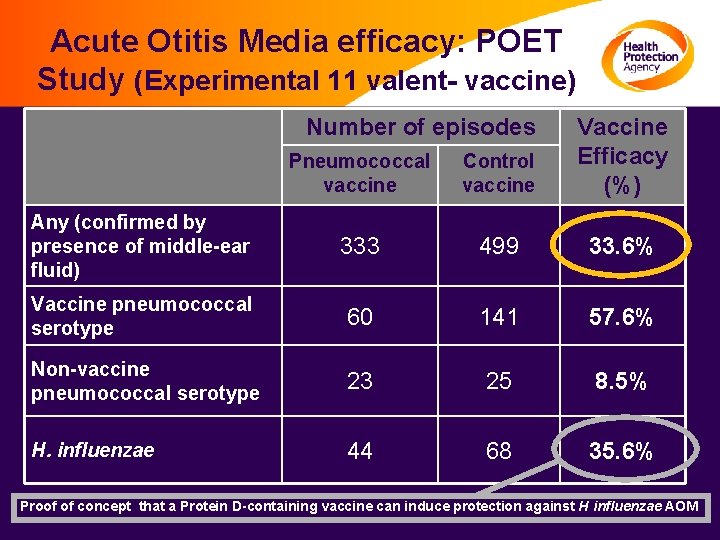
Acute Otitis Media efficacy: POET Study (Experimental 11 valent- vaccine) Number of episodes Pneumococcal vaccine Control vaccine Vaccine Efficacy (%) Any (confirmed by presence of middle-ear fluid) 333 499 33. 6% Vaccine pneumococcal serotype 60 141 57. 6% Non-vaccine pneumococcal serotype 23 25 8. 5% H. influenzae 44 68 35. 6% Proof of concept that a Protein D-containing vaccine can induce protection against H influenzae AOM Prymula R, et al. Lancet 2006; 367: 740– 748.
Serotype 3 Poolman J. et al. Vaccine 2009; 27: 3213 -22.
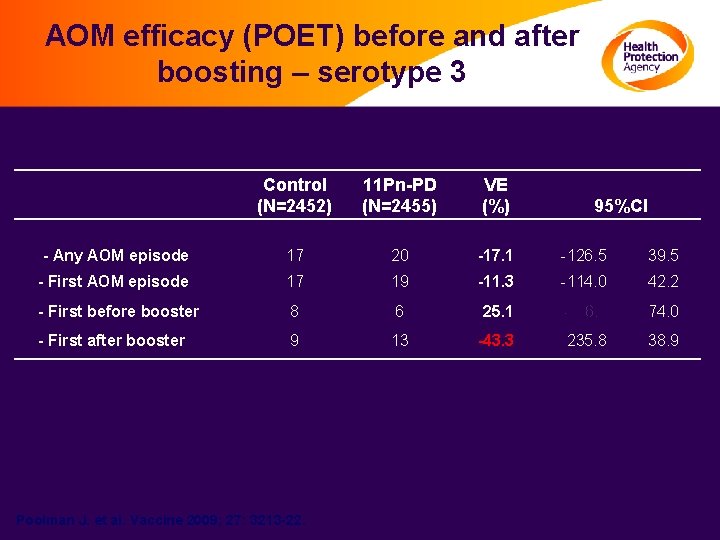
AOM efficacy (POET) before and after boosting – serotype 3 Control (N=2452) 11 Pn-PD (N=2455) VE (%) - Any AOM episode 17 20 -17. 1 -126. 5 39. 5 - First AOM episode 17 19 -11. 3 -114. 0 42. 2 - First before booster 8 6 25. 1 -116. 2 74. 0 - First after booster 9 13 -43. 3 -235. 8 38. 9 Poolman J. et al. Vaccine 2009; 27: 3213 -22. 95%CI
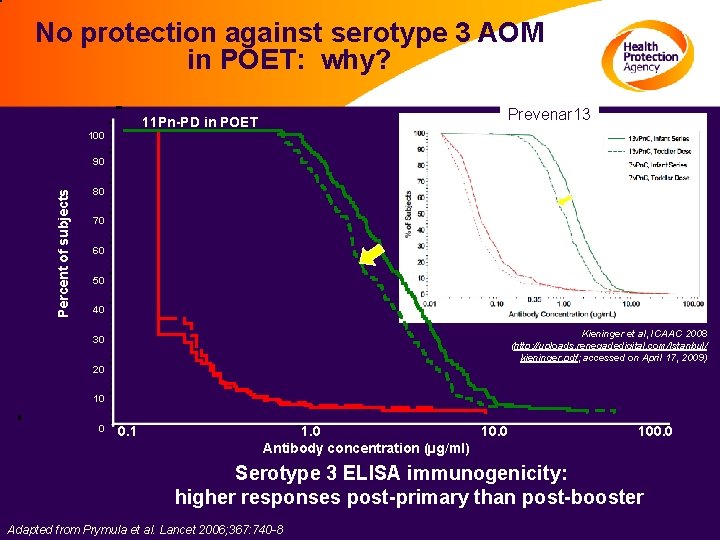
No protection against serotype 3 AOM in POET: why? Prevenar 13 11 Pn-PD in POET 100 11 Pn-PD post-primary Percent of subjects 90 80 HAV post-primary 70 60 50 40 Kieninger et al, ICAAC 2008 (http: //uploads. renegadedigital. com/Istanbul/ kieninger. pdf; accessed on April 17, 2009) 30 20 10 0 0. 1 1. 0 10. 0 Antibody concentration (µg/ml) 100. 0 Serotype 3 ELISA immunogenicity: higher responses post-primary than post-booster Adapted from Prymula et al. Lancet 2006; 367: 740 -8

Vaccination with PPV 23 does not protect against serotype 3 disease in the elderly Serotype 3 efficacy against IPD: PPV 23 vaccine Recent England & Wales data for serotype 3 VE = -29% [-73% to -4%] Statistical significant negative effect http: //www. advisorybodies. doh. gov. uk/jcvi/Pneumococcal_subgroup_minutes_15_January_2009_v 2. pdf


Prevenar 13 (Pfizer) 13 serotypes all conjugated to CRM 197 carrier protein and adsorbed on aluminium phosphate. 2. 2 µg of each serotype, except serotype 6 B (4. 4 µg) Licensed for a 2 dose primary series e. g. 2, 4 months of age with third dose recommended between 11 and 15 months. For unvaccinated children aged 12 to 23 months, the SPC recommends 2 doses, 2 months apart; the Green book, 1 dose. For children aged 2 to 5 years, both SPC and Green book recommend 1 dose.

From Prevenar to Prevenar 13 Infants should complete their course with Prevenar 13. 2 months PCV 7 PCV 13 4 months PCV 7 PCV 13 13 months PCV 7 PCV 13
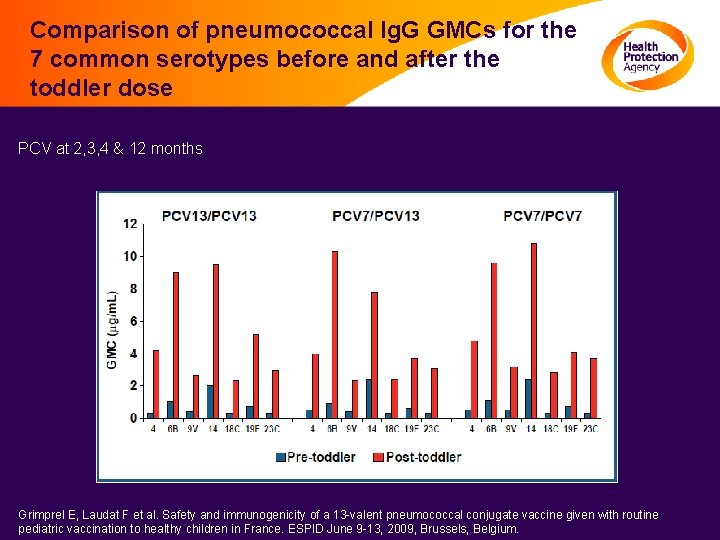
Comparison of pneumococcal Ig. G GMCs for the 7 common serotypes before and after the toddler dose PCV at 2, 3, 4 & 12 months Grimprel E, Laudat F et al. Safety and immunogenicity of a 13 -valent pneumococcal conjugate vaccine given with routine pediatric vaccination to healthy children in France. ESPID June 9 -13, 2009, Brussels, Belgium.
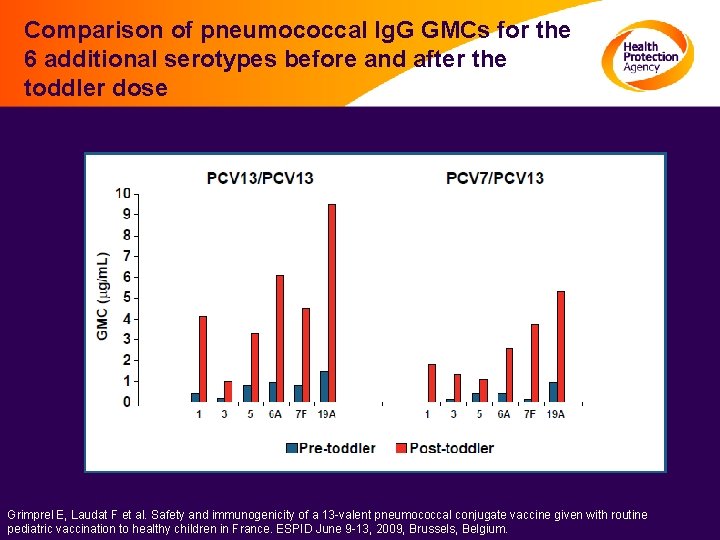
Comparison of pneumococcal Ig. G GMCs for the 6 additional serotypes before and after the toddler dose Grimprel E, Laudat F et al. Safety and immunogenicity of a 13 -valent pneumococcal conjugate vaccine given with routine pediatric vaccination to healthy children in France. ESPID June 9 -13, 2009, Brussels, Belgium.
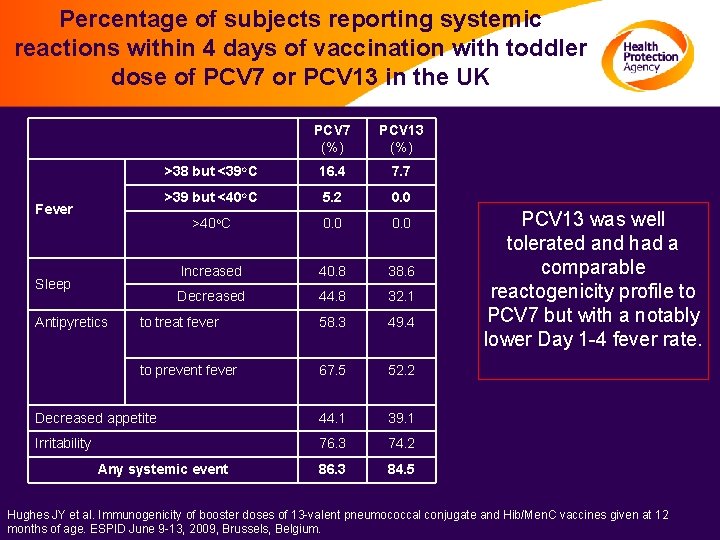
Percentage of subjects reporting systemic reactions within 4 days of vaccination with toddler dose of PCV 7 or PCV 13 in the UK PCV 7 (%) PCV 13 (%) >38 but <39 o. C 16. 4 7. 7 >39 but <40 o. C 5. 2 0. 0 >40 o. C 0. 0 Increased 40. 8 38. 6 Decreased 44. 8 32. 1 to treat fever 58. 3 49. 4 to prevent fever 67. 5 52. 2 Decreased appetite 44. 1 39. 1 Irritability 76. 3 74. 2 86. 3 84. 5 Fever Sleep Antipyretics Any systemic event PCV 13 was well tolerated and had a comparable reactogenicity profile to PCV 7 but with a notably lower Day 1 -4 fever rate. Hughes JY et al. Immunogenicity of booster doses of 13 -valent pneumococcal conjugate and Hib/Men. C vaccines given at 12 months of age. ESPID June 9 -13, 2009, Brussels, Belgium.
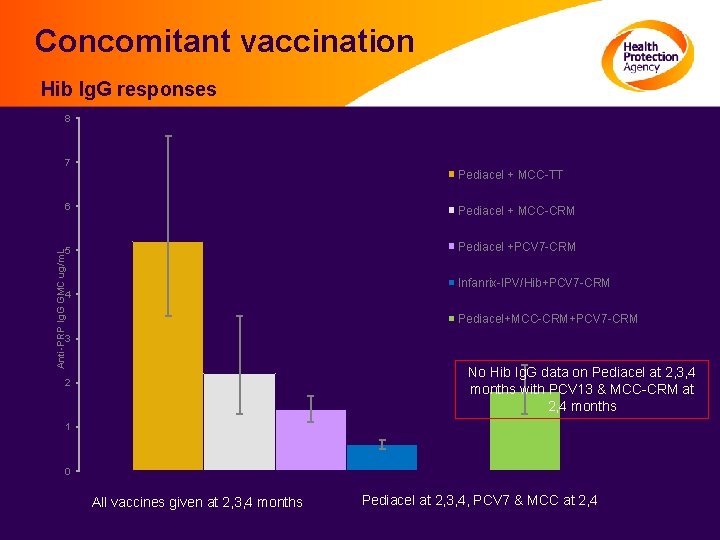
Concomitant vaccination Hib Ig. G responses 8 7 Pediacel + MCC-TT Pediacel + MCC-CRM 5 Pediacel +PCV 7 -CRM Anti-PRP Ig. G GMC ug/m. L 6 Infanrix-IPV/Hib+PCV 7 -CRM 4 Pediacel+MCC-CRM+PCV 7 -CRM 3 No Hib Ig. G data on Pediacel at 2, 3, 4 months with PCV 13 & MCC-CRM at 2, 4 months 2 1 0 All vaccines given at 2, 3, 4 months Pediacel at 2, 3, 4, PCV 7 & MCC at 2, 4

Vaccination schedule for those in a clinical risk group (Green book) Vaccine given and when to immunise Patient age at presentation Prevenar 13 (PCV) Pneumovax At-risk children 2 months to under 12 months of age. Vaccination according the routine immunisation schedule at 2, 4, 13 months of age. One dose after the second birthday. At-risk children second birthday 2 months to under 12 months of age who have asplenia or splenic dysfunction or who are Immunosuppressed. Vaccination according to the routine immunisation schedule at 2, 4 13 months of age. One dose after the second birthday. At-risk children 12 months to under 5 years of age. One dose. At-risk children 12 months to under 5 years of age who have asplenia or splenic dysfunction or who are immunosuppressed. Two doses, with an interval of 2 months between doses. One dose after the second birthday after the final dose of PCV. One dose after the second birthday and at Least 2 months after the final dose of PCV. At-risk children aged over 5 years and at-risk adults. PCV is not recommended. One dose.

Proposed changes to Green Book for clinical at risk groups: HIV Change from Pneumovax to 2 x PCV BMT Change from Pneumovax to 2 x PCV Chronic renal PCV every 5 years

Pneumovax Vaccine efficacy estimates for each age cohort 62 to 73 years 40% (13% to 59%; 95% CI) 74 to 79 years 25% (-12% to 49%; 95% CI) 80+ years 8% (-21% to 30%; 95% CI) Vaccine efficacy = 23% (6% to 36%; 95% CI) for all age groups. Vaccination of older people commenced in 2003, starting with people 80 years and over in August 2003, people aged from 75 years in April 2004 and from 65 years in April 2005. These cohorts were used to estimating vaccine effectiveness and the period used for analysis.
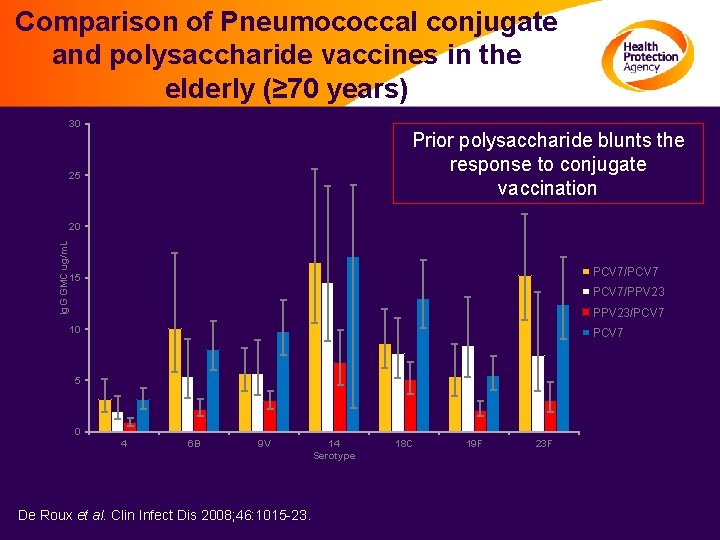
Comparison of Pneumococcal conjugate and polysaccharide vaccines in the elderly (≥ 70 years) 30 Prior polysaccharide blunts the response to conjugate vaccination 25 Ig. G GMC ug/m. L 20 PCV 7/PCV 7 15 PCV 7/PPV 23/PCV 7 10 PCV 7 5 0 4 6 B 9 V De Roux et al. Clin Infect Dis 2008; 46: 1015 -23. 14 Serotype 18 C 19 F 23 F
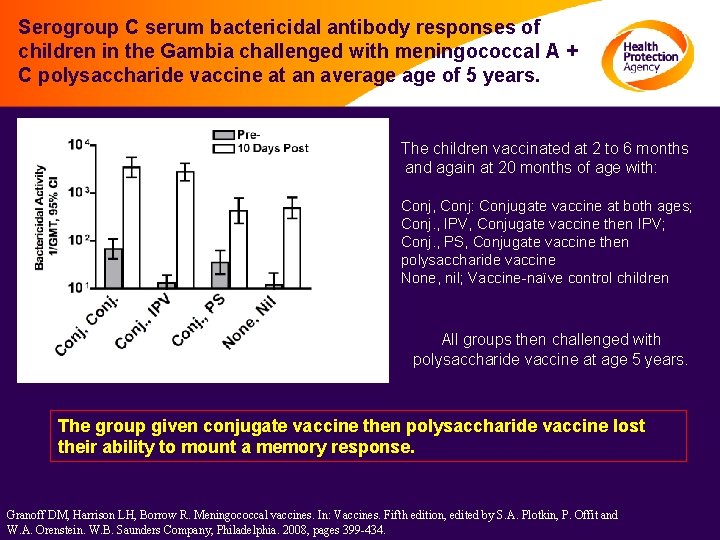
Serogroup C serum bactericidal antibody responses of children in the Gambia challenged with meningococcal A + C polysaccharide vaccine at an average of 5 years. The children vaccinated at 2 to 6 months and again at 20 months of age with: Conj, Conj: Conjugate vaccine at both ages; Conj. , IPV, Conjugate vaccine then IPV; Conj. , PS, Conjugate vaccine then polysaccharide vaccine None, nil; Vaccine-naïve control children All groups then challenged with polysaccharide vaccine at age 5 years. The group given conjugate vaccine then polysaccharide vaccine lost their ability to mount a memory response. Granoff DM, Harrison LH, Borrow R. Meningococcal vaccines. In: Vaccines. Fifth edition, edited by S. A. Plotkin, P. Offit and W. A. Orenstein. W. B. Saunders Company, Philadelphia. 2008, pages 399 -434.

Conclusions & more questions • Despite replacement disease, the reduction in IPD in children is substantial. • The phenomenon of replacement needs to be confronted and investigated. • Role of even higher valency conjugate vaccines? • Role of protein based pneumococcal vaccines? • Future of 23 -valent pneumococcal polysaccharide vaccine?

Acknowledgements HPA Cf. I: Liz Miller, Stefan Flasche, Pauline Kaye, Rashmi Malkani, Yoon Choi, Nick Andrews, Mary Slack, Robert George. HPA Manchester: Elaine Stanford. Pfizer: Paul Balmer GSK: Jan Poolman
- Slides: 35