New NIH Human Subjects Clinical Trials Information Ayisha

New NIH Human Subjects & Clinical Trials Information Ayisha Ali & Nadia Hajjar The goal of the NIH in updating these forms was to consolidate human subjects, inclusion enrollment, and clinical trial information previously collected across multiple agency forms

What is the difference between clinical research and a clinical trial? • Clinical research includes all research involving human participants. It does not include secondary studies using existing biological specimens or data collected without identifiers or data that are publicly available. • Clinical trials are clinical research studies involving human participants assigned to an intervention in which the study is designed to evaluate the effect(s) of the intervention on the participant and the effect being evaluated is a health-related biomedical or behavioral outcome

How can researchers determine whether a proposed study is a clinical trial? The following questions should be used to determine whether a study meets the NIH clinical trial definition: ▪ Does the study involve human participants? ▪ Are the participants prospectively assigned to an intervention? ▪ Is the study designed to evaluate the effect of the intervention on the participants? ▪ Is the effect being evaluated a health-related biomedical or behavioral outcome? Ø Ø Ø If the answers are all “yes, ” the study is a clinical trial. If any answers are “no, ” the study is not a clinical trial If the PI is unsure, encourage the PI to reach out to the program officer. * If the PI is still unsure after answering these questions, encourage them to reach out to the program officer for confirmation 2

New Human Subject Form Ø New form is included in all applications (whether or not human subjects or clinical trials are involved) Ø Required form fields vary based on a number of factors, including: Ø Whether study is delayed onset Ø Announcement-specific instructions Ø Human subject exemptions Ø Whether study involves a clinical trial 3

R & R Other Project Information Form Ø NIH will continue to collect Human Subjects information on the R & R Other Project Information form 4
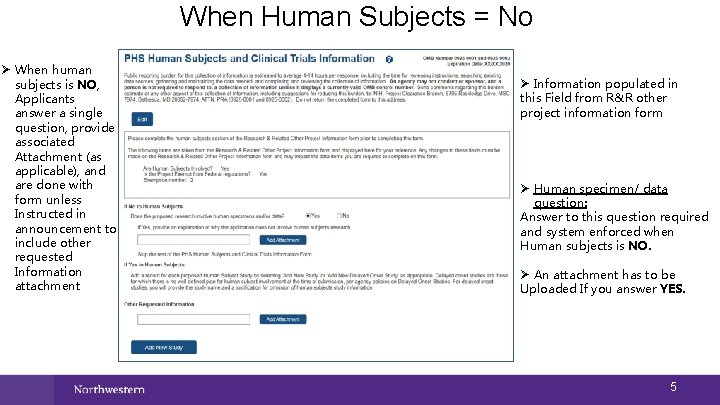
When Human Subjects = No Ø When human subjects is NO, Applicants answer a single question, provide associated Attachment (as applicable), and are done with form unless Instructed in announcement to include other requested Information attachment Ø Information populated in this Field from R&R other project information form Ø Human specimen/ data question: Answer to this question required and system enforced when Human subjects is NO. Ø An attachment has to be Uploaded If you answer YES. 5

When Human Subjects = Yes (not delayed onset) Ø Full Study records require quite a bit of data Ø New data collection leads applicants through clinical trial information collection requirements Ø Data collection expanded for new policies Check FOA to determine if attachment is needed If yes to Human Subject, add Study Title. Study title must be unique with in the application. 6
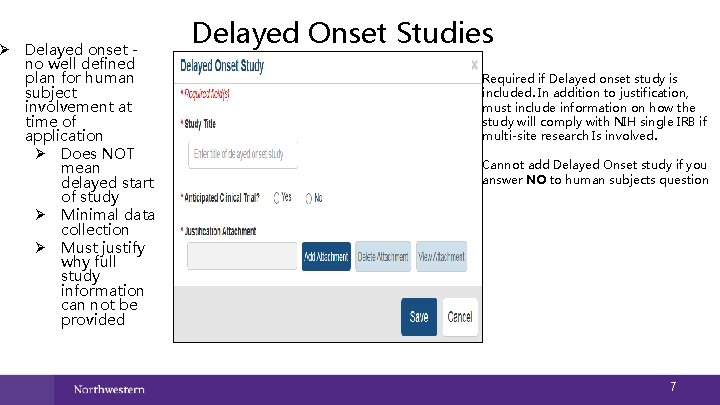
Ø Delayed onset no well defined plan for human subject involvement at time of application Ø Does NOT mean delayed start of study Ø Minimal data collection Ø Must justify why full study information can not be provided Delayed Onset Studies Required if Delayed onset study is included. In addition to justification, must include information on how the study will comply with NIH single IRB if multi-site research Is involved. Cannot add Delayed Onset study if you answer NO to human subjects question 7

Study Record: Section 1 -Basic Information Exemption 4 - Research involving the collection or study of existing data or specimens if publicly available or information recorded that subjects cannot be identified 8

Study Record: Section 2 - Study Population • Characteristics • Conditions-At least 1 entry is required but the PI can provide up to 20 study population characteristics. If available, use appropriate descriptors from (Me. SH) • Inclusion- Women, Minorities and Children all together • Recruitment and Retention Plan. Describe how you will recruit and retain participants. Address both planned and proposed recruitment Study Timeline. Provide a description or diagram describing the study timeline 9

Inclusion Enrollment Report Planned enrollment must be completed when answer to “using existing dataset” question is NO If YES then only complete cumulative enrollment. 10

Study Record: Section 3 Protection and Monitoring Plans Ø All of Section 3 is required for all studies involving human subjects unless noted otherwise Ø Data and Safety monitoring plan is only required if you answered “Yes” to all the questions in the clinical trial questionnaire Ø If not-clinical, but has human subjects, the overall structure of the study team is optional Ø If Yes to multi-site, single IRB plan must be included 11
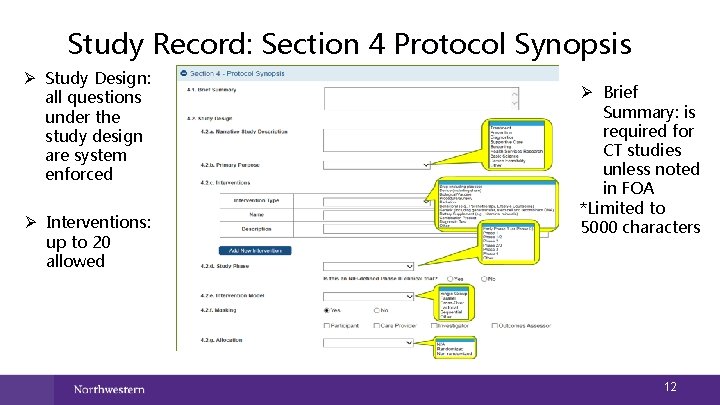
Study Record: Section 4 Protocol Synopsis Ø Study Design: all questions under the study design are system enforced Ø Interventions: up to 20 allowed Ø Brief Summary: is required for CT studies unless noted in FOA *Limited to 5000 characters 12
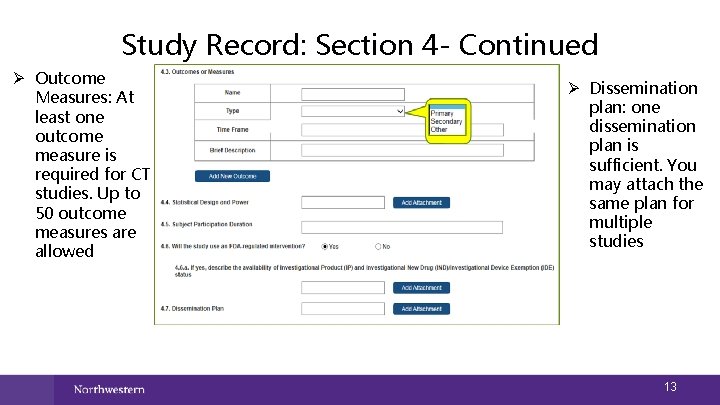
Study Record: Section 4 - Continued Ø Outcome Measures: At least one outcome measure is required for CT studies. Up to 50 outcome measures are allowed Ø Dissemination plan: one dissemination plan is sufficient. You may attach the same plan for multiple studies 13

Study Record: Section 5 - Other Clinical Trialrelated Attachments Ø Attachments in this section are allowed for CT only. Up to 10 attachments can be uploaded, only if noted in FOA. 14

Tips on How to connect with your PI on New Forms Ø Confirm with PI if the proposal is Clinical Trial or Not Clinical Trial Ø For some research grants NIH has specific FOA for CT and for not CT Ø PI’s are not familiar with new Human subjects forms Ø Send them the screen shots of the forms Ø Convert forms into PDF fillable forms Ø Set a meeting to complete these forms Note: Please do not try to answer any question on your own. Note: You will experience errors for copy and pasting the text in some fields. 15

NIH Single IRB Ø The policy affects multi-site studies involving non-exempt human subjects research funded by NIH, and applies to grant applications with due dates on or after January 25, 2018 Ø This policy applies to the domestic sites of NIH-funded multi-site studies where each site will conduct the same protocol involving non-exempt human subjects research. It does not apply to career development, research training or fellowship awards. 16

Single IRB at Proposal Stage Ø Single IRB plan: The justification must include the following elements: Ø Describe how you will comply with NIH policy on use of s. IRB for Multi-site research Ø Provide the name of the IRB that will serve as the s. IRB of record. Ø Indicate that all identified participating sites have agreed to rely on the proposed s. IRB and that any sites added after award will rely on the s. IRB. Ø Briefly describe how communication between sites and the s. IRB will be handled Ø Indicate that all participating sites will, prior to initiating the study, sign an authorization/reliance agreement that will clarify the roles and responsibilities of the s. IRB and participating sites. Ø Indicate which institution or entity will maintain records of the authorization/reliance agreements and of the communication plan. Note: Do not include the authorization/reliance agreement(s) or the communication plan(s) documents in your application. 17

Single IRB at Proposal Stage NU IRB of Record Letter of Support: Ø A letter reflecting the acknowledgement that the NU IRB is willing to serve as IRB of record for the study. Ø This must be obtained from NU IRB office and should be included in the proposal with single IRB plan. Sub-award Ø A letter of support from sub-awardee sites indicating they are willing to cede review to the NU IRB is required. Ø This letter should be uploaded into Info. Ed internal documents 18

QUESTIONS? 19
- Slides: 20