New legislation impacting IMP Brenda Van Assche 16

New legislation impacting IMP Brenda Van Assche – 16 Dec 2016 1
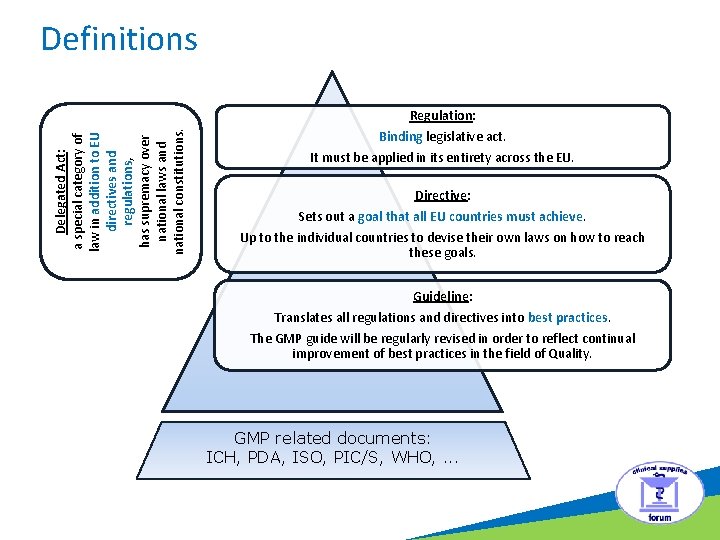
Definitions Delegated Act: a special category of law in addition to EU directives and regulations, has supremacy over national laws and national constitutions. Regulation: Binding legislative act. It must be applied in its entirety across the EU. Directive: Sets out a goal that all EU countries must achieve. Up to the individual countries to devise their own laws on how to reach these goals. Guideline: Translates all regulations and directives into best practices. The GMP guide will be regularly revised in order to reflect continual improvement of best practices in the field of Quality. GMP related documents: ICH, PDA, ISO, PIC/S, WHO, . . .
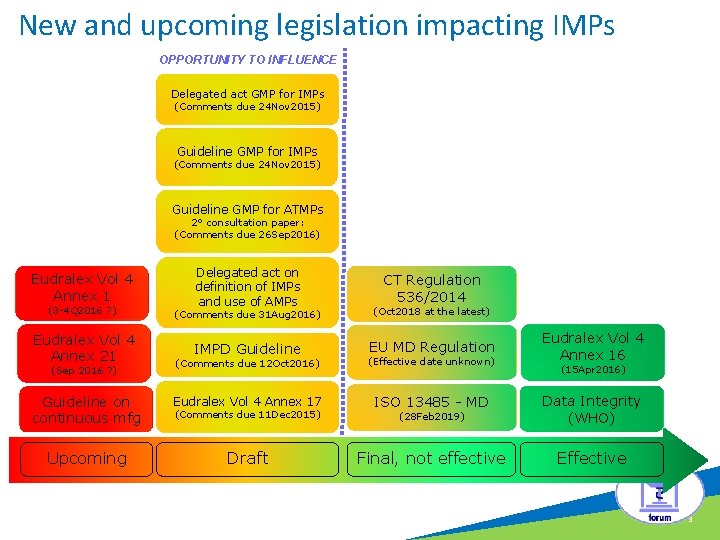
New and upcoming legislation impacting IMPs OPPORTUNITY TO INFLUENCE c Delegated act GMP for IMPs (Comments due 24 Nov 2015) Guideline GMP for ATMPs 2° consultation paper: (Comments due 26 Sep 2016) Eudralex Vol 4 Annex 1 Delegated act on definition of IMPs and use of AMPs CT Regulation 536/2014 (3 -4 Q 2016 ? ) (Comments due 31 Aug 2016) (Oct 2018 at the latest) Eudralex Vol 4 Annex 21 IMPD Guideline EU MD Regulation Eudralex Vol 4 Annex 16 (Comments due 12 Oct 2016) (Effective date unknown) Guideline on continuous mfg Eudralex Vol 4 Annex 17 ISO 13485 - MD (28 Feb 2019) Data Integrity (WHO) Upcoming Draft Final, not effective Effective (Sep 2016 ? ) (Comments due 11 Dec 2015) (15 Apr 2016) 3
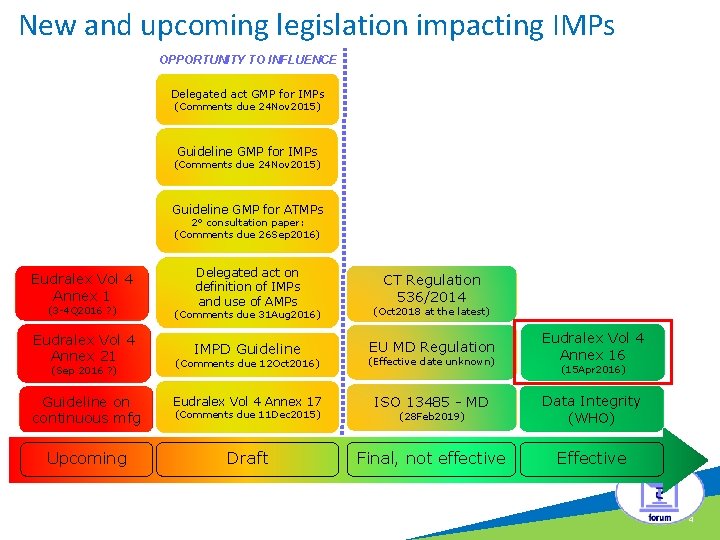
New and upcoming legislation impacting IMPs OPPORTUNITY TO INFLUENCE c Delegated act GMP for IMPs (Comments due 24 Nov 2015) Guideline GMP for ATMPs 2° consultation paper: (Comments due 26 Sep 2016) Eudralex Vol 4 Annex 1 Delegated act on definition of IMPs and use of AMPs CT Regulation 536/2014 (3 -4 Q 2016 ? ) (Comments due 31 Aug 2016) (Oct 2018 at the latest) Eudralex Vol 4 Annex 21 IMPD Guideline EU MD Regulation Eudralex Vol 4 Annex 16 (Comments due 12 Oct 2016) (Effective date unknown) Guideline on continuous mfg Eudralex Vol 4 Annex 17 ISO 13485 - MD (28 Feb 2019) Data Integrity (WHO) Upcoming Draft Final, not effective Effective (Sep 2016 ? ) (Comments due 11 Dec 2015) (15 Apr 2016) 4

Eudralex Vol 4 Annex 16: Final, Effective § Annex 16: § § “Certification by a Qualified Person and Batch release” Effective since 15 April 2016 The principles apply to IMP for human use Reasons for changes: to reflect the globalisation of the pharmaceutical supply chains and the introduction of new quality control strategies § Highlights: § § § Supply Chain Map Continous QP training Deviation Handling Quality Management System QP to QP agreement • EMA Q&A pending release
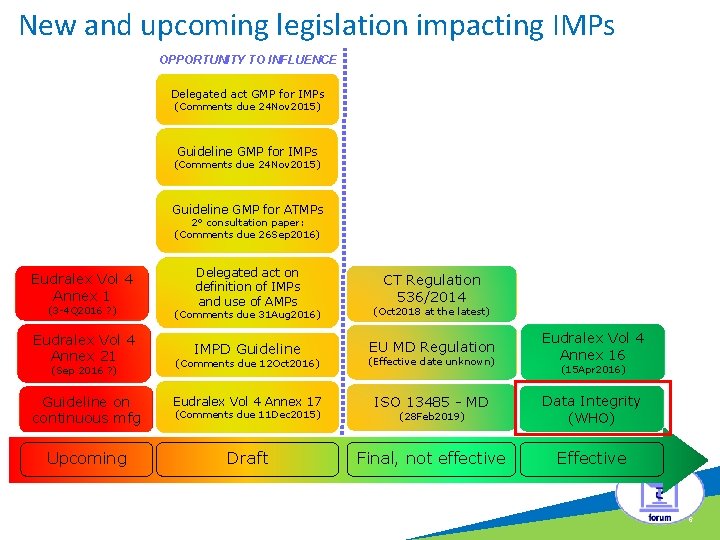
New and upcoming legislation impacting IMPs OPPORTUNITY TO INFLUENCE c Delegated act GMP for IMPs (Comments due 24 Nov 2015) Guideline GMP for ATMPs 2° consultation paper: (Comments due 26 Sep 2016) Eudralex Vol 4 Annex 1 Delegated act on definition of IMPs and use of AMPs CT Regulation 536/2014 (3 -4 Q 2016 ? ) (Comments due 31 Aug 2016) (Oct 2018 at the latest) Eudralex Vol 4 Annex 21 IMPD Guideline EU MD Regulation Eudralex Vol 4 Annex 16 (Comments due 12 Oct 2016) (Effective date unknown) Guideline on continuous mfg Eudralex Vol 4 Annex 17 ISO 13485 - MD (28 Feb 2019) Data Integrity (WHO) Upcoming Draft Final, not effective Effective (Sep 2016 ? ) (Comments due 11 Dec 2015) (15 Apr 2016) 6

Data Integrity • Data Integrity = The Topic of the Year Attributable Legible Contemporaneous Original Accurate • Current regulatory framework: Ø WHO guideline = current “State of the Art” Ø FDA Ø PIC/S • EU regulations: Ø EMA Q&A Ø MHRA: consultation paper (v 3) • EU Inspections versus Chapter 4 “Documentation” and Annex 11 “Computerised systems”
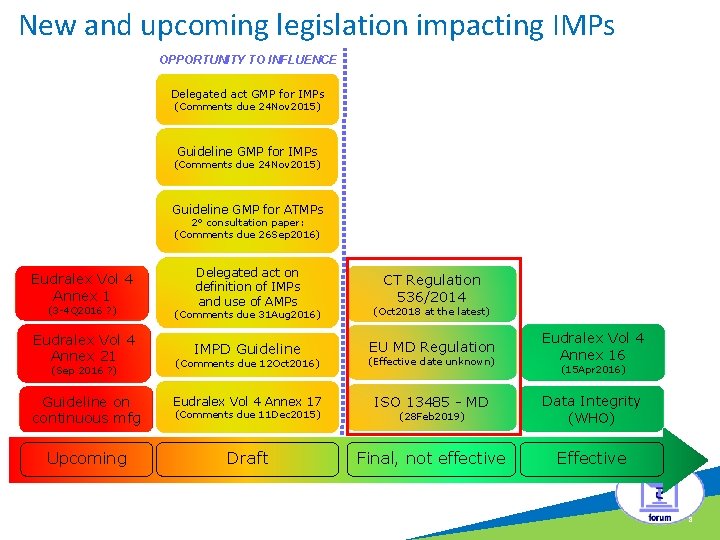
New and upcoming legislation impacting IMPs OPPORTUNITY TO INFLUENCE c Delegated act GMP for IMPs (Comments due 24 Nov 2015) Guideline GMP for ATMPs 2° consultation paper: (Comments due 26 Sep 2016) Eudralex Vol 4 Annex 1 Delegated act on definition of IMPs and use of AMPs CT Regulation 536/2014 (3 -4 Q 2016 ? ) (Comments due 31 Aug 2016) (Oct 2018 at the latest) Eudralex Vol 4 Annex 21 IMPD Guideline EU MD Regulation Eudralex Vol 4 Annex 16 (Comments due 12 Oct 2016) (Effective date unknown) Guideline on continuous mfg Eudralex Vol 4 Annex 17 ISO 13485 - MD (28 Feb 2019) Data Integrity (WHO) Upcoming Draft Final, not effective Effective (Sep 2016 ? ) (Comments due 11 Dec 2015) (15 Apr 2016) 8

Final, not effective • Clinical Trial Regulation 536/2014 Ø Effective as from Oct 2018, at the latest Expect more communication from EMA EO 2016 • EU Medical Device Regulation Ø Effective date unknown yet • ISO 13485 - Medical device Ø Effective as from 28 Feb 2019
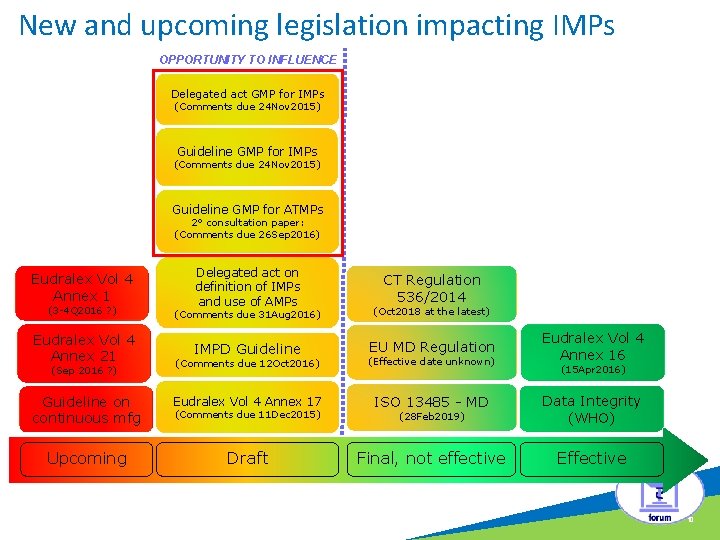
New and upcoming legislation impacting IMPs OPPORTUNITY TO INFLUENCE c Delegated act GMP for IMPs (Comments due 24 Nov 2015) Guideline GMP for ATMPs 2° consultation paper: (Comments due 26 Sep 2016) Eudralex Vol 4 Annex 1 Delegated act on definition of IMPs and use of AMPs CT Regulation 536/2014 (3 -4 Q 2016 ? ) (Comments due 31 Aug 2016) (Oct 2018 at the latest) Eudralex Vol 4 Annex 21 IMPD Guideline EU MD Regulation Eudralex Vol 4 Annex 16 (Comments due 12 Oct 2016) (Effective date unknown) Guideline on continuous mfg Eudralex Vol 4 Annex 17 ISO 13485 - MD (28 Feb 2019) Data Integrity (WHO) Upcoming Draft Final, not effective Effective (Sep 2016 ? ) (Comments due 11 Dec 2015) (15 Apr 2016) 10

Draft • “Commission Delegated Act on Principles and guidelines on GMP for IMPs for human use and inspection procedures” and “Detailed Commission guidelines on GMP for IMPs for human use” Ø Comments have been submitted by 24 Nov 2015, pending revisions. Comments focused on clear delineation between IMP vs commercial MP. Ø Directive 2003/94/EC is split into 2 new documents: one for IMP (= Delegated Act) and one for commercial MPs Ø Annex 13 will become obsolete when the guideline becomes operational

Draft • Guideline on GMP for ATMPs Ø ATMPs = Advanced Therapy Medicinal Products Ø What is an ATMP? Defined in Directive 2001/83/EC, as amended. An ATMP is a medicinal product, which is either: • a gene therapy medicinal product as defined in Part IV of Annex 1 to Directive 2001/83/EC • a somatic cell therapy medicinal product as defined in Part IV of Annex 1 to Directive 2001/83/EC • a tissue engineered product as defined in Article 21 (b) of the ATMP Regulation 1394/2007

Draft • Guideline on GMP for ATMPs Ø Stand-alone guideline document pursuant to Article 5 of Regulation 1394/2007, hence independent from EU GMP guidelines Part I & II and Annexes. One standard to reduce burden and allow flexibility for manufacturers to apply measures best suited to the specific product. Ø In-Scope: ATMPs for commercial as well as clinical use Ø Out-of-Scope: Manufacturing of ATMPs under the hospital exemption

Draft • Guideline on GMP for ATMPs – Current status Ø Current draft is developed from Annex 2 repetition of much of Volume 4 (personnel, premises, equipment, documentation, etc. ), however key components from Volume 4 are missing (key elements of pharmaceutical quality management system e. g. Change Control, QRM = reflected in risk based approach, no link to ICHQ 10) Ø Possible ways to go: 1) Include into Volume 4 as a new annex, 2) Stand-alone document with cross-references to relevant Volume 4 chapters and annexes, 3) Stand-alone document with potential repetition of GMP requirements from relevant Volume 4 chapters and annexes
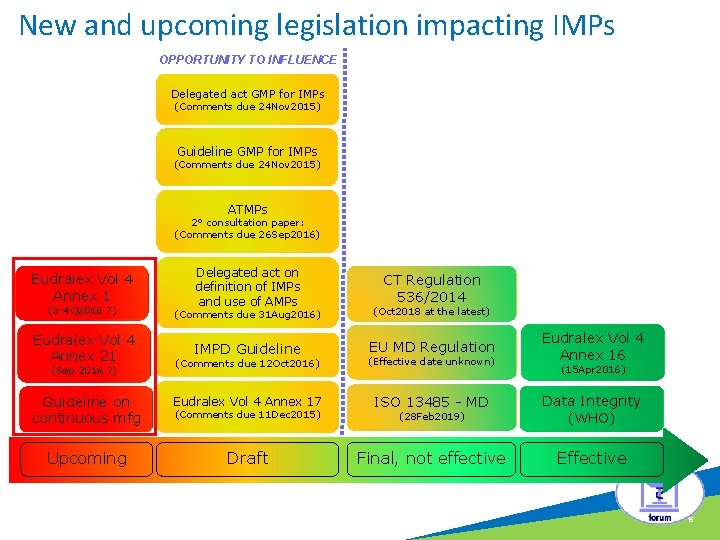
New and upcoming legislation impacting IMPs OPPORTUNITY TO INFLUENCE c Delegated act GMP for IMPs (Comments due 24 Nov 2015) Guideline GMP for IMPs (Comments due 24 Nov 2015) ATMPs 2° consultation paper: (Comments due 26 Sep 2016) Eudralex Vol 4 Annex 1 Delegated act on definition of IMPs and use of AMPs CT Regulation 536/2014 (3 -4 Q 2016 ? ) (Comments due 31 Aug 2016) (Oct 2018 at the latest) Eudralex Vol 4 Annex 21 IMPD Guideline EU MD Regulation Eudralex Vol 4 Annex 16 (Comments due 12 Oct 2016) (Effective date unknown) Guideline on continuous mfg Eudralex Vol 4 Annex 17 ISO 13485 - MD (28 Feb 2019) Data Integrity (WHO) Upcoming Draft Final, not effective Effective (Sep 2016 ? ) (Comments due 11 Dec 2015) (15 Apr 2016) 15

Thank You ! © 2015 Clinical Supplies Forum 16
- Slides: 16