NEW FACULTY ONBOARDING CHOP Research Institute Last updated

NEW FACULTY ONBOARDING CHOP Research Institute Last updated: September 2020 1

WELCOME Beverly L. Davidson, Ph. D Chief Scientific Strategy Officer Director, Center for Cellular and Molecular Therapeutics Richard Aplenc, MD, Ph. D, MSCE AVP and Chief Clinical Research Officer davidsonbl@email. chop. edu aplenc@email. chop. edu 2
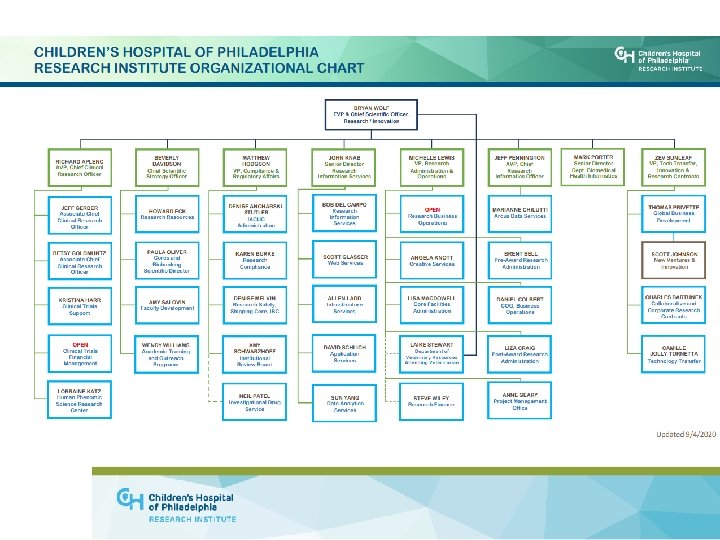

NEW FACULTY ONBOARDING Outline • • • Post-arrival assistance Business Administration (grants and contracts) Human Resources (HR) Faculty Affairs and Professional Development Office at Penn Office of Faculty Development at CHOP Research Institute Academic Training and Outreach Programs (ATOP) Research Safety Programs Research Compliance and Regulatory Affairs CHOP Conflict of Interest Program CHOP Institutional Review Board (IRB) 4

NEW FACULTY ONBOARDING Outline, continued. . • • • Clinical Research Support Office of Technology Transfer (OTT) Office of Entrepreneurship and Innovation (OEI) Office of Collaborative and Corporate Research Contracts (OCCRC) Research Institute Cores Grant review groups • Pre-review consultation for K’s, R’s, etc. • Research Information Services (RIS) 5

ONBOARDING Moving or starting your lab Howard Eck Director, Research Resources eck@email. chop. edu 215 -590 -4662 I am here! How do I…………? 6

ONBOARDING: Post Arrival • Establish a “punch list” • These are the items that need to be taken care of in order to make sure you are up and running as quickly as possible • Are there equipment purchases that need to be made right away? • Infrastructure, tele/data, minor renovations • A single point of contact for anything new for your lab e. g. : Electrical Telephones Keys Lockers Security Moving People/Equipment 7

ONBOARDING: Post-arrival • Lab Needs • Equipment installation, plumbing, electrical outlets, lab or office furniture, etc. • Office and Staff Needs • Keys, ID access, telephone connections, voicemail (if used), moving employees within your space • Archibus- CAFM database • Space Requests • Facilities Help Desk (4 -1046) • IT Help Desk (4 -4357) • Site Managers: • Tom Parkin (Abramson) • Anthony Campese (Colket) • Walter Morrison (Roberts) 8

Pre- and Post-Award Research Administration and Research Portfolio Management (Previously Sponsored Projects and Research Business Management) Michelle Lewis VP, Research Administration & Operations lewism 8@email. chop. edu 267 -426 -3006 9

Sponsored Projects and Research Business Management (Pre-Award & Post-Award) Three teams work to support the pre-award, post-award and incoming/outgoing sub-award processing and negotiation on behalf of CHOP Principal Investigators: • • • SPO = Sponsored Project Officer • Helps insure that the PI’s grant application(s) are submitted in compliance with the Sponsor guidelines and CHOP policies, and handles non-financial post-award items RBM = Research Business Manager • Supports PI’s in the management of their research and non-research portfolio’s including cost monitoring and financial closeout of awards • Meets with PI’s to determine budgets, salary, and resource allocations for new awards (at proposal stage) and post-award (at JIT and during the active award) Sr. GCS = Sr. Grant & Contract Specialist • Negotiates the incoming and outgoing subaward agreements as well as outgoing consulting agreements that support PIs’ collaborations with external individuals and institutions 10

Sponsored Projects and Research Business Management How can I find out who is my assigned RBM, SPO and/or GCS? Please visit: https: //sprbm. research. chop. edu/ The online tool is searchable by PI Name, Department or Division 11

Subject Matter Experts Michelle Lewis VP, Research Administration and Operations lewism 8@email. chop. edu Brent Bell Director, Pre-Award Research Administration 267 -425 -0509 bellb@email. chop. edu Liza Craig Director, Post-Award Research Administration 215 -590 -3845 craigl@email. chop. edu 12
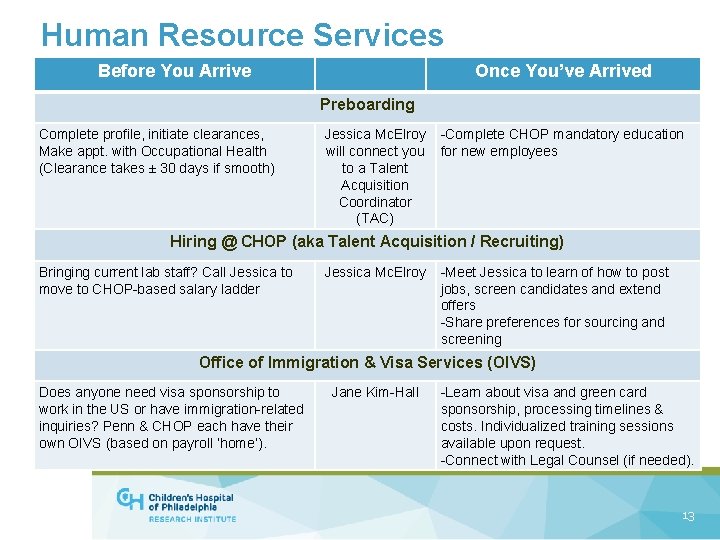
Human Resource Services Before You Arrive Once You’ve Arrived Preboarding Complete profile, initiate clearances, Make appt. with Occupational Health (Clearance takes ± 30 days if smooth) Jessica Mc. Elroy will connect you to a Talent Acquisition Coordinator (TAC) -Complete CHOP mandatory education for new employees Hiring @ CHOP (aka Talent Acquisition / Recruiting) Bringing current lab staff? Call Jessica to move to CHOP-based salary ladder Jessica Mc. Elroy -Meet Jessica to learn of how to post jobs, screen candidates and extend offers -Share preferences for sourcing and screening Office of Immigration & Visa Services (OIVS) Does anyone need visa sponsorship to work in the US or have immigration-related inquiries? Penn & CHOP each have their own OIVS (based on payroll ‘home’). Jane Kim-Hall -Learn about visa and green card sponsorship, processing timelines & costs. Individualized training sessions available upon request. -Connect with Legal Counsel (if needed). 13

Human Resource Services Before You Arrive Once You’ve Arrived General Human Resources (HR) If you have an HR question or concern and don’t know who to contact, Leon can help you connect with the right person. Leon Jones w/Heather Davis supporting the Practice Plans Performance management, employee relations, job descriptions and compensation. Connect to / liaison with other HR services. Organizational Development (OD) Unlikely that you’d have an OD need prior to your arrival, but if you can’t connect with who you need, Ruth is accessible for escalation. Maxine Johnson New leader support, training & development, talent management, engagement, retreats & in-tact team interventions, organizational analysis, design, etc. HR Leadership Unlikely that you’d have an HR leadership need prior to your arrival, but if you can’t connect with who you need, I’m accessible for escalation. Julie Josefowski You are in very capable hands with the team. I am at your disposal for questions, comments & concerns. 14

HR Personnel Jessica Mc. Elroy Leon Jones Jane Kim-Hall Manager Talent Acquisition 267 -226 -3423 Strategic HR Business Partner 267 -425 -1262 Program Manager Immigration Services 267 -425 -1244 Photo Not Available Julie Josefowski Director HR Business Partner Services 267 -425 -1264 Photo Not Available Heather Davis Maxine L Johnson Sr. HR Business Partner 267 -671 -6579 Sr. Manager HR 267 -425 -1258 15

FACULTY AFFAIRS & PROFESSIONAL DEVELOPMENT (FAPD) Perelman School of Medicine • Oversees the recruitment and appointment, promotion, and retention of distinguished faculty. • Offers professional development programs and support in career development, clinical skills, leadership and management, scholarship and teaching. • Promotes professionalism, well-being, and resilience. Victoria A. Mulhern Executive Director vmulhern@upenn. edu Stephanie Taitano Director of Faculty Professional Development, PSOM staitano@upenn. edu Kim Haebel CHOP Faculty Coordinator khaebel@upenn. edu 16

Faculty Life & Professional Development Program within the FAPD Office http: //www. med. upenn. edu/flpd/ Among many Faculty Life & Professional Development offerings are workshops and resources relating to the four full-time academic tracks: • • Tenure View more Clinician Educator View more Research View more Academic Clinician View more 17

OFFICE OF FACULTY DEVELOPMENT (OFD) CHOP Research Institute • Onboarding session • One-on-one meetings to welcome new CHOP faculty • New assistant professor gatherings for networking and discussing topics of shared interest • Research Institute pilot grants • Monthly Funding Forecaster • Office of Faculty Development web site • Annual Report on Faculty Demographics Amy Salovin Office of Faculty Development CHOP Research Institute salovina@chop. edu 18

Office of Faculty Development Research Institute Pilot Grant Programs • Jr. Investigator Pilot Grants § Supports work of Jr. Faculty (within 1 -4 years of appt) with a new mentor outside of current mentor to expand on needed expertise • K-Readiness Pilot Grants § For Sr. Fellows and Instructors o Provides salary support for staff that can assist with the research objectives of the trainee • Basic-Clinical Pilot Grants o To support new collaborations between basic and clinical researchers to advance translational science 19

FUNDING FORECASTER Distributed the first Tuesday of every month • Funding opportunities emailed by Office of Faculty Development to all Tenure, C-E and Research track faculty § Summary of CHOP/Penn Pilot and Limited Application grants § Recent NIH RFA’s and PAR’s. Do. D programs § Calendar of foundation and association grants 20

ACADEMIC TRAINING AND OUTREACH PROGRAMS (ATOP) Wendy Reed Williams, Ph. D williamsw@email. chop. edu Academic Training Career and Professional Development Support for Researchers-in. Training Outreach Programs STEM Pipeline Programs, Community Engagement and Research Training Support Specialty Programs and Diversity Workforce Diversity and Development of Targeted Research Programs and Pilot Grant Opportunities

Specialty Programs and Diversity Academic Training Outreach Programs • Career/Productivity Events for Early-Career Researchers • Postdoc Affairs • One-on-one Counseling • Mentor Support • PI Participation in Trainee Development Programming • Academic Training Advisory Council • Penn Postdoc Program and Career Services Liaison • CRISSP Undergraduate Research Internship • CHOP-RISES High School Internship • Symposia and Faculty Luncheon • Community Events • Responsible Conduct of Research Training • Training Wizard • Postdoctoral Fellowship for Academic Diversity • Liaison with CHOP Office of Diversity and Inclusion • Postdoc Communities and Ambassador Program • Cell and Gene Therapy Clinical Training Program • Research Institute Diversity Data • Preparing for Academic Teaching Workshop Jodi Leckrone, MEd leckrone@email. chop. edu Paulette Mc. Rae, Ph. D mcraep@email. chop. edu David Taylor, Ph. D taylord@email. chop. edu

RESEARCH SAFETY PROGRAMS: Who are we? Office of Research Safety (ORS) Office of the Institutional Biosafety Committee (IBC) Research Institute Shipping Core (RISC) Denise Melvin, Director Melvind@email. chop. edu 23

Office of Research Safety: What do we do? • • • Training programs (Bio, Chem, Lab Safety) Lab safety surveys and walkthroughs Individual lab consultations (Risk Assessments) PPE consultations COVID-19 guideline consultations Lab Safety Meetings (LSMs) Drills Online risk assessment questionnaire (RAQ) review IACUC/IBC/IRB protocol review Outreach Events (Research Safety Day) Special projects related to COVID-19 Incident response (24/7 on call) 24

Office of Institutional Biosafety Committee: What do we do? • Protocol review and triage • New protocols, amendments, 3 -year renewals • Review of IACUC and IRB protocols to ensure crossharmonization with IBC • Update e. IBC Smart. Form • Track biological agent inventory for all labs • Perform annual Biosafety Stand-down • Monitor NIH Guidelines to ensure compliance • Coordinate bi-monthly IBC meetings 25

Research Institute Shipping Core: What do we do? • Packaging, labeling, shipping and tracking of hazardous materials shipments • Consultations with Certified Shippers and Labs requiring shipping guidance • Assistance with identification and procurement of shipping permits • Training and certification for "Certified Shippers“ • Maintenance of Shipping website along with order form in i. Lab • Provide supplies to "Certified Shippers" at cost • Coordinate with alternate carriers (World Courier, etc. ) • Shipment of some patient medications 26

RESEARCH COMPLIANCE & REGULATORY AFFAIRS Matthew Hodgson AVP, Research Compliance & Regulatory Affairs hodgsonm@email. chop. edu 267 -426 -8723 Denise Ancharski-Stutler Admin Director, Office of IACUC Administration ancharskistutlerd@email. chop. edu 267 -425 -0145 Karen Burke Director, Research Compliance burkek 6@email. chop. edu 267 -425 -2447 Amy Schwarzhoff Director, Human Subjects Research schwarzhoffa@email. chop. edu 267 -426 -2346 Denise Melvin Director, Research Safety Programs melvind@email. chop. edu 267 -426 -7597 Adam Motsney Supervisor, Investigational Drug Service motsneya@email. chop. edu 215 -590 -1789 Katina Frangakis Manager, Shipping Core frangakisk@email. chop. edu 267 -425 -2079 27

Office of Research Compliance • Routine and for-cause quality assurance and monitoring services • Human subjects research • Investigational New Drug (IND) and Investigational Device Exemption (IDE) applications held by CHOP employees • Research billing • • Clinicaltrials. gov support Export control Policy and procedure documentation program Human Research Protection Program (HRPP) and Good Clinical Practice (GCP) training oversight • Research misconduct 28

CHOP CONFLICT OF INTEREST (COI) PROGRAM “Conflict of interest” refers to situations in which financial or other personal considerations may compromise, or have the appearance of compromising, an individual’s professional judgment Janet Holcombe Vice President, Chief Compliance and Privacy Officer 267 -426 -6037 holcombej@chop. edu Patrick Egan, Senior Director Compliance and Conflicts of Interest 267 -426 -6049 eganp@chop. edu 29

CHOP COI Requirements • Types of conflicts: • Clinical (outside interests may affect prescribing practices or clinical decision-making) • Purchasing (outside interests may affect decisionmaking with respect to purchasing or vendor relations) • Commitment (outside commitments may conflict with primary obligations to CHOP or with CHOP’s mission) • Research (research has potential to be biased as a result of outside interests – both those of the individual and of the Institution) 30

e. COI Electronic Disclosure System • Online disclosure system • Part of e. Research – interacts with e. IRB, e. SPA, e. Agreements • COI review required aspect of IRB approval, acceptance of funds, and approval of agreements • COI Training • Required every four years, integrated into e. COI disclosure process • Annual/Primary Disclosure Form • Required once per year in February • Reviewed by Department Chair/VP and COI Office • Project-specific Disclosure Form • • Pulls information from Annual/Primary form Required for each specific project Helps COI Office determine whether project could affect interest Reviewed by COI Office and Conflict of Interest Committee (if necessary) 31

COI Resources • Conflict of Interest OfficeOffice of Compliance and Privacy • 267 -426 -6044 • COI@email. chop. edu • COI @ CHOP site: https: //at. chop. edu/general-counsel/compliance-privacy/intranetpages/conflict-of-interest • COI policy: https: //at. chop. edu/communities/policyprocedure/administrative/Active/conf lict. pdf • Anonymous Reporting of Compliance Concerns: 1 -866 -246 -7456 or www. mycompliancereport. com (enter CHOP when prompted for institution access code) 32

INSTITUTIONAL REVIEW BOARD (IRB) Not Research Exempt Human Subjects Research Not Human Subjects Research Expedited Review Full Board Review Barbara Engel, MD, Ph. D Chair 267 -426 -6859 33 engelbc@email. chop. edu

IRB Resources • IRB webpage https: //irb. research. chop. edu/ What must be reviewed by the IRB https: //irb. research. chop. edu/what-must-be-reviewed-irb • Protocol and Consent Templates https: //irb. research. chop. edu/forms • SOPs https: //irb. research. chop. edu/policies • Contact https: //irb. research. chop. edu/contact IRB Help line (215 -590 -2830) IRBoffice@email. chop. edu • • • IRB Office Hours – 4 times per month on Thursday afternoons and Friday mornings (no appointment required – call Help line for schedule) • Meet with the Director, Assistant Director or one of the IRB chairs 34

CLINICAL RESEARCH SUPPORT OFFICE Richard Aplenc, MD, Ph. D, MSCE AVP & Chief Clinical Research Officer aplenc@email. chop. edu 267 -426 -7252 Clin. Research. Support@email. chop. edu 215 -590 -0574 35

CLINICAL RESEARCH SUPPORT OFFICE Jeffrey Gerber, MD, Ph. D Associate Chief Clinical Research Officer Elizabeth Goldmuntz, MD Associate Chief Clinical Research Officer Amanda Di. Nofia, MD Associate Chief Clinical Research Officer Focuses on biostatistical support for PIs Gerberj@email. chop. edu Focuses on clinical staffing support for PIs Goldmuntz@email. chop. edu Focuses on Cell Therapy INDs Dinofiaa@email. chop. edu 36

The CRSO Team • Research Staffing Core • Clinical research personnel provide study coordination activities on a full or part-time basis • Contact: Michele Toms, Administrative Director, Toms@email. chop. edu • Clinical Trials Financial Management • Budget preparation and negotiation • Research subject registration and charge review • Sponsor invoicing • Contact: CTFM@email. chop. edu • Clinical Trial Support • On. Core Clinical Trial Management System Support • REDCap Support • Recruitment Enhancement Core (REC) • Recruitment strategy consultation, sends recruitment emails • IND/IDE Support • Provides regulatory and operational support for CHOP physician-initiated FDA regulated clinical trials • Contact: Kristina Harr, Director, Harrk@email. chop. edu 37

Biostatistics at CHOP • Several groups provide biostatistics support at CHOP • CHOP based PENN faculty • Data Science and Biostatistics Unit (DSBU) • Biostatistics and Data Management Core (BDMC) • We are developing outward-facing summaries, published on RI intranet, of expertise, services, and personnel to provide information for investigators in need of data management, analysis, and grant support • Formal intake process being developed to assess current use across groups and need for expansion 38

Biostatistics at CHOP • UPENN Biostatistics Faculty Support • Justine Shults, Ph. D, lead biostatistician shultsj@email. chop. edu • Currently six faculty members • • • Jing Huang, Ph. D Yimei Li, Ph. D Di Shu, Ph. D Justine Shults, Ph. D Rai Xiao, Ph. D Jarcy Zee, Ph. D 39

Biostatistics at CHOP • Data Science and Biostatistics Unit (DSBU) • Heather Griffis, Ph. D, Director; griffish@email. chop. edu • Staffed by Ph. D and Masters-level research statisticians and statistical scientists who pull, clean, manage, and analyze data • Close collaboration with REDCap team and Informatics groups in Department of Biomedical Health and Informatics to provide integrated data management and analytic support • Extensive expertise in administrative data • PHIS, HCUPS databases, Medicaid, NAMCS, NHDS, Premier Perspective, OPTUM, and Market. Scan. • Expertise in geographic information systems and geospatial analyses 40

Biostatistics at CHOP • Westat Biostatistics and Data Management Core (BDMC) https: //bdmc. research. chop. edu/contact. php • Biostatistics support • Study design, power calculations, analytic plans for grant applications • Data management services • Database development and maintenance • Preparation data for FDA submission 41

Office of Technology Transfer (OTT) Office of Entrepreneurship & Innovation (OEI) Office of Collaborative & Corporate Research Contracts (OCCRC) Zev Sunleaf Vice President-Technology Transfer, Innovation & Research Contracts sunleafs@email. chop. edu 267 -425 -3029 42

OTT: IP and Licensing • Manage the intellectual property (IP- i. e. patents and copyrights) created across CHOP • Make patent filing decisions based on prior art and business case • Bring research from bench to bedside • Work with inventors to market technologies and find licensees and partners • Negotiate license and option agreements • Distribute income Camille Jolly-Tornetta Director, Office of Technology Transfer jollytornc@chop. edu https: //www. research. chop. edu/office-technology-transfer-commercialization-innovation 43

OCCRC Collaborative and Corporate Research Contracts • Negotiates industry-funded sponsored research contracts and clinical trial agreements, and unfunded collaboration agreements • Unfunded collaborative research agreements (CRA) with corporate and academic partners and consortiums • Industry funded clinical trials (CTA) or sponsored research agreements (SRA) • Confidential disclosure agreements (CDA/NDA) • Agreements to send or receive materials (MTA) • Agreements to send or receive data (DUA, BAA) • Personal faculty consulting agreements (FCA) www. research. chop. edu/office-of-collaborative-and-corporate-research-contracts Charles Bartunek Director-Office of Collaborative & Corporate Research Contracts bartunekc@email. chop. edu 44

OCCRC Collaborative and Corporate Research Contracts • Agreement submission portals: • Sophia • MTA, DUA, CDA, unfunded Collaboration Agreement, CORE Services Agreement • e. SPA • Industry-funded Sponsored Research Agreement • e. Agreements • Faculty consulting agreement • e. SIFTER • New Clinical Trial Agreement or an amendment to a CTA Questions? researchcontracts@email. chop. edu 45

OEI: Entrepreneurship & Innovation • Create a culture of Innovation at CHOP • Innovation Rounds • Innovation Series • Office Hours • Sprint • Targeted Outcomes • Alternative Revenue Streams • Licensable Assets • New CHOP Startup companies Tom Privette Head-Global Business Development & External Partnering privettet@email. chop. edu https: //www. chop. edu/centers-programs/office-entrepreneurship-and-innovation 46

CHOP RESEARCH INSTITUTE CORES The CHOP Research Institute Cores provide state-of-the-art technologies, technical expertise and education to facilitate discovery, and support the research missions of our investigators https: //www. research. chop. edu/our-research/core-facilities Paula M. Oliver, Ph. D Lisa Mac. Dowell, MBA, CPA, CIA Scientific Director for Cores 267 -426 -2839 oliverp 1@email. chop. edu Director, Core Facilities Administration 267 -426 -7896 macdowell@email. chop. edu Find CHPS cores here Find PSOM cores here 47

Grant Review Communities Umbrella for grant groups at CHOP G R C Grant Proposal Success & others

Grant Review Communities Neurology Affinity Group Grants Club Mike Robinson Pathology/Cell Biology Jan Burkhardt Yair Argon Mickey Marks K Advisory Dept. of Pediatrics Grant Proposal Success (GPS) Program 49 Matthew Weitzman

Grant Review Communities AIMS 50 Help you identify whether you… Share experience in grant writing …are asking the right scientific question. Learn about each other’s science …are using the best experimental approach. Build communities to promote collaboration …have assembled the best team for the project.

Grant Proposal Success Program Existing Campus Groups Pathology Breakfast Club 51 Cancer GPS Micro GPS Neuro GPS* Richards Society GPS Basic Science K (Ba. Sci. K) GPS Workshop Logo indicates primary organizing institution, though members from either institution are welcome! * Neuro-GPS is a separate group from the Neurology Affinity Group Grants Club
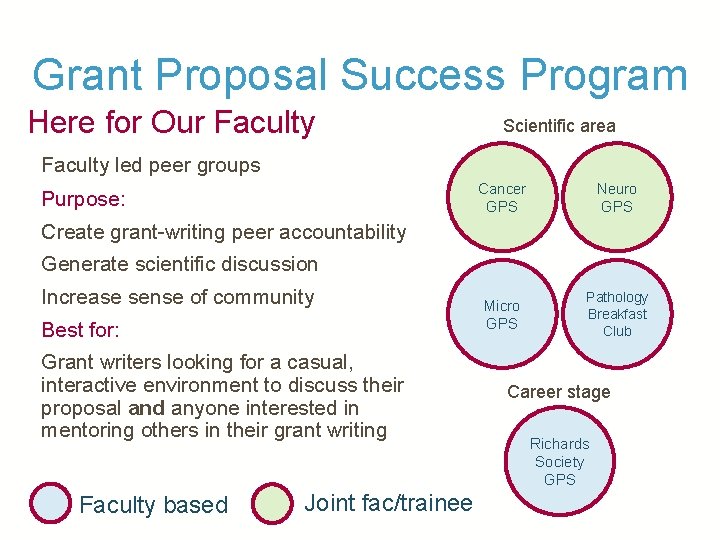
Grant Proposal Success Program Here for Our Faculty Scientific area Faculty led peer groups Purpose: Cancer GPS Neuro GPS Micro GPS Pathology Breakfast Club Create grant-writing peer accountability Generate scientific discussion Increase sense of community Best for: Grant writers looking for a casual, interactive environment to discuss their proposal and anyone interested in mentoring others in their grant writing Faculty based Joint fac/trainee Career stage Richards Society GPS

Grant Proposal Success Program Supporting Our Trainees GPS Workshop Ba. Sci. K GPS 5 month comprehensive training program • • Offered annually, competitive enrollment Unparalleled multi-part educational series Supported Grant Mechanisms: F 32, K-Series, and equiv. foundation awards Best for: Trainees who are ready to write and submit, and like working in an interactive format GPS Workshop Pediatrics K Advisory Individual grant review • • Specialized panels built for each individual Customized to meet your goals Supported Grant Mechanisms: K-Series proposals Best for: Trainees 2 -3 months from submission who are seeking writing independence Basic Science K (Ba. Sci. K) GPS Pediatrics K Club

Research Information Services Management Team John Knab Senior Director knabj@email. chop. edu 267 -425 -2163 Bob Del Campo Assistant Director delcampo@email. chop. edu 267 -426 -1999 Allen Ladd Infrastructure Services Manager ladd@email. chop. edu 267 -426 -9263 Scott Glasser Associate Director, Web Strategy glassers@email. chop. edu 267 -425 -1991 Our Portfolio: Dave Schlich Application Services Manager schlichd@email. chop. edu 267 -426 -7818 Sun Yang Business Analytics Manager yangs@email. chop. edu 267 -426 -2518 • • • End User Technology Services Infrastructure/Cloud Services Web Services Application Services Business Analytics Services 54

Research Information Services • Provides Technology Services Focused on the Research Institute: • • • Web (Online presence and communications) • Data needs (High Performance Compute, retention, large datasets) • End User Devices: Requisitions / Setup & Deployments / Moves / Software Installs • Business Data Analytics / Reporting Applications (Business & Study) Systems Infrastructure / Cloud Services • Research IS Help/Request Resources: • Research Information Services portal: https: //www. research. chop. edu/research-information-services • Via Email: Research. IS@email. chop. edu *Please note: to report a problem with an existing service, contact the IS Help Desk / x: 4 HELP/ 215 -590 -4357 55

Enterprise (Hospital) Information Services • • Provides Enterprise Technology Services outside of our portfolio (e. g. ): • Network Access • VPN (remote access) • Email (Office 365) • Enterprise Applications (e. g. Workday, Coupa, Service. Now, etc. ) • Mobile Devices (e. g. mobile phone, etc. ) and Wired Phones • Conference Room Audio / Visual Enterprise IS Help/Request Resources: • Service. Now portal: https: //chop. service-now. com/esp • To request a new service or move / alter an existing (ex: mobile device, remote access, etc. ): • To report a problem with an existing service, please contact the IS Help Desk (x: 4 -HELP / 215 -590 -4357) Help for Research IS managed services requested through Enterprise (Hospital) IS will automatically route to the Research IS team 56

THANK YOU! 57
- Slides: 57