New Drug Update 2016 2017 Rachael Mc Caleb

New Drug Update 2016 -2017 Rachael Mc. Caleb, Pharm. D, BCPS Assistant Professor, Department of Pharmacy Practice University of Arkansas for Medical Sciences College of Pharmacy

Disclosures • I have nothing to disclose concerning possible financial or personal relationships with commercial entities that may have a direct or indirect interest in the subject matter of this presentation. 2

Pharmacist Objectives • Recognize drugs that gained FDA approval within the last year • Describe indications, doses, formulations, adverse effects, and drug interactions of recently approved drugs • Summarize clinical data for recently approved drugs • Identify information regarding the recently approved drugs that should be communicated to patients 3

Technician Objectives • List new drugs that gained FDA approval within the last 12 months • Identify doses and formulation of the recently approved drugs • Be able to state the indication for the recently approved drugs 4
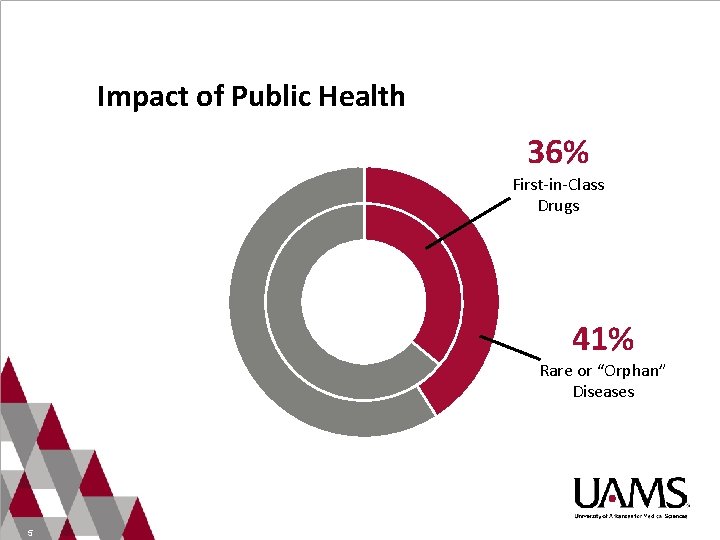
Impact of Public Health 36% First-in-Class Drugs 41% Rare or “Orphan” Diseases 5
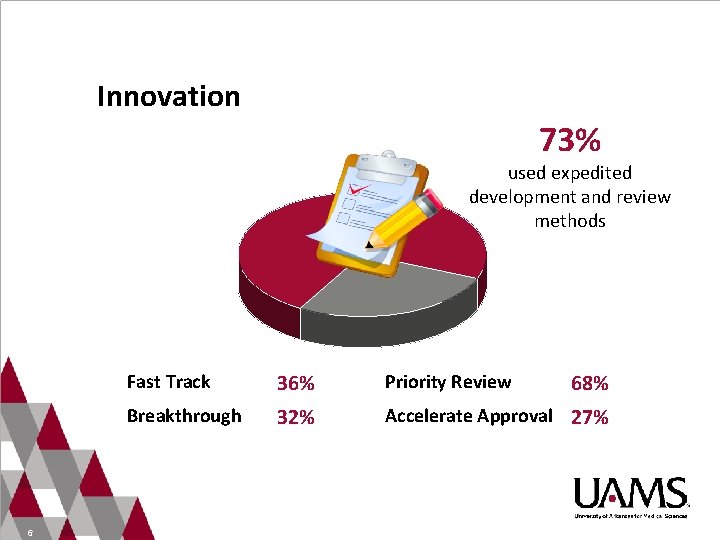
Innovation 73% used expedited development and review methods 6 Fast Track 36% Priority Review 68% Breakthrough 32% Accelerate Approval 27%
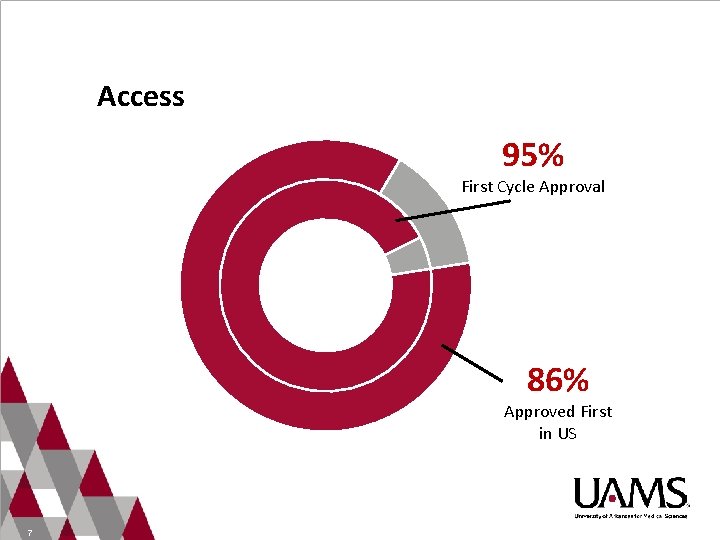
Access 95% First Cycle Approval 86% Approved First in US 7
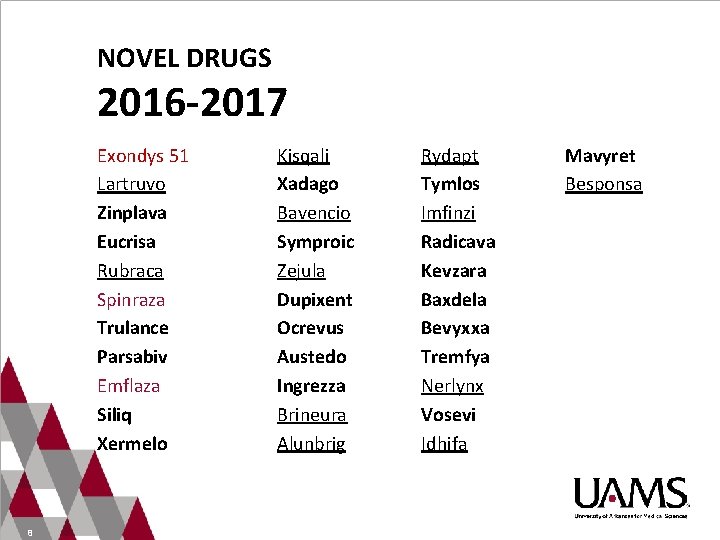
NOVEL DRUGS 2016 -2017 Exondys 51 Lartruvo Zinplava Eucrisa Rubraca Spinraza Trulance Parsabiv Emflaza Siliq Xermelo 8 Kisqali Xadago Bavencio Symproic Zejula Dupixent Ocrevus Austedo Ingrezza Brineura Alunbrig Rydapt Tymlos Imfinzi Radicava Kevzara Baxdela Bevyxxa Tremfya Nerlynx Vosevi Idhifa Mavyret Besponsa

Hepatology 9

Sofosbuvir/velpatasvir/voxilaprevir (Vosevi®) • Indication: Treatment of chronic hepatitis C virus (HCV) genotype 1, 2, 3, 4, 5, or 6 infection in adults without cirrhosis or with compensated cirrhosis who have previously been treated with an HCV regimen containing an NS 5 A inhibitor • Approval Date: July 18, 2017 • MOA: – Sofosbuvir is an inhibitor of the HCV NS 5 B RNA-dependent RNA polymerase – Velpatasvir is an inhibitor of HCV NS 5 A – Voxilaprevir is an inhibitor of hepatitis C virus (HCV) NS 3/4 A protease 10 Vosevi (sofosbuvir, velpatasvir, voxilaprevir) [prescribing information]. Foster City, CA: Gilead Sciences Inc; July 2017.

Sofosbuvir/velpatasvir/voxilaprevir (Vosevi®) • Dosage: One tablet (sofosbuvir 400 mg, velpatasvir 100 mg, and voxilaprevir 100 mg) once daily for 12 weeks • Boxed Warning: Risk of Hepatitis B reactivation • Adverse Reactions: Headache, fatigue, diarrhea, and nausea • Precautions: Bradycardia • Cost: ~$90, 000/treatment 11 Vosevi (sofosbuvir, velpatasvir, voxilaprevir) [prescribing information]. Foster City, CA: Gilead Sciences Inc; July 2017.
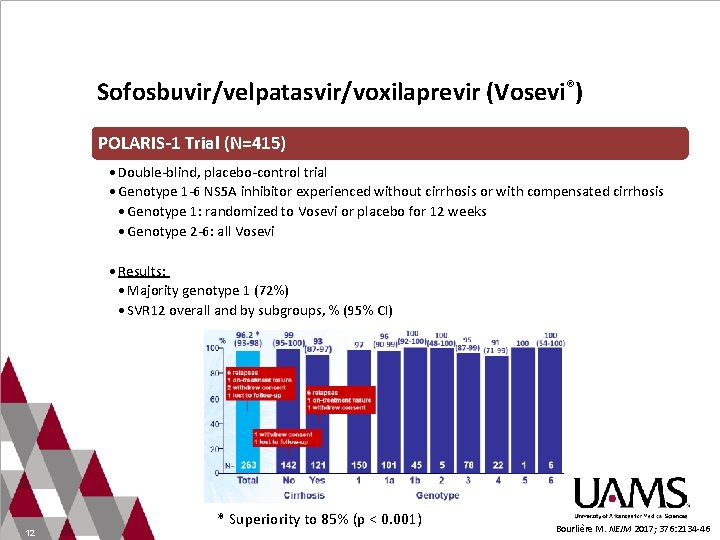
Sofosbuvir/velpatasvir/voxilaprevir (Vosevi®) POLARIS-1 Trial (N=415) • Double-blind, placebo-control trial • Genotype 1 -6 NS 5 A inhibitor experienced without cirrhosis or with compensated cirrhosis • Genotype 1: randomized to Vosevi or placebo for 12 weeks • Genotype 2 -6: all Vosevi • Results: • Majority genotype 1 (72%) • SVR 12 overall and by subgroups, % (95% CI) 12 * Superiority to 85% (p < 0. 001) Bourlière M. NEJM 2017; 376: 2134 -46
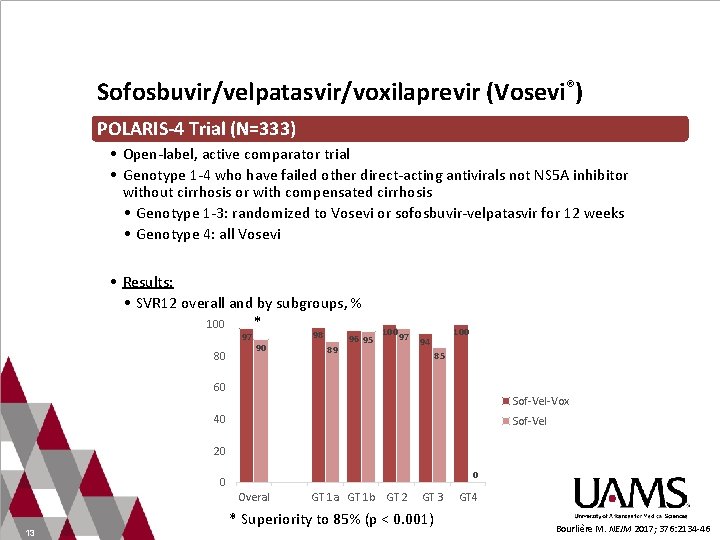
Sofosbuvir/velpatasvir/voxilaprevir (Vosevi®) POLARIS-4 Trial (N=333) • Open-label, active comparator trial • Genotype 1 -4 who have failed other direct-acting antivirals not NS 5 A inhibitor without cirrhosis or with compensated cirrhosis • Genotype 1 -3: randomized to Vosevi or sofosbuvir-velpatasvir for 12 weeks • Genotype 4: all Vosevi • Results: • SVR 12 overall and by subgroups, % 100 80 97 * 90 98 89 96 95 100 97 100 94 85 60 Sof-Vel-Vox 40 Sof-Vel 20 0 0 Overal 13 GT 1 a GT 1 b GT 2 GT 3 * Superiority to 85% (p < 0. 001) GT 4 Bourlière M. NEJM 2017; 376: 2134 -46

Sofosbuvir/velpatasvir/voxilaprevir (Vosevi®) POLARIS-2 & 3 Trials (N=219) • Open-label, active comparator trial • POLARIS 2: Genotype 1, 2 and 4 -6 without cirrhosis or with compensated cirrhosis • POLARIS 3: Genotype 3 who were direct-acting antivirals naïve with compensated cirrhosis • Randomized to Vosevi for 8 weeks or sofosbuvir-velpatasvir for 12 weeks • Results: • POLARIS 2: • SVR rate: Vosevi (95%) and sof-vel (98%) [Voseiv did not meet noninferiority to sof-vel] • POLARIS 3: • SVR rate was 96% for both treatment groups 14 Lacobson IM, et al. Gastroenterology. 2017; 153: 113 -22.

Glecaprevir/pibrentasvir (Mavyret®) • Indication: Treatment of chronic hepatitis C virus (HCV) genotype 1, 2, 3, 4, 5, or 6 infection in adults without cirrhosis or with compensated cirrhosis • Approval Date: August 3, 2017 • MOA: – Glecaprevir is an inhibitor of hepatitis C virus (HCV) NS 3/4 A protease – Pibrentasvir is an inhibitor of HCV NS 5 A 15 Mavyret (glecaprevir/pibrentasvir) [prescribing information]. North Chicago, IL: Abb. Vie Inc; August 2017.

Glecaprevir/pibrentasvir (Mavyret®) • Dosage: – Treatment naïve: 8 weeks (no cirrhosis) or 12 weeks (compensated cirrhosis) – Treatment experience: 8 -16 weeks depending on genotype and prior treatment • Boxed Warning: Risk of Hepatitis B reactivation • Adverse Reactions: Headache and fatigue • Cost: $15, 840/month https: //www. cbsnews. com/news/fda-approves-mavyret-abbvie-drug-to-treat-hepatitis-c/ 16 Mavyret (glecaprevir/pibrentasvir) [prescribing information]. North Chicago, IL: Abb. Vie Inc; August 2017.

Glecaprevir/pibrentasvir (Mavyret®) Clinical Trials Snapshot – ENDURANCE 1/3/4 (Treatment naïve without cirrhosis) – SURVEYOR 1/2 (Treatment naïve without cirrhosis or with compensated cirrhosis (only 2)) – EXPEDITION 1/4 (Treatment naïve with compensated cirrhosis) • 4 (CKD stage 4/5) – MAGELLAN 1 (NS 5 A experience without cirrhosis or with compensated cirrhosis) – ENDURANCE-3 • Open-label, active-control (sofosbuvir and daclatasvir) 17 Mavyret (glecaprevir/pibrentasvir) [prescribing information]. North Chicago, IL: Abb. Vie Inc; August 2017.

Glecaprevir/pibrentasvir (Mavyret®) Clinical Trials Snapshot – Results: • N=2300 • 92 -100% of patients who received Mavyret for 8, 12 or 16 weeks duration had no virus detected in the blood 12 weeks after finishing treatment – Overall 98 percent cure rate • 97. 5% cure rate – GT 1 -6 treatment naïve patients without cirrhosis after 8 weeks of treatment • 98% cure rate with compensated cirrhosis patients 18 Mavyret (glecaprevir/pibrentasvir) [prescribing information]. North Chicago, IL: Abb. Vie Inc; August 2017.

Dermatology 19

Crisaborole (Eucrisa®) • Indication: Topical treatment of mild to moderate atopic dermatitis in patients ≥ 2 years of age • Approval Date: December 14, 2016 • MOA: Inhibits phosphodiesterase 4 (PDE-4) and results in increased intracellular cyclic adenosine monophosphate (c. AMP) levels 20 Eucrisa (crisaborole) [prescribing information]. Palo Alto, CA: Anacor Pharmaceuticals Inc; December 2016.

Crisaborole (Eucrisa®) • Dosage: Apply a thin film to affected area(s) 2 times daily • Adverse Reactions: Application site pain • Cost: 2% ointment (60 g tube): $696. 00 21 Eucrisa (crisaborole) [prescribing information]. Palo Alto, CA: Anacor Pharmaceuticals Inc; December 2016.

Crisaborole (Eucrisa®) Clinical Trial Snapshot Trial 1 & 2 (N=1522) • Multicenter, double-blind, vehicle-control trials • Patients age 2 -79 years • Randomized to crisaborole or vehicle applied twice daily for 28 days • Results: Trial 1 Trial 2 Day 29 Crisaborole (N=503) Vehicle (N=256) Crisaborole (N=513) Vehicle (N=250) ISGA 0/1 w/2 -grade improvement from baseline 32. 8% 25. 4% 31. 4% 18. 0% Investigator's Static Global Assessment (ISGA) 22 Eucrisa (crisaborole) [prescribing information]. Palo Alto, CA: Anacor Pharmaceuticals Inc; December 2016.

Dupilumab (Dupixent®) • Indication: Treatment of adult moderate-severe atopic dermatitis • Approval Date: March 28, 2017 • MOA: Human monoclonal Ig. G 4 antibody targeting IL-4 Rα subunit, dual interleukin-4 (IL-4) and interleukin-13 (IL-13) cytokine antagonism JP Morgan Healthcare Conference, January 14, 2014 23 Dupixent (dupilumab) [prescribing information]. Tarrytown, NY: Regeneron Pharmaceuticals; March 2017.

Dupilumab (Dupixent®) • Dosage: 600 mg then 300 mg subcutaneous every other week • Adverse Events: Injection site reactions, conjunctivitis, blepharitis, oral herpes, keratitis, eye pruritus, and other herpes simplex virus infection • Precautions: Hypersensitivity, conjunctivitis/keratitis, and comorbid asthma • Cost: $3414/monthly for maintenance 24 Dupixent (dupilumab) [prescribing information]. Tarrytown, NY: Regeneron Pharmaceuticals; March 2017.
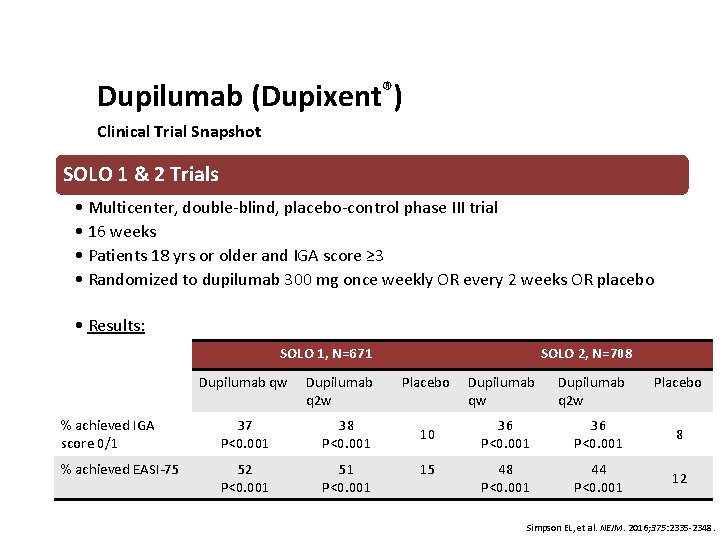
Dupilumab (Dupixent®) Clinical Trial Snapshot SOLO 1 & 2 Trials • Multicenter, double-blind, placebo-control phase III trial • 16 weeks • Patients 18 yrs or older and IGA score ≥ 3 • Randomized to dupilumab 300 mg once weekly OR every 2 weeks OR placebo • Results: SOLO 1, N=671 Dupilumab qw Dupilumab q 2 w % achieved IGA score 0/1 37 P<0. 001 38 P<0. 001 % achieved EASI-75 52 P<0. 001 51 P<0. 001 SOLO 2, N=708 Placebo Dupilumab qw 10 15 Dupilumab q 2 w Placebo 36 P<0. 001 8 48 P<0. 001 44 P<0. 001 12 Simpson EL, et al. NEJM. 2016; 375: 2335 -2348.

Dupilumab (Dupixent®) Clinical Trial Snapshot LIBERTY AD CHRONOS Trial (N=161) • Multicenter, double-blind, placebo-control phase III trial 52 week • Patients 18 years or older, ISGA score ≥ 3, EASI score ≥ 16 • Randomized to dupilumab 300 mg once weekly OR every 2 weeks OR placebo PLUS topical corticosteroids • Results: Dupilumab qw Dupilumab q 2 w Placebo % achieved ISGA score 0/1 40 P<0. 0001 36 P<0. 0001 13 % achieved EASI-75 64 P<0. 0001 65 P<0. 0001 22 Eczema Area and Severity Index (EASI) 26 Blauvelt A, et al. The Lancet, 2017; 389: 2287 -303.

Oxymetazoline hydrochloride (Rhofade®) • Indication: Treatment of persistent facial erythema associated with rosacea in adults • Approval Date: January 19, 2017 • MOA: Relatively selective alpha 1 A-receptor agonist that when applied topically may decrease erythema through direct vasoconstriction https: //www. rhofadehcp. com/persistent-erythema 27 Rhofade (oxymetazoline) [prescribing information]. Irvine, CA: Allergan; January 2017.

Oxymetazoline hydrochloride (Rhofade®) • Dosage: Apply a pea-sized amount to entire face once daily • Adverse Reactions: application site dermatitis, worsening inflammatory lesions of rosacea, and application site pruritus, erythema, and pain • Precautions: May impact blood pressure, potentiation of vascular insufficiency, and risk of angle closure glaucoma • Cost: 1% cream (30 g) $570. 00 28 Rhofade (oxymetazoline) [prescribing information]. Irvine, CA: Allergan; January 2017.

Oxymetazoline hydrochloride (Rhofade®) Clinical Trial Snapshot Trial 1 & 2 (N=885) • Double-blind, vehicle-controlled trials • Randomized to oxymetazoline or vehicle once daily for 29 days • Assessed success using the 5 -point clinician erythema assessment (CEA) scale and the 5 -point subject self-assessment (SSA) scale • Success defined as a 2 -grade improvement on both CEA and SSA • Results: 29 Trial 1 Trial 2 Time-point (hr) Rhofade (N=222) Vehicle (N=218) Rhofade (N=224) Vehicle (N=221) 3 12% 6% 14% 7% 6 16% 8% 13% 5% 9 18% 6% 16% 9% 12 15% 6% 12% 6% Rhofade (oxymetazoline) [prescribing information]. Irvine, CA: Allergan; January 2017.

Brodalumab (Siliq®) • Indication: Treatment of moderate to severe plaque psoriasis in adult patients • Approval Date: February 15, 2017 • MOA: Humanized Ig. G 2 monoclonal antibody that selectively binds with the interleukin 17 A (IL-17 A) cytokine and inhibits interaction with the IL-17 receptor – Inhibits the release of proinflammatory cytokines and chemokines 30 Siliq (brodalumab) [prescribing information]. Bridgewater, NJ: Valeant Pharmaceuticals; February 2017.

Brodalumab (Siliq®) • Dosage: 210 mg subcutaneous at weeks 0, 1, and 2 then 210 mg once every 2 weeks – Discontinue if inadequate response after week 12 or 16 • Boxed Warning: Suicidal ideation and behavior • Adverse Events: arthralgia, headache, fatigue, neutropenia, influenza, and tinea infections • Precautions: Infections, tuberculosis, Crohn’s disease, and immunizations • Cost: $6300 then $4200/month 31 Siliq (brodalumab) [prescribing information]. Bridgewater, NJ: Valeant Pharmaceuticals; February 2017.

Brodalumab (Siliq®) Clinical Trial Snapshot AMAGINE 2 & 3 Trials • Multicenter, double-blind, placebo-control phase III • Patients with moderate to severe plaque psoriasis • Randomized to brodalumab 140 or 210 mg, ustekinumab, or placebo every 2 weeks for 12 weeks • Results: PASI 75 % (95% CI), p value vs UST s. PGA score 0 -1 % (95% CI), p value vs UST PASI 100 % (95% CI), p value vs UST 32 AMAGINE-2 UST BRO 140 N=300 N=610 70 (6567 (6375) 70), 0. 33 BRO 210 PLB N=612 N=315 86 (83 -89), 6 (4 -9) 0. 08 AMAGINE-3 UST BRO 140 N=313 N=629 69 (6469 (65 -73), 74) 0. 95 4 (2 -7) 61 (5567) 58 (5462), 0. 49 79 (75 -82), 4 (2 -7) <0. 001 57 (5263) 60 (56 -64), 80 (76 -83), 0. 44 <0. 001 1 (0 -2) 22 (1727) 26 (2229), 0. 08 44 (41 -49), 0. 3 (0 -2) <0. 001 19 (1423) 27 (24 -31), 37 (33 -41), 0. 007 <0. 001 PLB N=309 8 (5 -12) BRO 210 N=624 85 (82 -88), 0. 007 Psoriasis Area Severity Index (PASI); Static Physicians Global Assessment (s. PGA) Lebwohl, M, et al. NEJM, 373: 1318 -1328.

Guselkumab (Tremfya®) • Indication: Treatment of moderate to severe plaque psoriasis in adult patients • Approval Date: July 13, 2017 • MOA: Humanized Ig. G 2 monoclonal antibody that selectively binds with the interleukin IL-23 cytokine and inhibits interaction with the IL-23 receptor – Inhibits the release of proinflammatory cytokines and chemokines https: //www. tremfyahcp. com/plaque-psoriasis/mechanism-of-action. html 33 Tremfya (guselkumab) [prescribing information]. Horsham, PA: Janssen Biotech, Inc; July 2017.

Guselkumab (Tremfya®) • Dosage: 100 mg subcutaneous at weeks 0 & 4, then every 8 weeks • Adverse Reactions: Upper respiratory infections, headache, injection site reactions, arthralgia, diarrhea, gastroenteritis, tinea infections, and herpes simplex infections • Precautions: Infections, tuberculosis • Cost: $11, 620. 80/dose – $81, 345. 60 annually 34 Tremfya (guselkumab) [prescribing information]. Horsham, PA: Janssen Biotech, Inc; July 2017.

Guselkumab (Tremfya®) Clinical Trial Snapshot VOYAGE 1 & 2 Trials • Multicenter, double-blind phase III trials • Moderate to severe plaque psoriasis • Guselkumab 100 mg, adalimumab, or placebo • Study design VOYAGE 1 VOYAGE 2 Blauvelt, A, et al. J Am Acad Dermatol, 76: 405 -417. Reich K, et al. J Am Acad Dermatol, 76: 418 -431.
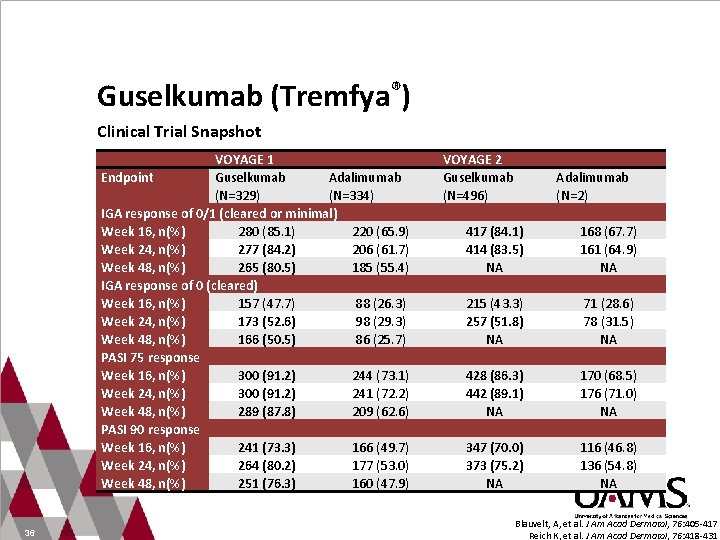
Guselkumab (Tremfya®) Clinical Trial Snapshot Endpoint VOYAGE 1 Guselkumab Adalimumab (N=329) (N=334) IGA response of 0/1 (cleared or minimal) Week 16, n(%) 280 (85. 1) 220 (65. 9) Week 24, n(%) 277 (84. 2) 206 (61. 7) Week 48, n(%) 265 (80. 5) 185 (55. 4) IGA response of 0 (cleared) Week 16, n(%) 157 (47. 7) 88 (26. 3) Week 24, n(%) 173 (52. 6) 98 (29. 3) Week 48, n(%) 166 (50. 5) 86 (25. 7) PASI 75 response Week 16, n(%) 300 (91. 2) 244 (73. 1) Week 24, n(%) 300 (91. 2) 241 (72. 2) Week 48, n(%) 289 (87. 8) 209 (62. 6) PASI 90 response Week 16, n(%) 241 (73. 3) 166 (49. 7) Week 24, n(%) 264 (80. 2) 177 (53. 0) Week 48, n(%) 251 (76. 3) 160 (47. 9) 36 VOYAGE 2 Guselkumab (N=496) Adalimumab (N=2) 417 (84. 1) 414 (83. 5) NA 168 (67. 7) 161 (64. 9) NA 215 (43. 3) 257 (51. 8) NA 71 (28. 6) 78 (31. 5) NA 428 (86. 3) 442 (89. 1) NA 170 (68. 5) 176 (71. 0) NA 347 (70. 0) 373 (75. 2) NA 116 (46. 8) 136 (54. 8) NA Blauvelt, A, et al. J Am Acad Dermatol, 76: 405 -417. Reich K, et al. J Am Acad Dermatol, 76: 418 -431.

Guselkumab (Tremfya®) Clinical Trial Snapshot NAVIGATE Trial (N=853) • • Multicenter, double-blind phase III trial Adult patients with moderate to severe plaque psoriasis All patients received ustekinumab at Weeks 0 and 4 At week 16, patients with inadequate response to ustekinumab (n=268) • Randomized to continue ustekinumab or guselkumab 100 mg • Responders continue ustekinumab (n=585) • Results: • Higher mean number of visits at which patients had an IGA score of 0 or 1 compared to ustekinumab (1. 5 vs 0. 7; p<0. 001) • More patients achieving an IGA score of 0 or 1 compared to ustekinumab (31. 1% vs 14. 3%; p<0. 001) • Greater proportions of guselkumab patients achieved PASI 75/90/100 37 Langley RG, et al. Br J Dermatol. Published Online First. [7/21/2017]

Infection Disease 38

Bezlotoxumab (Zinplava®) • Indication: Reduce recurrence of Clostridium difficile infection (CDI) in patients who are receiving standard-of-care treatment for CDI • Approval Date: October 21, 2016 • MOA: Human Ig. G 1 monoclonal antibody which binds to C. difficile toxin B and neutralizes it to prevent its toxic effects. http: //en. pharmacodia. com/web/drug/1_9806. html 39 Zinplava (bezlotoxumab) [prescribing information]. Whitehouse Station, NJ; Merck & Co, Inc: October 2016.

Bezlotoxumab (Zinplava®) • Dosage: 10 mg/kg single infusion during antibacterial treatment for CDI • Pharmacokinetics: Elimination half-life of ~19 days • Adverse Reactions: Nausea, pyrexia, and headache • Precautions: Exacerbation of heart failure (HF) • Cost: 100 kg patient - $4560. 00 40 Zinplava (bezlotoxumab) [prescribing information]. Whitehouse Station, NJ; Merck & Co, Inc: October 2016.

Bezlotoxumab (Zinplava®) Clinical Trial Snapshot MODIFY 1 & 2 Trials (N=2655) • Multicenter, double-blind, placebo control trials • Adults with primary or recurrent C. difficile infections • Randomized to single dose bezlotoxumab (10 mg/kg), actoxumab plus bezlotoxumab (10 mg/kg), or placebo • MODIFY 1 included actoxumab 10 mg/kg arm [data suggest lack of efficacy and not included in MODIFY 2] • Also received standard-of-care antibiotics (metronidazole, vancomycin, or fidaxomicin) for 10 to 14 days 41 Wilcox MH, et al. NEJM. 2017; 376: 305 -17.
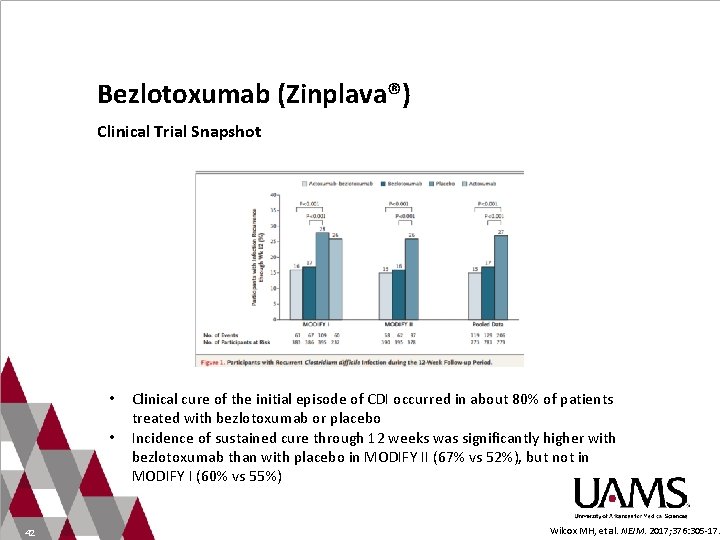
Bezlotoxumab (Zinplava®) Clinical Trial Snapshot • • 42 Clinical cure of the initial episode of CDI occurred in about 80% of patients treated with bezlotoxumab or placebo Incidence of sustained cure through 12 weeks was significantly higher with bezlotoxumab than with placebo in MODIFY II (67% vs 52%), but not in MODIFY I (60% vs 55%) Wilcox MH, et al. NEJM. 2017; 376: 305 -17.
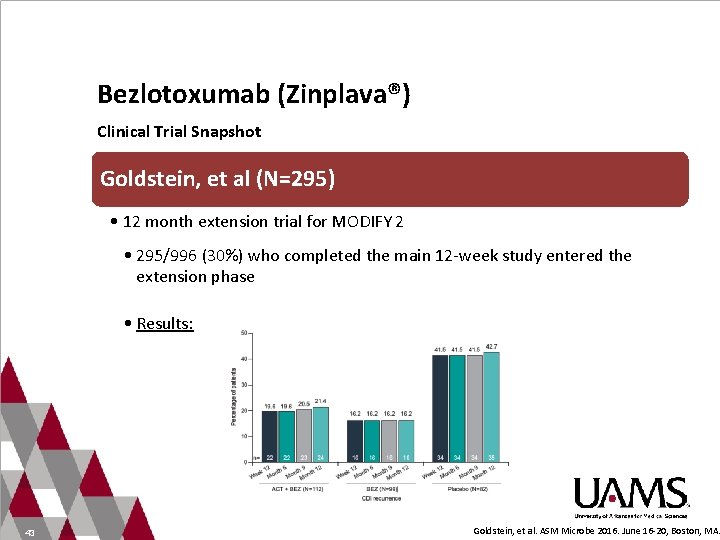
Bezlotoxumab (Zinplava®) Clinical Trial Snapshot Goldstein, et al (N=295) • 12 month extension trial for MODIFY 2 • 295/996 (30%) who completed the main 12 -week study entered the extension phase • Results: 43 Goldstein, et al. ASM Microbe 2016. June 16 -20, Boston, MA.

Delafloxacin (Baxdela®) • Indication: Treatment of acute bacterial skin and skin structure infections • Approval Date: June 19, 2017 • MOA: Inhibits DNA gyrase (topoisomerase II) and topoisomerase IV enzymes • Unique Characteristics: – No issue with QTc interval or other cardiac events and phototoxicity – Activity is enhanced in acidic conditions 44 Baxdela (delafloxacin) [prescribing information]. Lincolnshire, IL: Melinta Therapeutics Inc; June 2017. http: //www. contagionlive. com/news/what-makes-delafloxacin-a-unique-antibiotic-for-skin-infections

Delafloxacin (Baxdela®) • Dosage: – Oral – 450 mg every 12 hours for 5 to 14 days – IV – 300 mg every 12 hours for 5 to 14 days – Renal adjustments: • GFR 15 – 29 m. L/min: IV 200 mg every 12 hours • GFR <15 m. L/min: use not recommended • Adverse Reactions: Nausea, diarrhea, headache, transaminase elevations and vomiting • Cost: Not currently available 45 Baxdela (delafloxacin) [prescribing information]. Lincolnshire, IL: Melinta Therapeutics Inc; June 2017.

Delafloxacin (Baxdela®) Clinical Trial Snapshot Study 1 (N=660) • Multicenter, double-blind trial • Patient with acute bacterial skin and skin structure infections • Randomized to IV delafloxacin 300 mg q 12 hr OR vancomycin plus aztreonam Study 2 (N=850) • Multicenter, double-blind trial • Patient with acute bacterial skin and skin structure infections • Randomized to IV delafloxacin 300 mg q 12 hr (6 doses) then oral 450 mg q 12 hr OR vancomycin plus aztreonam 46 Baxdela (delafloxacin) [prescribing information]. Lincolnshire, IL: Melinta Therapeutics Inc; June 2017.
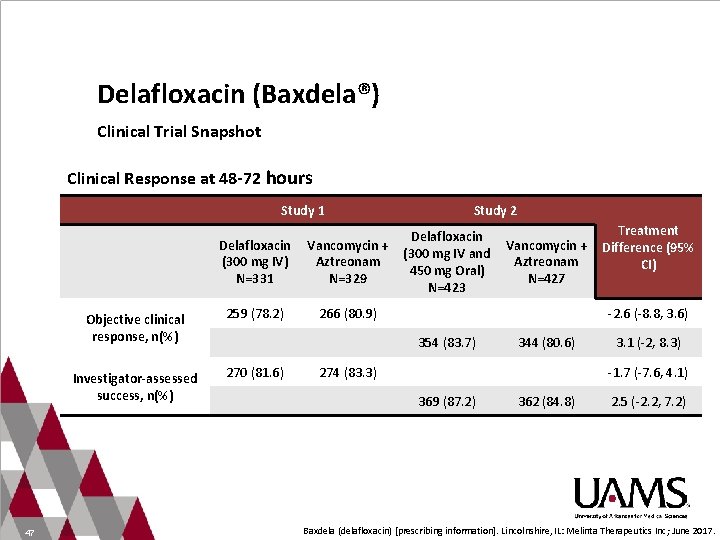
Delafloxacin (Baxdela®) Clinical Trial Snapshot Clinical Response at 48 -72 hours Study 1 Study 2 Treatment Delafloxacin Vancomycin + Difference (95% (300 mg IV and (300 mg IV) Aztreonam CI) 450 mg Oral) N=331 N=329 N=427 N=423 47 Objective clinical response, n(%) 259 (78. 2) Investigator-assessed success, n(%) 270 (81. 6) 266 (80. 9) -2. 6 (-8. 8, 3. 6) 354 (83. 7) 344 (80. 6) 274 (83. 3) 3. 1 (-2, 8. 3) -1. 7 (-7. 6, 4. 1) 369 (87. 2) 362 (84. 8) 2. 5 (-2. 2, 7. 2) Baxdela (delafloxacin) [prescribing information]. Lincolnshire, IL: Melinta Therapeutics Inc; June 2017.

Cardiology 48

Betrixaban (Bevyxxa®) • Indication: Prophylaxis of VTE in adults hospitalized for an acute medical illness who are at risk for thromboembolic complications • Approval Date: June 23, 2017 • MOA: Factor Xa (FXa) inhibitor, selectively blocks the active site of Fxa, decreases thrombin generation (TG) 49 Bevyxxa (betrixaban) [prescribing information]. South San Francisco, CA: Portola Pharmaceuticals Inc; June 2017.

Betrixaban (Bevyxxa®) • Dosage: 160 mg as a single dose on day 1, followed by 80 mg once daily for 35 to 42 days – Cr. Cl 15 -30 m. L/min: reduce dose by 50% • Adverse Reactions: Bleeding • Precautions: Risk of bleeding, severe renal impairment, concomitant P-gp inhibitors • Cost: Not currently available 50 Bevyxxa (betrixaban) [prescribing information]. South San Francisco, CA: Portola Pharmaceuticals Inc; June 2017.

Betrixaban (Bevyxxa®) Clinical Trial Snapshot APEX Trial (N=7513) • Multicenter, double-blinded, active-control trial • Patients 40 years or older hospitalized for at 96 hours for acute medical illness with reduced mobility or risk factors for VTE • Randomized to enoxaparin 40 mg daily 10± 4 days plus oral placebo OR enoxaparin placebo daily 10± 4 days plus oral betrixaban 160 mg then 80 mg for 35 -42 days • Analyses performed on different cohorts • Cohort 1: elevated D-dimer level • Cohort 2: age 75 years or older 51 Cohen AT, et al. NEJM. 2016; 375: 534 -44.
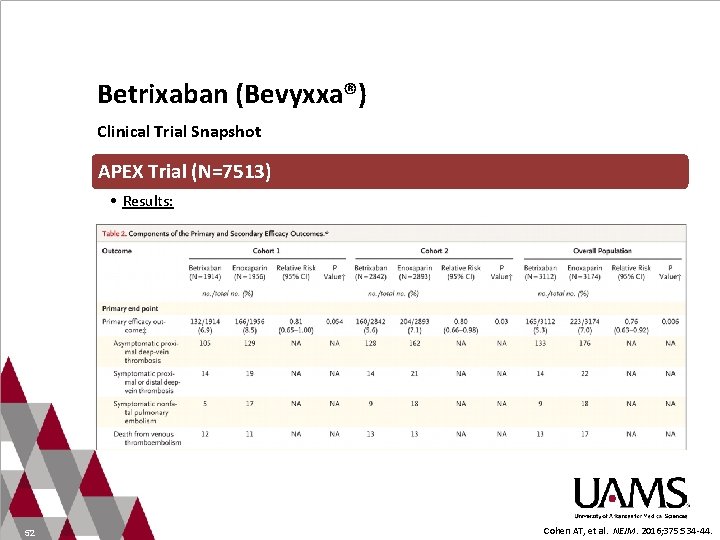
Betrixaban (Bevyxxa®) Clinical Trial Snapshot APEX Trial (N=7513) • Results: 52 Cohen AT, et al. NEJM. 2016; 375: 534 -44.

Immunology 53

Sarilumab (Kevzara®) • Indication: Treatment of moderate to severe active rheumatoid arthritis in adults who have had an inadequate response or intolerance to one or more DMARDs • Approval Date: May 22, 2017 • MOA: Inhibitor of interleukin-6 (IL-6) mediated signaling through s. IL-6 R and m. IL-6 R, leads to reduction in CRP levels 54 Kevzara (sarilumab) [prescribing information]. Bridgewater, NJ: Sanofi-Aventis; May 2017.

Sarilumab (Kevzara®) • Dosage: 200 mg subcutaneous every 2 weeks • Adverse Reactions: Neutropenia, increased ALT, injection site erythema, upper respiratory infections and urinary tract infections • Precautions: Serious infections, lab abnormalities, GI perforation, live vaccines • Cost: $43, 200 annually 55 Kevzara (sarilumab) [prescribing information]. Bridgewater, NJ: Sanofi-Aventis; May 2017.

Sarilumab (Kevzara®) Clinical Trial Snapshot Genovese et al. (N=1197) • Moderate to severe RA • Sarilumab 200 or 150 mg OR placebo q 2 wks • Concomitant MTX • Results: • DAS 28 -CRP <2. 6 (remission) at 24 weeks • Sarilumab 200 mg: 33. 1% Sarilumab 150 mg: 37. 8% Placebo: 20% • DAS 28 -CRP <2. 6 (remission) at 52 weeks • Sarilumab 200 mg: 55. 6% Sarilumab 150 mg: 47. 8% Placebo: 38. 7% • Sarilumab resulted in less radiographic progression of damage compared to placebo 56 Disease Activity Score 28 -joint count C reactive protein (DAS 28 -CRP) Genovese MC, et al. Arthritis Rheumatol. 2015; 67: 1424 -1437.

Sarilumab (Kevzara®) Clinical Trial Snapshot TARGET Trial (N=546) • Moderate to severe RA • Sarilumab 200 or 150 mg OR placebo q 2 wks • Concomitant methotrexate • Results: • Statistically significant improvements in ACR 20/50/70 at 24 weeks compared to placebo • Statistically significant improvements in DAS 28 -CRP and greater proportion achieved remission (DAS 28 -CRP <2. 6) compared to placebo • Statistically significant and clinically relevant improvements in physical function compared with placebo 57 Fleischmann R, et al. Arthritis Rheumatol. 2017; 69: 277 -90.

Sarilumab (Kevzara®) Clinical Trial Snapshot MONARCH Trial (N=369) • Moderate to severe RA • Sarilumab 200 mg or adalimumab 40 mg q 2 wks • Results: • Sarilumab was superior to adalimumab in the primary end point of change from baseline in DAS 28 -ESR (− 3. 28 vs − 2. 20; p<0. 0001) • Statistically significant improvements in ACR 20/50/70 response rates compared to adalimumab • Greater proportion achieved remission compared to adalimumab (7. 1% vs 2. 7%; p=0. 0468) • Neutropenia occurred more frequently with sarilumab compared to adalimumab but no significant difference in infection rates 58 Burmester GR, et al. Ann Rheum Dis. Published Online First: [11/17/2016].

Endocrinology 59

Dapagliflozin/saxagliptin (Qtern®) • Indication: Improve glycemic control in adults with type 2 diabetes mellitus (T 2 DM) • Approval Date: February 28, 2017 • MOA: – Dapagliflozin, a sodium-glucose cotransporter 2 (SGLT-2) inhibitor – Saxagliptin, a dipeptidyl peptidase-4 (DPP-4) inhibitor 60 Dapagliflozin/saxagliptin (Qtern) [prescribing information]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP ; February 2017.

Dapagliflozin/saxagliptin (Qtern®) • Dosage: 10 mg dapagliflozin/5 mg saxagliptin tablet once daily – Discontinue if GFR <60 m. L/min • Adverse Reactions: Upper respiratory tract infection, urinary tract infection, and dyslipidemia • Precautions: Pancreatitis, heart failure, hypotension, ketoacidosis, AKI, UTI, hypoglycemia, increased LDL, bladder cancer • Cost: Not currently available 61 Dapagliflozin/saxagliptin (Qtern) [prescribing information]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP ; February 2017.

Dapagliflozin/saxagliptin (Qtern®) Clinical Trial Snapshot Combination of saxagliptin and dapagliflozin with metformin versus placebo • Statistically significant reductions in Hb. A 1 c in comparison to patients treated with placebo Combination of saxagliptin and dapagliflozin added to metformin versus saxagliptin or dapagliflozin alone added to metformin • Statistically superior reductions in Hb. A 1 c in comparison to patients treated with single agent plus metformin 62 Dapagliflozin/saxagliptin (Qtern) [prescribing information]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP ; February 2017.

Etelcalcetide (Parsabiv®) • Indication: Treatment of secondary hyperparathyroidism (HPT) in adults with chronic kidney disease (CKD) on hemodialysis • Approval Date: February 8, 2017 • MOA: Synthetic peptide calcimimetic, activates the calcium-sensing receptor (Ca. SR) on the parathyroid gland, resulting in decreased PTH secretion 63 Parsabiv (etelcalcetide) [prescribing information]. Thousand Oaks, CA: Amgen Inc; February 2017.

Etelcalcetide (Parsabiv®) • Dosage: 5 mg IV bolus 3 times weekly following hemodialysis – Maximum maintenance dose – 15 mg 3 times weekly • Minimum maintenance dose – 2. 5 mg 3 times weekly • Adverse Reactions: Hypocalcemia, muscle spasms, diarrhea, nausea, vomiting, headache, and paresthesia • Precautions: Hypocalcemia (sometimes severe), worsening heart failure, upper GI bleeding, and adynamic bone • Cost: Anticipated availability is currently undetermined 64 Parsabiv (etelcalcetide) [prescribing information]. Thousand Oaks, CA: Amgen Inc; February 2017.

Etelcalcetide (Parsabiv®) Clinical Trial Snapshot Study 1 & 2 (N=1023) • CKD patients with secondary hyperparathyroidism receiving hemodialysis • IV etelcalcetide thrice weekly (max dose 15 mg) • PTH target < 300 pg/m. L • Results: • >30% reduction from baseline in PTH during the etelcalcetide: 77% versus 11% in Study 1, and 79% versus 11% in Study 2 • PTH levels of 300 pg/m. L or less during the etelcalcetide: 52% versus 6% in Study 1, and 56% versus 5% in Study 2 65 Parsabiv (etelcalcetide) [prescribing information]. Thousand Oaks, CA: Amgen Inc; February 2017.

Etelcalcetide (Parsabiv®) Clinical Trial Snapshot Block et al (NCT 1896232) (N=683) • Randomized, double-blind, active-control trial • IV etelcalcetide vs oral cinacalcet for 26 weeks • Results: • >30% reduction in PTH from baseline • Etelcalcetide (68. 2%) • Cinacalcet (57. 7%) • P for noninferiority, <0. 001; P for superiority, 0. 004 • Etelcalcetide treated patients achieved lower calcium and phosphate concentrations at week 26 • Heart failure events: • 10 (3%) etelcalcetide: 5(serious) • 2 (0. 6%) cinacalcet: 1 (serious) 66 Block GA, et al. JAMA; 317: 156 -164.

Gastroenterology 67

Plecanatide (Trulance®) • Indication: Treatment of chronic idiopathic constipation in adult patients • Approval Date: January 19, 2017 • MOA: Plecanatide and its active metabolite bind agonize guanylate cyclase-C on the luminal surface of intestinal epithelium, intestinal fluid increases and GI transit time is increased – Same class as linaclotide (Linzess®) 68 Trulance (plecanatide) [prescribing information]. New York, NY: Synergy Pharmaceuticals; January 2017.

Plecanatide (Trulance®) • Dosage: 3 mg once daily – Can be crushed • Boxed Warning: Risk of serious dehydration in pediatric patients • Adverse Reactions: Diarrhea, abdominal distension and tenderness, flatulence and increases in ALT and AST • Cost: $424. 17/ 30 tablets 69 Trulance (plecanatide) [prescribing information]. New York, NY: Synergy Pharmaceuticals; January 2017.
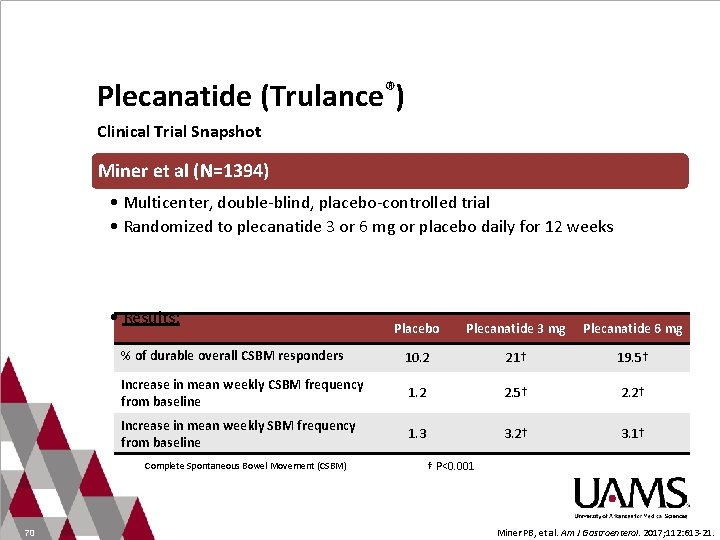
Plecanatide (Trulance®) Clinical Trial Snapshot Miner et al (N=1394) • Multicenter, double-blind, placebo-controlled trial • Randomized to plecanatide 3 or 6 mg or placebo daily for 12 weeks • Results: Placebo Plecanatide 3 mg Plecanatide 6 mg % of durable overall CSBM responders 10. 2 21† 19. 5† Increase in mean weekly CSBM frequency from baseline 1. 2 2. 5† 2. 2† Increase in mean weekly SBM frequency from baseline 1. 3 3. 2† 3. 1† Complete Spontaneous Bowel Movement (CSBM) 70 † P<0. 001 Miner PB, et al. Am J Gastroenterol. 2017; 112: 613 -21.

Telotristat etiprate (Xermelo®) • Indication: Treatment of carcinoid syndrome diarrhea (in combination with somatostatin analog therapy) in adults with symptoms inadequately controlled by somatostatin analog therapy • Approval Date: February 28, 2017 • MOA: Inhibits tryptophan hydroxylase (TPH), TPH is the rate-limiting enzyme in serotonin synthesis, reducing the frequency of carcinoid syndrome diarrhea 71 Xermelo (telotristat ethyl) [prescribing information]. The Woodlands, TX: Lexicon Pharmaceuticals Inc; February 2017.

Telotristat etiprate (Xermelo®) • Dosage: 250 mg by mouth three times daily – Can use with octreotide: administer octreotide at least 30 minutes after • Adverse Reactions: Headache and nausea • Cost: $6196. 80 per month 72 Xermelo (telotristat ethyl) [prescribing information]. The Woodlands, TX: Lexicon Pharmaceuticals Inc; February 2017.

Telotristat etiprate (Xermelo®) Clinical Trial Snapshot TELESTAR Trial (N=135) • Double-blind, placebo-controlled phase III trial • 12 weeks, additional 24 weeks open-label extension • Patients with 4 -12 bowel movements/day on somatostatin analogs • 1: 1: 1 randomization to placebo, telotristat etiprate 250 mg or 500 mg • Results: • Statistically significant decrease in bowel movements/day (placebo= -0. 9, 250 mg dose= -1. 7, 500 mg dose= -2. 1) p<0. 001 • Odds Ratio: 250 mg: 3. 49 (95% CI, 1. 33 to 9. 16) and 500 mg: 3. 11 (95% CI, 1. 20 to 8. 10) • Statistically significant reductions in u 5 -HIAA levels for both telotristat etiprate doses compared to placebo 73 Kulke MH, et al. J Clin Oncol. 2017; 35: 14 -23.

Naldemedine (Symproic®) • Indication: Treatment of opioid-induced constipation (OIC) in adults with chronic noncancer pain • Approval Date: March 23, 2017 • MOA: Functions as a peripherally acting mu-opioid receptor antagonist, decrease the constipating effects of opioids 74 Symproic (naldemedine) [prescribing information]. Stamford, CT: Purdue Pharma; August 2017.

Naldemedine (Symproic®) • Dosage: 0. 2 mg by mouth daily • Adverse Reactions: Abdominal pain, diarrhea, and nausea • Precautions: – Gastrointestinal Perforations: reported with use of other peripherally acting opioid antagonist agents – Opioid Withdrawal: patients with disruptions to the blood-brain barrier at increased risk • Cost: Not available 75 Symproic (naldemedine) [prescribing information]. Stamford, CT: Purdue Pharma; August 2017.

Naldemedine (Symproic®) Clinical Trial Snapshot COMPOSE 1 & 2 Trials (N=1100) • Multicenter, double-blind, phase III trials • Patients with stable opioid regimen (≥ 30 mg morphine equivalent) for ≥ 1 month • Randomized to receive naldemedine 0. 2 mg or placebo for 12 weeks • Results: Responders, n(%) COMPOSE 1 COMPOSE 2 Naldemedine Placebo (N=273) (N=272) Naldemedine Placebo (N=276) (N=274) 130 (48) 94(35) 13 (5, 21) p=0. 002 145 (53) 76 Treatment Difference (95% CI) 92 (34) 19 (11, 27) P<0. 0001 Hale M, et al. Lancet Gastroenterol Hepatol. 2017; 2: 555 -564.

Musculoskeletal 77

Ocrelizumab (Ocrevus®) • Indication: Treatment of relapsing or primary progressive forms of multiple sclerosis (PPMS) in adult patients • Approval Date: March 28, 2017 • MOA: Selectively targets and binds to the cell surface to deplete CD 20 expressing B-cells through antibody-dependent cell-mediated phagocytosis and cytotoxicity, as well as complement-mediated cytolysis Themswire. com 78 Ocrevus (ocrelizumab) [prescribing information]. South San Francisco, CA: Genetech, Inc; March 2017.

Ocrelizumab (Ocrevus®) • Dosage: – Start dose: 300 mg intravenous infusion, followed two weeks later by a second 300 mg intravenous infusion – Subsequent doses: 600 mg intravenous infusion every 6 months • Adverse Reactions: – RMS: Upper respiratory tract infections and infusion reactions – PPMS: Upper/lower respiratory tract infections, infusion reactions, and skin infections • Precautions: Infusion reactions, infections, and malignancies • Cost: $19, 500/300 mg 79 Ocrevus (ocrelizumab) [prescribing information]. South San Francisco, CA: Genetech, Inc; March 2017.

Ocrelizumab (Ocrevus®) Clinical Trial Snapshot ORATORIO Trial (N=732) • Age 18 -55 with PPMS • Randomized to receive IV ocrelizumab 600 mg or placebo • Results: • Fewer patients had disability progression confirmed at 12 weeks compared to placebo (32. 9 vs 39. 3%; HR 0. 76 (0. 59, 0. 98) RRR 24%; p=0. 03) • Fewer patients had disability progression confirmed at 24 weeks compared to placebo (29. 6 vs 35. 7%; HR 0. 75 (0. 58, 0. 98) RRR 25%; p=0. 04) • At week 120, total volume of brain lesions decreased by 3. 4% with ocrelizumab and increased by 7. 4% with placebo (P<0. 001) 80 Montalban X, et al. NEJM. 2017; 376: 209 -220.

Ocrelizumab (Ocrevus®) Clinical Trial Snapshot OPERA 1 & 2 Trials (N=1656) • Age 18 -55 with RMS • Randomized to receive IV ocrelizumab 600 mg q 24 wks or interferon beta-1 a 44 mcg three times weekly • Results: • Annualized relapse rate was lower with ocrelizumab vs interferon beta-1 a • OPERA 1: 0. 16 vs. 0. 29; 46% lower rate with ocrelizumab; P<0. 001 • OPERA 2: 0. 16 vs. 0. 29; 47% lower rate; P<0. 001 • Fewer patients had disability progression confirmed at 12 and 24 weeks compared to interferon beta-1 a • Mean number of MRI confirmed throughout week 96 • OPERA 1: 94% lower number of lesions with ocrelizumab, P<0. 001 • OPERA 2: 95% lower number of lesions, P<0. 001 81 Hauser SL, et al. NEJM. 2017; 376: 221 -234.

Abaloparatide (Tymlos®) • Indication: Treatment of postmenopausal women with osteoporosis at high risk for fracture • Approval Date: April 28, 2017 • MOA: Analog of human parathyroid hormone peptide, stimulates of osteoblast function and increased bone mass 82 Tymlos (abaloparatide) [prescribing information]. Waltham, MA: Radius Health; April 2017.

Abaloparatide (Tymlos®) • Dosage: 80 mcg subcutaneous once daily – Supplied in prefilled pen that delivers 30 daily doses • Boxed Warning: Risk of osteosarcoma • Adverse Reactions: Hypercalciuria, dizziness, nausea, headache, palpitations, fatigue, upper abdominal pain and vertigo • Precautions: Orthostatic hypotension, hyperca; cemia, and hypercalciuria • Cost: $1950/month 83 Tymlos (abaloparatide) [prescribing information]. Waltham, MA: Radius Health; April 2017.
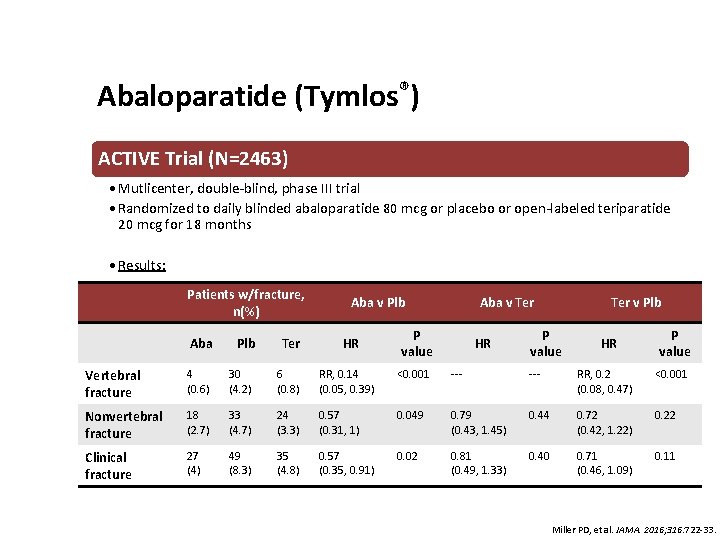
Abaloparatide (Tymlos®) ACTIVE Trial (N=2463) • Mutlicenter, double-blind, phase III trial • Randomized to daily blinded abaloparatide 80 mcg or placebo or open-labeled teriparatide 20 mcg for 18 months • Results: Patients w/fracture, n(%) Aba v Plb HR Aba v Ter P value HR Ter v Plb P value HR P value Aba Plb Ter Vertebral fracture 4 (0. 6) 30 (4. 2) 6 (0. 8) RR, 0. 14 (0. 05, 0. 39) <0. 001 --- RR, 0. 2 (0. 08, 0. 47) <0. 001 Nonvertebral fracture 18 (2. 7) 33 (4. 7) 24 (3. 3) 0. 57 (0. 31, 1) 0. 049 0. 79 (0. 43, 1. 45) 0. 44 0. 72 (0. 42, 1. 22) 0. 22 Clinical fracture 27 (4) 49 (8. 3) 35 (4. 8) 0. 57 (0. 35, 0. 91) 0. 02 0. 81 (0. 49, 1. 33) 0. 40 0. 71 (0. 46, 1. 09) 0. 11 Miller PD, et al. JAMA. 2016; 316: 722 -33.
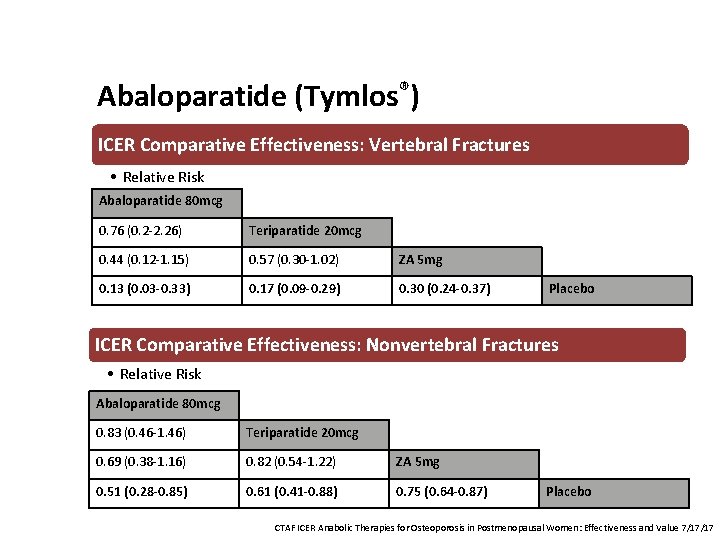
Abaloparatide (Tymlos®) ICER Comparative Effectiveness: Vertebral Fractures • Relative Risk Abaloparatide 80 mcg 0. 76 (0. 2 -2. 26) Teriparatide 20 mcg 0. 44 (0. 12 -1. 15) 0. 57 (0. 30 -1. 02) ZA 5 mg 0. 13 (0. 03 -0. 33) 0. 17 (0. 09 -0. 29) 0. 30 (0. 24 -0. 37) Placebo ICER Comparative Effectiveness: Nonvertebral Fractures • Relative Risk Abaloparatide 80 mcg 0. 83 (0. 46 -1. 46) Teriparatide 20 mcg 0. 69 (0. 38 -1. 16) 0. 82 (0. 54 -1. 22) ZA 5 mg 0. 51 (0. 28 -0. 85) 0. 61 (0. 41 -0. 88) 0. 75 (0. 64 -0. 87) Placebo CTAF ICER Anabolic Therapies for Osteoporosis in Postmenopausal Women: Effectiveness and Value 7/17/17

Neurology 86

Deutetrabenazine (Austedo®) • Indication: Treatment of chorea associated with Huntington’s disease • Approval Date: April 3, 2017 • MOA: Causes reversible reduction of dopamine release by selectively inhibiting pre-synaptic vesicular monoamine transporter type 2 (VMAT 2); decreasing the uptake of monoamines (including dopamine, serotonin, norepinephrine, and histamine) into synaptic vesicles and depleting the monoamine stores. 87 Austedo (deutetrabenazine) tablets [prescribing information]. North Wales, PA; Teva Pharmaceuticals USA, Inc, April 2017.

Deutetrabenazine (Austedo®) • Dosage: Initially, 6 mg daily; increase dose weekly by 6 mg up to maximum of 48 mg/day – Conversion from tetrabenazine: See package insert – Therapy interrupted for >7 days: Retitrate • Boxed Warning: Increased risk of depression and suicidal thoughts • Adverse Reactions: Drowsiness, diarrhea, dry mouth, and fatigue • Precautions: Neuroleptic malignant syndrome, parkinsonism, somnolence • Cost: $11, 836. 80/month (48 mg max dose) 88 Austedo (deutetrabenazine) tablets [prescribing information]. North Wales, PA; Teva Pharmaceuticals USA, Inc, April 2017.

Deutetrabenazine (Austedo®) Clinical Data Snapshot Huntington Study Group Trial (N=90) • Multicenter, randomized, double-blind, placebo control trial • 90 ambulatory patients with baseline total maximal chorea score ≥ 8 • Received either deutetrabenzine or placebo for 12 weeks • Results: • Statistically significant improvement in total maximal chorea; mean between-group difference was -2. 5 units (95% CI, – 3. 7 to – 1. 3) (P < . 001) • Significant more patients in deutetrabenzine group reported success by the PGIG scale compared to placebo (51% vs 20%; treatment difference 31. 3; 95% CI, 12. 4 -49. 8, P=0. 002) • Similar improvements in CGIC scale and SF-36 subscale • No significant difference in Berg Balance Test 89 Huntington Study Group, et al. JAMA. 2016; 316: 40 -50.

Valbenazine (Ingrezza®) • Indication: Treatment of adults with tardive dyskinesia • Approval Date: April 11, 2017 • MOA: Causes reversible reduction of dopamine release by selectively inhibiting pre-synaptic vesicular monoamine transporter type 2 (VMAT 2); alleviating the symptoms associated with dopamine hypersensitivity Davis MC, et al. NEJM. 2017; 376: 2503 -06. 90 Ingrezza (valbenazine) [prescribing information]. San Diego, CA: Neurocrine Biosciences Inc; April 2017.

Valbenazine (Ingrezza®) • Dosage: 40 mg once daily can increase to 80 mg once daily in one week – Dosage adjustment • Strong CYP 3 A 4 inducer ---- use not recommended • Strong CYP 3 A 4 inhibitor ---- 40 mg once daily maximum dose • Strong CYP 2 D 6 inhibitor ----- dose reduction may be necessary – Renal/Hepatic Impairment Dosing • Cr. Cl <30 m. L/min ---- use not recommended • Child-Pugh class B/C ---- 40 mg once daily • Adverse Reactions: somnolence and QT prolongation (not clinically significant at concentrations expected with recommended dosing • Precautions: Somnolence and QT prolongation • Cost: 40 mg - $6, 330/month 91 Ingrezza (valbenazine) [prescribing information]. San Diego, CA: Neurocrine Biosciences Inc; April 2017.

Valbenazine (Ingrezza®) Clinical Data Snapshot KINECT 3 Trial (N=234) • Multicenter, double-blind, placebo control, phase III trial • Randomized to valbenazine 40 or 80 mg OR placebo once daily • At 6 weeks, placebo group was re-randomized to either valbenazine 40 or 80 mg once daily • Therapy was stopped at week 48 and analysis occurred at week 52 • Results: • LS mean change from Baseline to week 6 in AIMS dyskinesia score was: • 80 mg: − 3. 2 vs − 0. 1 placebo group (P<. 001) • 40 mg: − 1. 9 vs − 0. 1 placebo group; (P=. 002) • Reduction in AIMS dyskinesia score continued during week 12 -48 • At week 52, score increased following discontinuation Hauser RA, et al. Am J Psychiatry. 2017; 174: 476 -84.
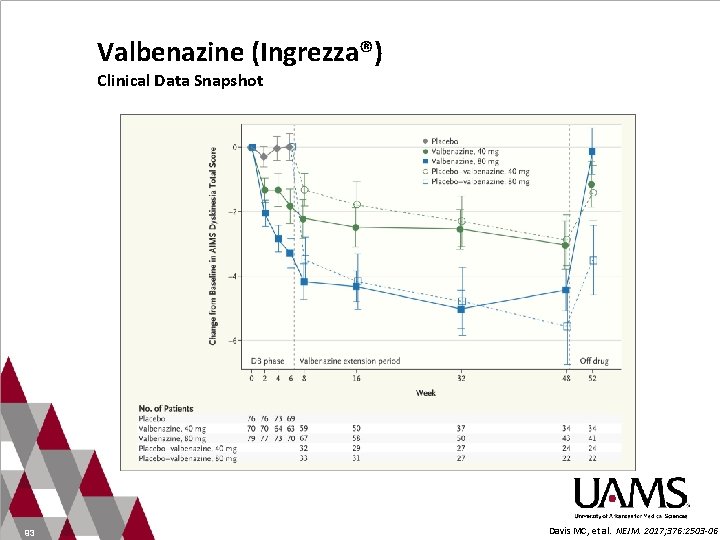
Valbenazine (Ingrezza®) Clinical Data Snapshot 93 Davis MC, et al. NEJM. 2017; 376: 2503 -06.

Deutetrabenazine (Austedo®) Clinical Data Snapshot ARM-TD Trial (N=117) • Multicenter, double-blind, placebo control trial • Randomized to deutetrabenazine or placebo once daily fro 12 weeks • Results: • LS mean change from Baseline to week 12 in AIMS dyskinesia score was: • Deutetrabenazine: − 3. 0 vs − 1. 6 placebo group (treatment difference 1. 4, P=0. 019) Fernandez HH, et al. Neurology. 2017; 88: 2003 -10.

Edaravone (Radicava®) • Indication: Treatment of amyotrophic lateral sclerosis (ALS) • Approval Date: May 5, 2017 • MOA: Free radical and peroxynitrite scavenger that prevents oxidative damage to cell membranes and may contribute to inhibiting the progression of ALS 95 Radicava (edaravone) [prescribing information]. Jersey City, NJ: MT Pharma America Inc; May 2017.

Edaravone (Radicava®) • Dosage: – Initial cycle: 60 mg IV once daily for 14 days, followed by a 14 -day drug-free period – Subsequent cycles: 60 mg IV once daily for 10 days within a 14 -day period, followed by a 14 -day drug-free period – 60 minute infusion • Adverse Reactions: Contusion, gait disturbance, and headache • Cost: 30 mg - $651. 60 – ~$18, 000/month 96 Radicava (edaravone) [prescribing information]. Jersey City, NJ: MT Pharma America Inc; May 2017.

Edaravone (Radicava®) Clinical Trial Snapshot Edaravone ALS Study Group Trial (N=137) • Double-blind, parallel-group, placebo-control trial • Japanese patients with ALS • Functionality retained most ADLs, normal respiratory function, disease 2 or less years • Randomized to edaravone 60 mg or placebo for 6 months • Results: • LS mean change from Baseline to week 24 in ALSFRS-R score was: • − 5. 01 edaravone vs − 7. 5 placebo group (treatment difference 2. 49; 95% CI 0. 99 -3. 98, P=0. 0013) 97 Writing Group; Edaravone (MCI-186) ALS 19 Study Group, et al. Lancet Neurol. 2017; 16: 505 -12.

Safinamide (Xadago®) • Indication: Adjunctive treatment to levodopa/carbidopa in patients with Parkinson disease (PD) experiencing "off" episodes – Not for monotherapy • Approval Date: March 21, 2017 • MOA: Inhibitor of MAO-B, blocking the catabolism of dopamine • Safinamide more selective for MAO-B vs MAO-A (1000 -fold) than selegiline (127 fold) and rasagiline (103 -fold) – Reduces diet restrictions – MAO-B inhibition is reversible • Better clinical manageability and limiting possible drug–drug interactions 98 Xadago (safinamide) [prescribing information]. Louisville, KY: US World. Meds; June 2017.

Safinamide (Xadago®) • Dosage: 50 mg once daily, can increase up to 100 mg daily in 2 weeks • Adverse Reactions: Dyskinesia, fall, nausea, and insomnia • Contraindications: Other MAO inhibitors, opioids, SNRIs, tricyclic antidepressants, cyclobenzaprine, methylphenidate, amphetamine, St. John's wort • Cost: 50 and 100 mg - $803. 88 (30) 99 Xadago (safinamide) [prescribing information]. Louisville, KY: US World. Meds; June 2017.

Safinamide (Xadago®) Clinical Trial Snapshot Borgohain et al (N=669) • Multicenter, double-blind, placebo-control, phase III trial • Randomized to safinamide 100 or 50 mg daily or placebo for 24 weeks • Results: • Significant difference in mean total “on” time with both safinamide groups compared to placebo • Significant improvements in motor scores with both safinamide groups compared to placebo • No significant between-group differences with treatment-emergent adverse events 100 Borgohain R, et al. Movement Disorders. 2014; 29: 229 -37.

Safinamide (Xadago®) Clinical Trial Snapshot SETTLE Trial (N=549) • Multicenter, double-blind, placebo-control, phase III trial • Randomized to safinamide daily or placebo for 24 weeks • Results: • Similar results as Study 016 • 0. 96 hour improvement from placebo • Did not reach significance for improvement in motor scores 101 Schapira HV, et al. JAMA Neurology. 2017; 74: 216 -24.

Oncology 102

Rucaparib (Rubraca®) • Indication: Treatment of advanced ovarian cancer in patients with deleterious BRCA mutation (germline and/or somatic) who have been treated with two or more chemotherapies • Approval Date: December 19, 2016 • MOA: Inhibits poly (ADP-ribose) polymerase (PARP) enzymes, which play a role in DNA repair, resulting in DNA damage, apoptosis, and cell death 103 Rubraca (rucaparib) [prescribing information]. Boulder, CO: Clovis Oncology; February 2017.

Rucaparib (Rubraca®) • Doseage: 600 mg twice daily – Available in multiple tablet strengths (200, 250, and 300 mg) – Dose reductions for toxicities • Adverse Reactions: – Nausea, fatigue, anemia, vomiting, decreased appetite, thrombocytopenia – Increase SCr, ALT, AST, cholesterol and pancytopenia • Cost: – $8244/month or ~$100, 000 annually 104 Rubraca (rucaparib) [prescribing information]. Boulder, CO: Clovis Oncology; February 2017.

Rucaparib (Rubraca®) Clinical Trial Snapshot Study 1 & 2 (N=106) • Two multicenter, single arm, open label trials • 100% had an ECOG performance status of 0 or 1 and all had received at least 2 prior platinum-based chemotherapies • Rucaparib 600 mg twice daily • Results: • Investigator-assessed • Objective response rate: 54% (complete response 9%; partial response 45% • Median duration of response: 9. 2 (6. 6, 11. 6) • Independent radiology review • Objective response rate: 42% • Median duration of response: 6. 7 (5. 5, 11. 1) 105 Rubraca (rucaparib) [prescribing information]. Boulder, CO: Clovis Oncology; February 2017.

Ribociclib (Kisqali®) • Indication: Treatment of advanced or metastatic hormone receptor-positive, breast cancer – Used in combination with an aromatase inhibitor • Approval Date: March 13, 2017 • MOA: Cyclin-depended kinase inhibitor specifically for 4 and 6 to block retinoblastoma protein phosphorylation and prevents progression through the cell cycle at G 1 phase 106 Kasqali (ribociclib) [prescribing information]. East Hanover, NJ: Novartis Pharmaceuticals; March 2017.

Ribociclib (Kisqali®) • Dosage: 600 mg by mouth – Once daily for 21 days, 7 days off – Can dose adjust down to 200 mg for toxicities • Adverse Reactions: Peripheral edema, nausea, vomiting, neutropenia, leukopenia, decrease platelet count, alopecia • Precautions: QT prolongation, neutropenia and hepatobiliary toxicity • Cost: 600 mg dose - $13, 140. 00 per month – ~$250/tablet 107 Kasqali (ribociclib) [prescribing information]. East Hanover, NJ: Novartis Pharmaceuticals; March 2017.

Ribociclib (Kisqali®) Clinical Trial Snapshot MONALEESA-2 (N=668) • Multicenter, double-blind, placebo-control phase III trial • Randomized to ribociclib 600 mg plus letrozole or placebo plus letrozole • Median f/u: 15. 3 months • Results: • PFS significantly longer with ribociclib group • Hazard ratio, 0. 56; 95% CI, 0. 43 to 0. 72; P=3. 29× 10− 6 for superiority • PFS at 18 months: (63% ribociclib and 42. 2% placebo) • OS not nature • Safety – more neutropenia, anemia, and increases in ALT and AST w/ribociclib 108 Hortobagyi GN, et al. NEJM; 375: 1738 -1748.

Neratinib (Nerlynx®) • Indication: Extended adjuvant treatment of early stage HER 2 positive breast cancer • Approval Date: July 17, 2017 • MOA: Irreversible tyrosine kinase inhibitor of HER 1, HER 2, and HER 4, demonstrates antitumor activity in EGFR and/or HER 2 expressing cancer 109 Nerlynx (neratinib) [prescribing information]. Los Angeles, CA: Puma Biotechnology, Inc. ; July 2017.

Neratinib (Nerlynx®) • Dosage: 240 mg (6 40 mg tablets) once daily with food for 1 year • Adverse Reactions: Diarrhea, nausea, abdominal pain/distention, fatigue, vomiting, rash, stomatitis, decreased appetite, muscle spasms, dyspepsia, AST or ALT increase, nail disorder, dry skin, weight decreased and urinary tract infection • Precautions: Diarrhea and hepatotoxicity • Cost: $12, 600/month 110 Nerlynx (neratinib) [prescribing information]. Los Angeles, CA: Puma Biotechnology, Inc. ; July 2017.

Neratinib (Nerlynx®) Clinical Trial Snapshot Exte. NET Trial (N=2840) • Multicenter, double-blind, placebo control, phase III trial • Stage 1 -2 HER 2 positive breast cancer completed trastuzumab therapy at least 2 yrs prior • Randomized to neratinib 240 mg or placebo • Median f/u 24 months • Results: Invasive disease-free survival event, n 2 -yr invasive disease-free survival rate, % (95% CI) Neratinib (N=1420) Placebo (N=1420) HR (95% CI) 70 109 0. 67 (0. 50 -0. 91) p=0. 0091 93. 9 (92. 4 -95. 2) 91. 6 (90 -93) 562 (40) 23 (2) Adverse events Diarrhea (Grade 3 -4), n (%) 111 Chan A, et al. Lancet Oncol. 2016; 17: 367 -77.

Niraparib (Zejula®) • Indication: Maintenance treatment of recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer in patients who are in a complete or partial response to platinum-based chemotherapy – In patients with or without BRCA 1/2 mutations • Approval Date: March 27, 2017 • MOA: Poly (ADP-ribose) polymerase (PARP) enzyme inhibitor, inhibiting PARP enzymatic activity results in DNA damage, apoptosis and cell death http: //rubraca. com/about-rubraca/how-rubraca-works 112 Zejula (niraparib) [prescribing information]. Waltham, MA: Tesaro Inc; March 2017.

Niraparib (Zejula®) • Dosage: 300 mg by mouth daily – Begin no later than 8 weeks following platinum-containing regimen • Adverse Reactions: Thrombocytopenia, anemia, neutropenia, leukopenia, palpitations, nausea, constipation, vomiting, mucositis, diarrhea, dyspepsia, fatigue, and AST/ALT elevation • Precautions: Myelodysplastic Syndrome/Acute Myeloid Leukemia, Bone marrow suppression, and CV effects • Cost: $17, 700/ month 113 Zejula (niraparib) [prescribing information]. Waltham, MA: Tesaro Inc; March 2017.
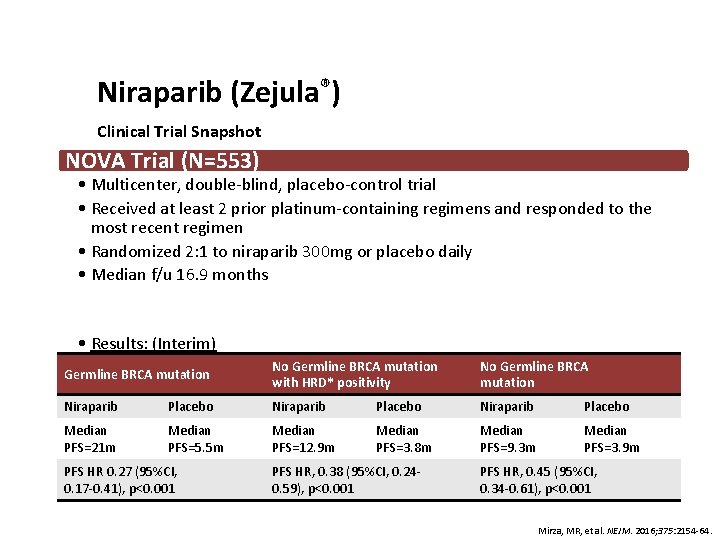
Niraparib (Zejula®) Clinical Trial Snapshot NOVA Trial (N=553) • Multicenter, double-blind, placebo-control trial • Received at least 2 prior platinum-containing regimens and responded to the most recent regimen • Randomized 2: 1 to niraparib 300 mg or placebo daily • Median f/u 16. 9 months • Results: (Interim) Germline BRCA mutation No Germline BRCA mutation with HRD* positivity No Germline BRCA mutation Niraparib Placebo Median PFS=21 m Median PFS=5. 5 m Median PFS=12. 9 m Median PFS=3. 8 m Median PFS=9. 3 m Median PFS=3. 9 m PFS HR 0. 27 (95%CI, 0. 17 -0. 41), p<0. 001 PFS HR, 0. 38 (95%CI, 0. 240. 59), p<0. 001 PFS HR, 0. 45 (95%CI, 0. 34 -0. 61), p<0. 001 Mirza, MR, et al. NEJM. 2016; 375: 2154 -64.
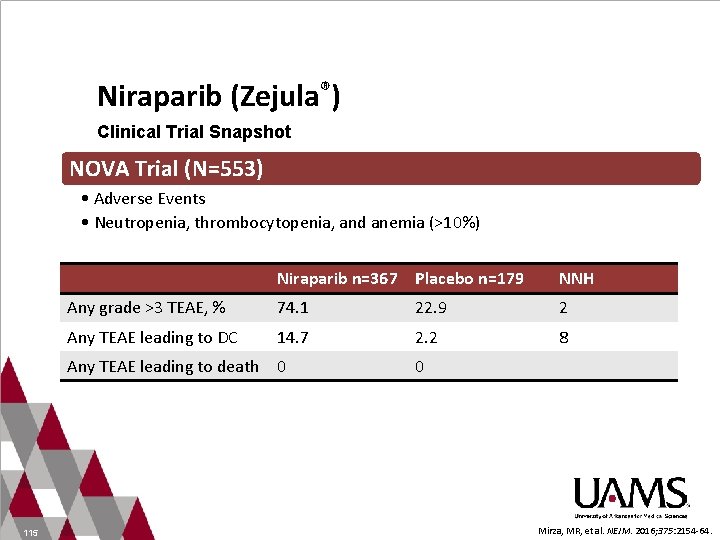
Niraparib (Zejula®) Clinical Trial Snapshot NOVA Trial (N=553) • Adverse Events • Neutropenia, thrombocytopenia, and anemia (>10%) Niraparib n=367 Placebo n=179 NNH Any grade >3 TEAE, % 74. 1 22. 9 2 Any TEAE leading to DC 14. 7 2. 2 8 Any TEAE leading to death 0 115 0 Mirza, MR, et al. NEJM. 2016; 375: 2154 -64.

Brigatinib (Alunbrig®) • Indication: Treatment of ALK-positive metastatic non-small cell lung cancer (NSCLC) in patients who have progressed on or are intolerant to crizotinib • Approval Date: April 28, 2017 • MOA: ALK tyrosine kinase inhibitor, was designed for potent activity against a broad range of ALK resistance mutations 116 Alunbrig (brigatinib) [prescribing information]. Cambridge, MA: Ariad Pharmaceuticals Inc; April 2017.

Brigatinib (Alunbrig®) • Dosage: 90 mg by mouth daily after 7 days increase to 180 mg daily – Minimum dose: 60 mg daily • Adverse Reactions: Nausea, diarrhea, fatigue, cough, and headache • Precautions: Interstitial lung disease, HTN, bradycardia, visual disturbance, CPK elevations, pancreatic enzyme elevations, and hyperglycemia • Cost: $95/tablet (30 mg tablet) – ~$17, 000/month (180 mg dosing) 117 Alunbrig (brigatinib) [prescribing information]. Cambridge, MA: Ariad Pharmaceuticals Inc; April 2017.

Brigatinib (Alunbrig®) Clinical Trial Snapshot ALTA (N=222) • Two-arm, open-label, multicenter trial • Locally advanced/metastatic ALK-positive NSCLC with progression on crizotinib • Randomized to brigatinib 90 or 180 mg once daily • Median f/u: 8 months • Results: • Objective response rate: • 45% brigatinib 90 mg 54% brigatinib 180 mg • Mean progression free survival: • 9. 2 months brigatinib 90 mg 12. 9 months brigatinib 180 mg • TEAEs: GI symptoms, headache, cough 118 Kim DW, et al. J Clin Oncol. 2017; 35: 2490 -98.

Oncology Besponsa® (inotuzumab ozogamicin) • Treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia • Humanized CD 22 -directed monoclonal antibody-drug • IV infusion • Boxed warning: Hepatotoxicity and increased risk of post-transplant non-relapse mortality • Clinical Trial Results: • 218 evaluated patients • 35. 8% who received Besponsa experienced complete remission (CR) for a median 8. 0 months • 17. 4% who received alternative chemotherapy experienced CR for a median 4. 9 months 119 Besponsa (inotuzumab ozogamicin) [prescribing information]. Philadelphia, PA: Wyeth Pharmaceuticals Inc; August 2017.
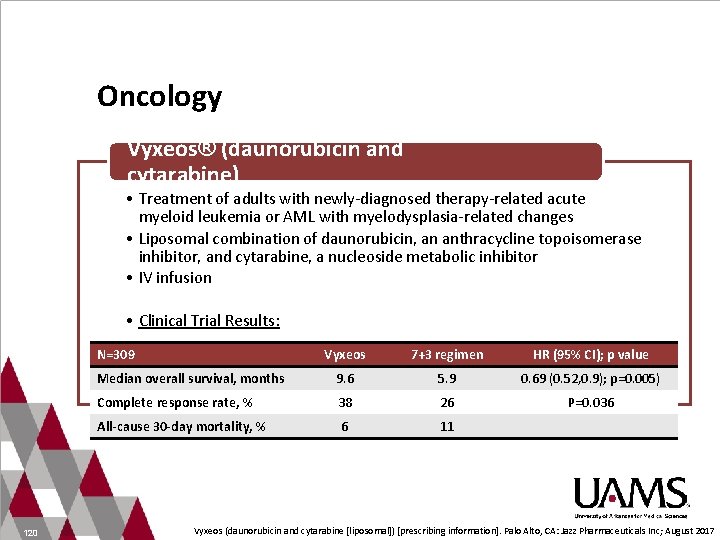
Oncology Vyxeos® (daunorubicin and cytarabine) • Treatment of adults with newly-diagnosed therapy-related acute myeloid leukemia or AML with myelodysplasia-related changes • Liposomal combination of daunorubicin, an anthracycline topoisomerase inhibitor, and cytarabine, a nucleoside metabolic inhibitor • IV infusion • Clinical Trial Results: N=309 120 Vyxeos 7+3 regimen HR (95% CI); p value Median overall survival, months 9. 6 5. 9 0. 69 (0. 52, 0. 9); p=0. 005) Complete response rate, % 38 26 P=0. 036 All-cause 30 -day mortality, % 6 11 Vyxeos (daunorubicin and cytarabine [liposomal]) [prescribing information]. Palo Alto, CA: Jazz Pharmaceuticals Inc; August 2017

Enasidenib (IDHIFA®) • Indication: Treatment of relapsed or refractory acute myeloid leukemia (AML) in patients with an isocitrate dehydrogenase-2 (IDH 2) mutation • Approval Date: August 1, 2017 • MOA: Small molecule inhibitor of the isocitrate dehydrogenase 2 (IDH 2) enzyme that works by blocking several enzymes that promote cell growth 121 Idhifa (enasidenib) [prescribing information]. Summit, NJ: Celgene Corporation; August 2017.

Enasidenib (IDHIFA®) • Dosage: 100 mg daily by mouth until disease progression • Boxed Warning: Differentiation syndrome • Adverse Reactions: Nausea, vomiting, diarrhea, increased levels of bilirubin, and decreased appetite • Precautions: Electrolyte imbalance, hematologic effects, hepatotoxicity, GI toxicity, and tumor lysis syndrome • Cost: $29, 846. 40/month 122 Idhifa (enasidenib) [prescribing information]. Summit, NJ: Celgene Corporation; August 2017.

Enasidenib (IDHIFA®) Study 1 (N=199) • Multicenter, open-label, single arm, two-cohort trial • Patients with relapsed or refractory AML and IDH 2 mutation received enasidenib 100 mg daily • Median f/u: 6. 6 months • Results: IDHIFA N=199 Endpoint 37 (19); (13 -25) Complete remission, n(%); (95% CI) Complete remission w/partial hematological recovery, n(%); (95% CI) 123 9 (4); (2 -8) Median duration of response, months (95% CI) 8. 2 (4. 7 -19. 4) 9. 6 (0. 7 -NA) Idhifa (enasidenib) [prescribing information]. Summit, NJ: Celgene Corporation; August 2017.

Midostaurin (Rydapt®) • Indication: – Newly diagnosed acute myeloid leukemia (AML) that is FLT 3 mutationpositive • Not monotherapy – Aggressive systemic mastocytosis (ASM), systemic mastocytosis with associated hematological neoplasm (SM-AHN), or mast cell leukemia (MCL) • Approval Date: April 27, 2017 • MOA: Small molecule that inhibits multiple receptor tyrosine kinases 124 Rydapt (midostaurin) [prescribing information]. East Hanover, NJ: Novartis Pharmaceuticals Corporation; April 2017.

Midostaurin (Rydapt®) • Dosage: – AML: 50 mg orally twice daily with food on days 8 to 21 of each induction cycle (in combination with daunorubicin and cytarabine) and on days 8 to 21 of each consolidation – ASM, SM-AHN, and MCL: 100 mg orally twice daily with food • Adverse Reactions: Febrile neutropenia, nausea, mucositis, vomiting, headache, petechiae, musculoskeletal pain, epistaxis, device-related infection, hyperglycemia, and upper respiratory tract infection • Precautions: Pulmonary toxicity • Cost: $4497/cycle for AML OR ~$18, 000/month 125 Rydapt (midostaurin) [prescribing information]. East Hanover, NJ: Novartis Pharmaceuticals Corporation; April 2017.

Midostaurin (Rydapt®) Clinical Trial Snapshot RATIFY Trial (N=717) • • Multicenter, double-blind, placebo control, phase III trial Patients with newly diagnosed AML not previously treated with antineoplastic Randomized to standard chemotherapy plus midostaurin or placebo Median f/u: 59 months • Results: 126 Midostaurin N=360 Placebo N=357 HR (95% CI) Media overall survival, months 74. 7 25. 6 0. 78 (0. 63 -0. 96), p=0. 009 Event-free survival, months 8. 2 3. 0 p=0. 002 Stone RM, et al. NEJM. 2017; 377: 454 -64.

Oncology Bavencio® (avelumab) • Adults and pediatric patients ≥ 12 years with metastatic Merkel cell carcinoma (MCC) • Patients with locally advanced or metastatic urothelial carcinoma (UC) who have disease progression during or following platinum-containing chemotherapy • Programmed death ligand-1 (PD-L 1) blocking antibody • IV infusion • Clinical Trial Results: • JAVELIN Merkel 200 Trial – MCC • Overall response rate: 33% • Duration of response: 2. 8 – 23. 3+ months • JAVELIN Solid Tumor Trial – UC • Overall response rate: 16. 1% • Duration of response: 1. 4 – 17. 4+ months 127 Bavencio (avelumab) [prescribing information]. Rockland, MA: EMD Serono Inc; May 2017.

Oncology Imfinzi® (durvalumab) • Treatment of patients with locally advanced or metastatic urothelial carcinoma who: • Have disease progression during or following [within 12 months] platinumcontaining chemotherapy • Programmed death-ligand 1 (PD-L 1) blocking antibody • IV infusion N=182 Imfiniz 10 mg/kg q 2 wks • Boxed warning: Hepatotoxicity and increased risk of post-transplant non. Objective response rate, n (%) 31 (17%) relapse mortality • Clinical Trial Results: Response of 6 months or longer, n 31 Response of 12 months or longer, 128 5 Imfinzi (durvalumab) [prescribing information]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; April 2017.
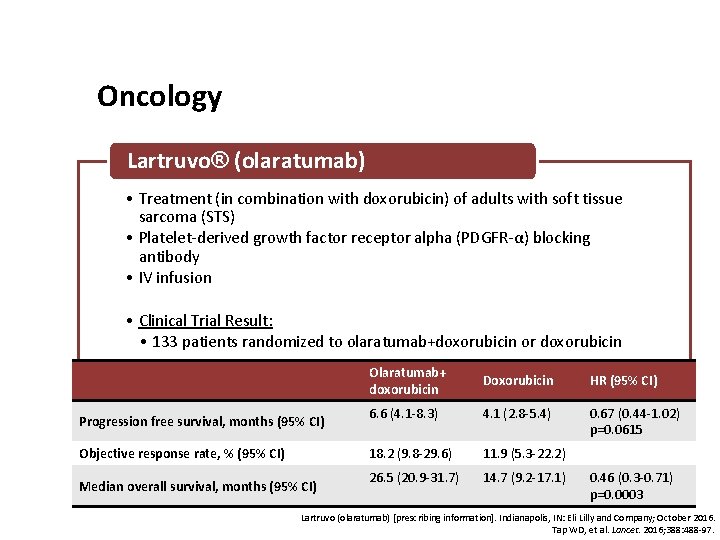
Oncology Lartruvo® (olaratumab) • Treatment (in combination with doxorubicin) of adults with soft tissue sarcoma (STS) • Platelet-derived growth factor receptor alpha (PDGFR-α) blocking antibody • IV infusion • Clinical Trial Result: • 133 patients randomized to olaratumab+doxorubicin or doxorubicin Progression free survival, months (95% CI) Objective response rate, % (95% CI) Median overall survival, months (95% CI) Olaratumab+ doxorubicin Doxorubicin HR (95% CI) 6. 6 (4. 1 -8. 3) 4. 1 (2. 8 -5. 4) 0. 67 (0. 44 -1. 02) p=0. 0615 18. 2 (9. 8 -29. 6) 11. 9 (5. 3 -22. 2) 26. 5 (20. 9 -31. 7) 14. 7 (9. 2 -17. 1) 0. 46 (0. 3 -0. 71) p=0. 0003 Lartruvo (olaratumab) [prescribing information]. Indianapolis, IN: Eli Lilly and Company; October 2016. Tap WD, et al. Lancet. 2016; 388: 488 -97.

New Drug Update 2016 -2017 Rachael Mc. Caleb, Pharm. D, BCPS Assistant Professor, Department of Pharmacy Practice University of Arkansas for Medical Sciences College of Pharmacy

Additional Information Will not be discussed during this presentation 131

Genetic Disease 132

Cerliponase alfa (Brineura®) • Indication: To slow the loss of ambulation in symptomatic pediatric patients 3 years of age and older with late infantile neuronal ceroid lipofuscinosis type 2 (CLN 2) • Approval Date: April 27, 2017 • MOA: Hydrolytic lysosomal N-terminal tripeptidyl peptidase – Deficiency in TPP 1 activity results in the accumulation of lysosomal storage, leading to progressive decline in motor function 133 Brineura (cerliponase alfa) [prescribing information]. Novato, CA: Bio. Marin Pharmaceutical Inc; April 2017.

Cerliponase alfa (Brineura®) • Dosage: Intraventricular infusion – 300 mg once every other week followed by administration of 2 m. L intraventricular electrolytes • Adverse Reactions: pyrexia, ECG abnormalities, decreased/increased CSF protein, vomiting, seizures, hypersensitivity, hematoma, headache, irritability, pleocytosis, device-related infection, bradycardia, feeling jittery, and hypotension • Cost: $64, 800/dose 134 Brineura (cerliponase alfa) [prescribing information]. Novato, CA: Bio. Marin Pharmaceutical Inc; April 2017.

Cerliponase alfa (Brineura®) Clinical Trial Snapshot Package Insert Trial (N=24) • Single arm trial • Cerliponase alfa 300 mg QOW for 48 weeks (extension 96 weeks) • Compare to historical cohort • Results: • 22 patients treated with Brineura and evaluated for efficacy at week 96, 21 (95%) did not decline • 21/42 (50%) patients in the historical cohort experienced an unreversed (sustained) 2 -category decline or unreversed score of 0 in the Motor domain of the CLN 2 Clinical Rating Scale over 96 weeks 135 Brineura (cerliponase alfa) [prescribing information]. Novato, CA: Bio. Marin Pharmaceutical Inc; April 2017.

C 1 esterase inhibitor (Haegarda®) • Indication: Prophylaxis against hereditary angioedema (HAE) in adults and adolescents • Approval Date: July 2017 • MOA: C 1 inhibitor therapy in patients with a C 1 inhibitor deficiency (as in HAE), is thought to suppress the activation of plasma kallikrein and factor XIIa which prevents bradykinin production 136 Haegarda (C 1 Esterase Inhibitor [Human]) [prescribing information]. Kankakee, IL: CSL Behring LLC; June 2017.

C 1 esterase inhibitor (Haegarda®) • Dosage: 60 IU/kg q 3 -4 days – Lyophilized powder supplied in single-use vials • Adverse Reactions: Injection site reaction, hypersensitivity, nasopharyngitis and dizziness • Cost: – 2000 unit vial ($2256. 00) – 3000 unit vial ($3384. 00) 137 Haegarda (C 1 Esterase Inhibitor [Human]) [prescribing information]. Kankakee, IL: CSL Behring LLC; June 2017.

C 1 esterase inhibitor (Haegarda®) Clinical Trial Snapshot COMPACT Trial (N=90) • Multicenter, double-blind, placebo-control, crossover trial • Randomized to either Haegarda 60 IU/kg or 40 IU/kg for 16 weeks and placebo for the other 16 weeks • Results: Haegarda 60 IU/kg (N=45) Haegarda 40 IU/kg (N=45) Placebo Haegarda Placebo Number of HAE Attacks (Number/month) Mean (SD) 0. 5 (0. 8) 4 (2. 3) 1. 2 (2. 3) 3. 6 (2. 1) Min, Max 0. 0, 3. 1 0. 6, 11. 3 0. 0, 12. 5 0. 0, 8. 9 Treatment difference LS Mean (95% CI) P-value 138 -3. 5 (-4. 2, -2. 8) -2. 4 (-3. 4, -1. 5) <0. 001 Haegarda (C 1 Esterase Inhibitor [Human]) [prescribing information]. Kankakee, IL: CSL Behring LLC; June 2017.

Musculoskeletal 139

Nusinersen (Spinraza®) • Indication: Treatment of spinal muscular atrophy (SMA) in pediatric and adult patients • Approval Date: December 23, 2016 • MOA: Nusinersen is an antisense oligonucleotide (ASO) designed to treat SMA caused by mutations in chromosome 5 q that lead to SMN protein deficiency 140 Spinraza (nusinersen) [prescribing information]. Cambridge, MA: Biogen; June 2017.

Nusinersen (Spinraza®) • Dosage: 12 mg per dose – Loading dose: 4 doses (3 doses every 14 days then 1 dose 30 days after the third dose) – Maintenance dose: every 4 months • Adverse Reactions: joint contracture and pyrexia • Cost: Based on 1 year, LD + MD 141 $30000/m. L, provided in a singleuse 5 m. L vial $150, 000/vial = 1 dose Year 1 Years 2 and each year after 6 doses, 1 st year. 3 doses every year thereafter $900, 000 $450, 000 Spinraza (nusinersen) [prescribing information]. Cambridge, MA: Biogen; June 2017.

Nusinersen (Spinraza®) Clinical Trial Snapshot Study 1 (N=20) • Phase II, open label, dose-escalation study • Improvement in developmental motor milestones (HINE-2 and CHOPINTEND) Ongoing trials • Phase 3 in infantile-onset SMA • Study CS 4: Patients with later-onset SMA • Interim analysis (N=126) • Study stopped based on highly statistically significant treatment effect • Findings not yet reviewed by FDA 142 Spinraza (nusinersen) [prescribing information]. Cambridge, MA: Biogen; June 2017.

Eteplirsen (Exondys 51®) • Indication: Treatment of Duchenne muscular dystrophy (DMD) in patients who have a confirmed mutation of the DMD gene that is amenable to exon 51 skipping. • Approval Date: September 19, 2016 • MOA: Binds to exon 51 of dystrophin pre-messenger RNA (m. RNA), resulting in exclusion of this exon during m. RNA processing. 143 Exondys 51 (eteplirsen) [prescribing information]. Cambridge, MA; Sarepta Therapeutics, Inc: September 2016.

Eteplirsen (Exondys 51®) • Dosage: 30 mg/kg once weekly – Administered by IV infusion over 35 to 60 minutes – Supplied as: • 100 mg/2 m. L and 500 mg/2 m. L single dose vial • Adverse Reactions: Balance disorder and vomiting (>35% incidence compared to placebo) • Cost: 100 mg/2 m. L - $1920. 00 144 Exondys 51 (eteplirsen) [prescribing information]. Cambridge, MA; Sarepta Therapeutics, Inc: September 2016.

Eteplirsen (Exondys 51®) Clinical Data Snapshot Mendell, et al. (2013) (N=12) • Randomized, double-blind, placebo-controlled • Boys aged 7 -13 years with DMD and ability to walk 200 -400 m on 6 MWT • 3 cohorts (placebo, 30 mg/kg/wk, 50 mg/kg/wk) for 24 weeks • At week 24, placebo cohort randomized to 30 mg/kg/wk or 50 mg/kg/wk (open-label extension) • Results: • Statistically significant increase in dystrophin positive fibers by week 48 • Statistical differences compared to placebo for 6 MWT as a 67. 3 m difference from baseline in the 30 mg/kg group and 87. 4 m in the 50 mg/kg group 145 Mendell JR, et al. Ann Neurol. 2013; 74: 637 -47.

Eteplirsen (Exondys 51®) Clinical Data Snapshot Mendell et al. (2016) • Evaluated longitudinal effect of eteplirsen versus historical control • Patients: ambulatory DMD patients ≥ 7 years old • Results: • At 36 months eteplirsen patients showed a statistically and clinically meaningful difference in 6 MWT decline of 151 m compared to placebo • Loss of ambulation over 3 years • Eteplirsen group: 2/12 • Placebo group: 6/13 146 Mendell JR, et al. Ann of Neurol. 2016; 79: 257 -271.

Deflazacort (Emflaza®) • Indication: Treatment of Duchenne muscular dystrophy (DMD) in patients ≥ 5 years • Approval Date: February 9, 2017 • MOA: Corticosteroid prodrug; its active metabolite acts on the glucocorticoid receptor to exert anti-inflammatory and immunosuppressive effects http: //reference. medscape. com/drug/emflaza-deflazacort-1000120 147 Emflaza (deflazacort) [prescribing information]. Northbrook, IL: Marathon Pharmaceuticals; February 2017.

Deflazacort (Emflaza®) • Dosage: 0. 9 mg/kg/day by mouth • Adverse Reactions: Cushingoid appearance, weight increased, increased appetite, upper respiratory tract infection, cough, pollakiuria, hirsutism, central obesity, and nasopharyngitis • Precautions: Alterations in endocrine function. Immunosuppression, GI perforation, behavior/mood disturbances, effects on bone, and ophthalmic effects • Cost: – 6, 18, 30, and 36 mg tablet - $294/tab – Oral suspension 22. 75 mg/m. L - $298/m. L 148 Emflaza (deflazacort) [prescribing information]. Northbrook, IL: Marathon Pharmaceuticals; February 2017.
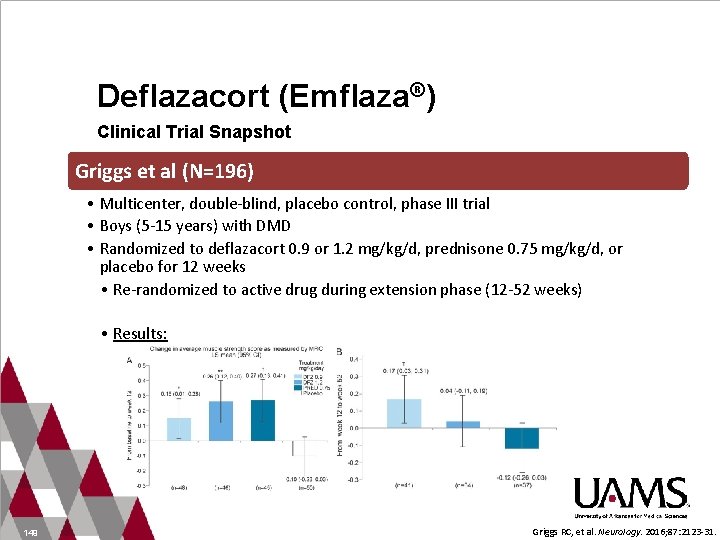
Deflazacort (Emflaza®) Clinical Trial Snapshot Griggs et al (N=196) • Multicenter, double-blind, placebo control, phase III trial • Boys (5 -15 years) with DMD • Randomized to deflazacort 0. 9 or 1. 2 mg/kg/d, prednisone 0. 75 mg/kg/d, or placebo for 12 weeks • Re-randomized to active drug during extension phase (12 -52 weeks) • Results: 149 Griggs RC, et al. Neurology. 2016; 87: 2123 -31.
- Slides: 149