NEW DIABETES TREATMENTS Richard Sachson MD 1552 BC

NEW DIABETES TREATMENTS Richard Sachson MD
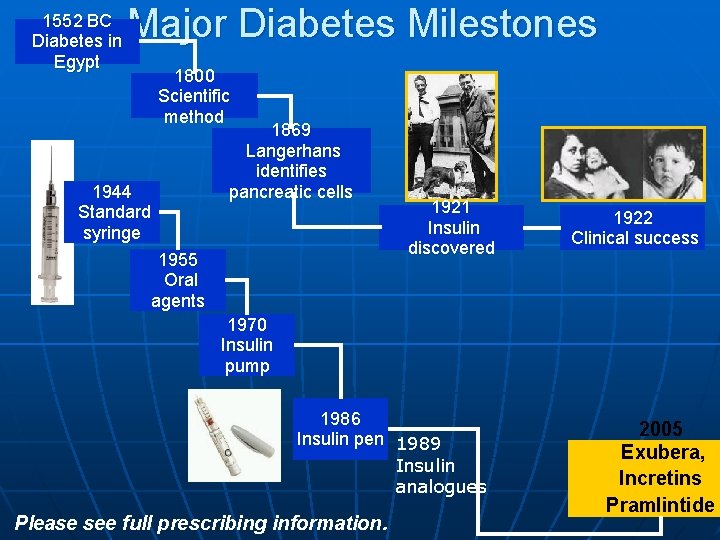
1552 BC Diabetes in Egypt Major Diabetes Milestones 1800 Scientific method 1944 Standard syringe 1869 Langerhans identifies pancreatic cells 1955 Oral agents 1921 Insulin discovered 1922 Clinical success 1970 Insulin pump 1986 Insulin pen 1989 Insulin analogues Please see full prescribing information. 2005 Exubera, Incretins Pramlintide
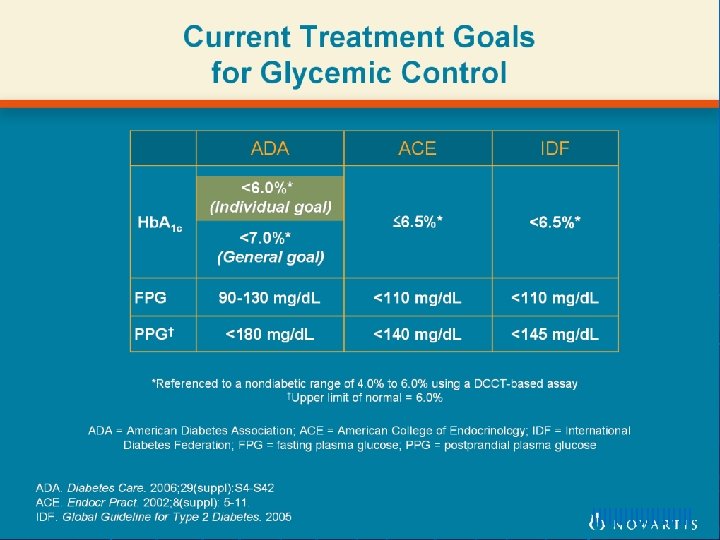
Current Treatment Goals for Glycemic Control llllllllll

New Diabetes Therapeutic Agents n Incretins • GLP 1 analogues: Exenatide- Byetta • DPP 4 Inhibitors: Sitagliptin- Januvia n n Pramlintide-Symlin Inhaled insulin-Exubera

INCRETINS


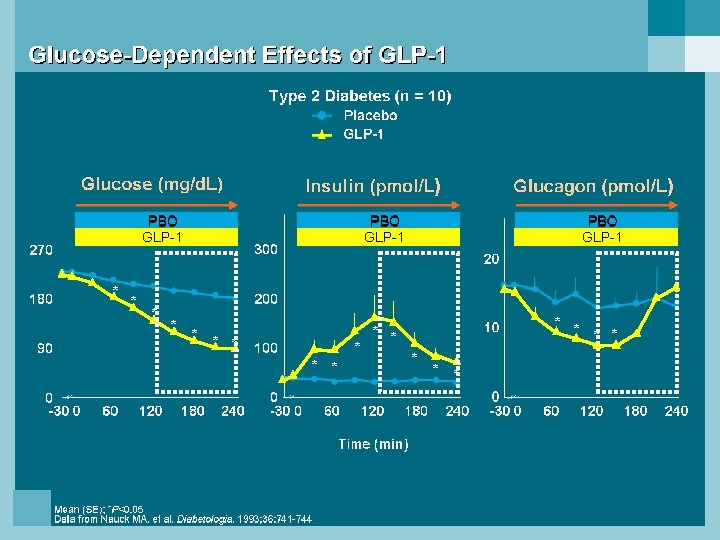
GLP-1 and GIP Are Incretin Hormones GLP-1 § § § Is released from L cells in ileum and colon 1, 2 Stimulates insulin response from beta cells in a glucose-dependent manner 1 Inhibits gastric emptying 1, 2 Reduces food intake and body weight 2 Inhibits glucagon secretion from alpha cells in a glucose-dependent manner 1 Deficient in type 2 diabetes GIP § § § Is released from K cells in duodenum 1, 2 Stimulates insulin response from beta cells in a glucose-dependent manner 1 Has minimal effects on gastric emptying 2 Has no significant effects on satiety or body weight 2 Does not appear to inhibit glucagon secretion from alpha 1, 2 cells 1, 2 Normal levels but decreased responsiveness in type 2 diabetes 1. Meier JJ et al. Best Pract Res Clin Endocrinol Metab. 2004; 18: 587– 606. 2. Drucker DJ. Diabetes Care. 2003; 26: 2929– 2940.

DPP-4 Degrades GLP-1
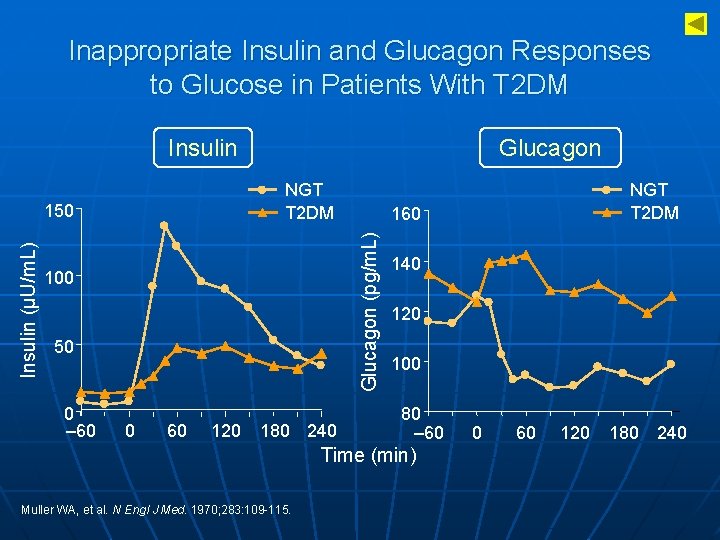
Inappropriate Insulin and Glucagon Responses to Glucose in Patients With T 2 DM Insulin NGT T 2 DM 100 50 0 – 60 0 60 120 180 240 120 100 80 – 60 Time (min) Muller WA, et al. N Engl J Med. 1970; 283: 109 -115. NGT T 2 DM 160 Glucagon (pg/m. L) 150 Insulin (μU/m. L) Glucagon 0 60 120 180 240

EXENATIDE

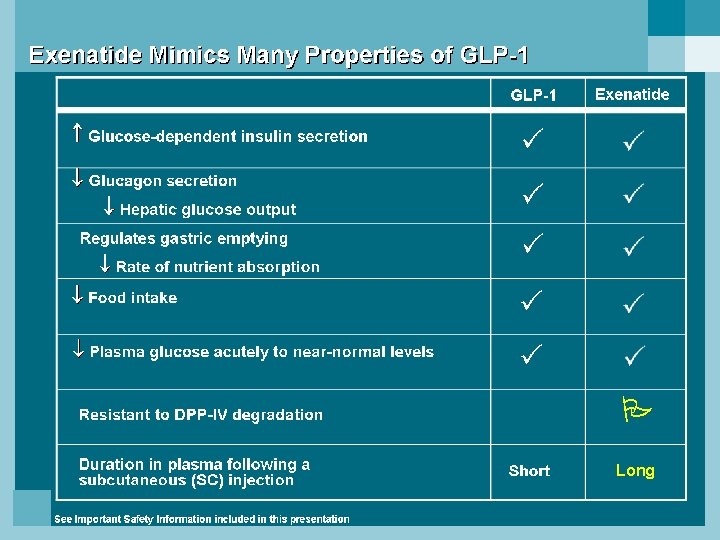
Long

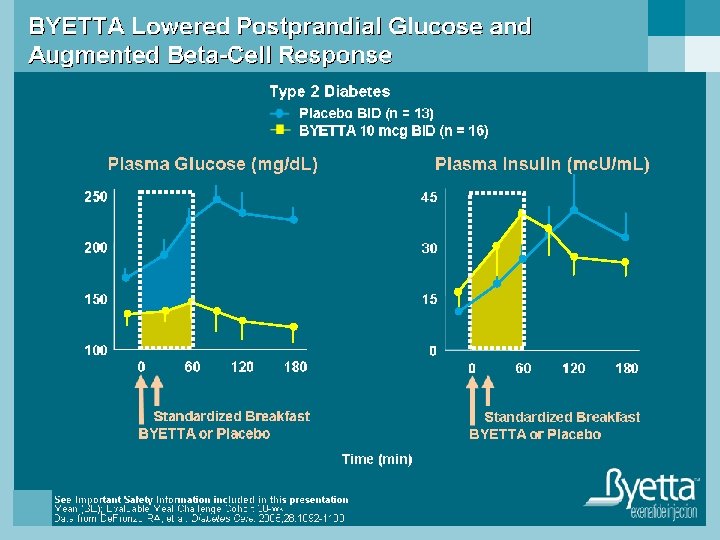
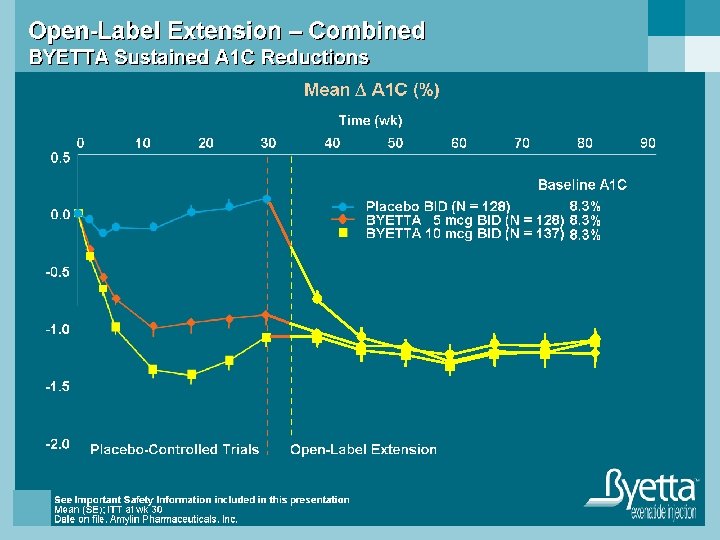




Byetta Prescribing Considerations n Adjunctive therapy to improve glycemic control in type 2 diabetic patients who are taking • Metformin • SFU • TZD

SIDAGLIPTIN- JANUVIA
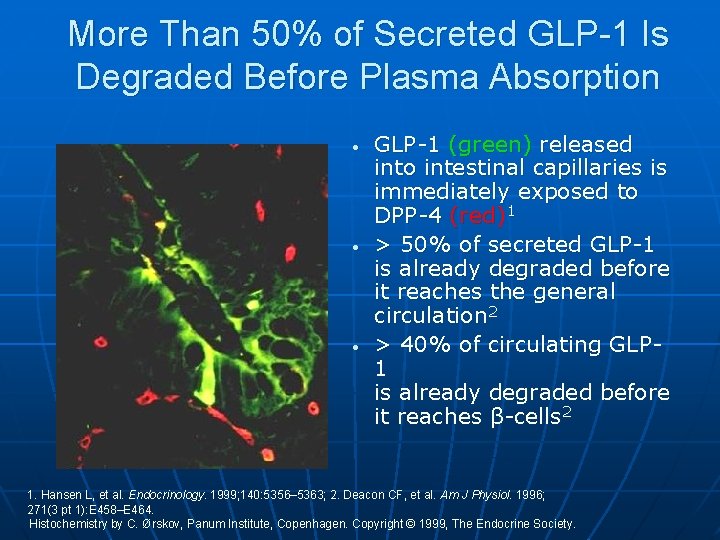
More Than 50% of Secreted GLP-1 Is Degraded Before Plasma Absorption • • • GLP-1 (green) released into intestinal capillaries is immediately exposed to DPP-4 (red)1 > 50% of secreted GLP-1 is already degraded before it reaches the general circulation 2 > 40% of circulating GLP 1 is already degraded before it reaches β-cells 2 1. Hansen L, et al. Endocrinology. 1999; 140: 5356– 5363; 2. Deacon CF, et al. Am J Physiol. 1996; 271(3 pt 1): E 458–E 464. Histochemistry by C. Ørskov, Panum Institute, Copenhagen. Copyright © 1999, The Endocrine Society.
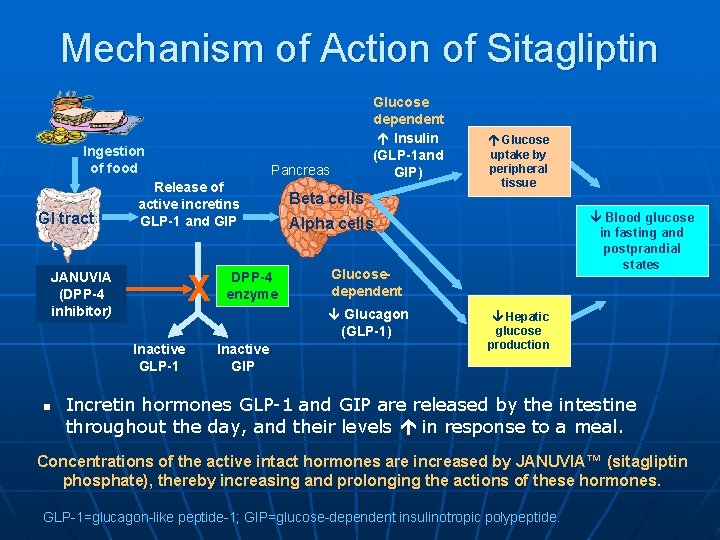
Mechanism of Action of Sitagliptin Ingestion of food GI tract X Inactive GLP-1 n Pancreas Release of active incretins GLP-1 and GIP JANUVIA (DPP-4 inhibitor) Glucose dependent Insulin (GLP-1 and GIP) DPP-4 enzyme Beta cells Blood glucose in fasting and postprandial states Alpha cells Glucosedependent Glucagon (GLP-1) Inactive GIP Glucose uptake by peripheral tissue Hepatic glucose production Incretin hormones GLP-1 and GIP are released by the intestine throughout the day, and their levels in response to a meal. Concentrations of the active intact hormones are increased by JANUVIA™ (sitagliptin phosphate), thereby increasing and prolonging the actions of these hormones. GLP-1=glucagon-like peptide-1; GIP=glucose-dependent insulinotropic polypeptide.
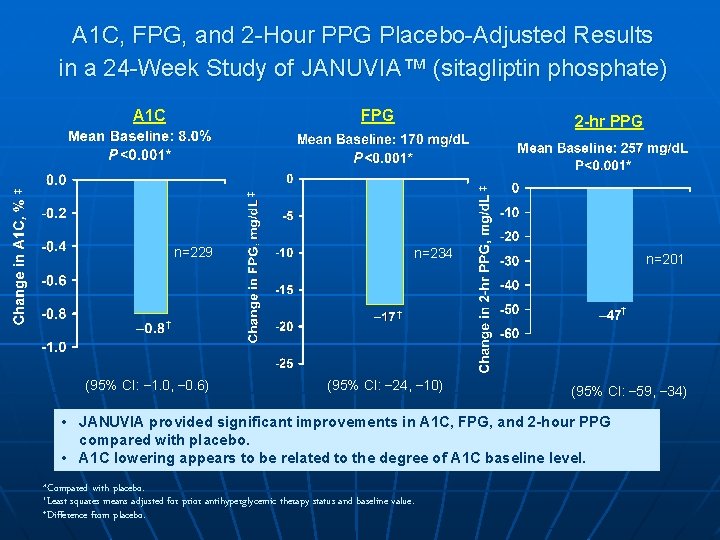
A 1 C, FPG, and 2 -Hour PPG Placebo-Adjusted Results in a 24 -Week Study of JANUVIA™ (sitagliptin phosphate) A 1 C FPG ‡ ‡ ‡ 2 -hr PPG n=229 n=234 n=201 † † † (95% CI: – 1. 0, – 0. 6) (95% CI: – 24, – 10) (95% CI: – 59, – 34) • JANUVIA provided significant improvements in A 1 C, FPG, and 2 -hour PPG compared with placebo. • A 1 C lowering appears to be related to the degree of A 1 C baseline level. *Compared with placebo. †Least squares means adjusted for prior antihyperglycemic therapy status and baseline value. ‡Difference from placebo.
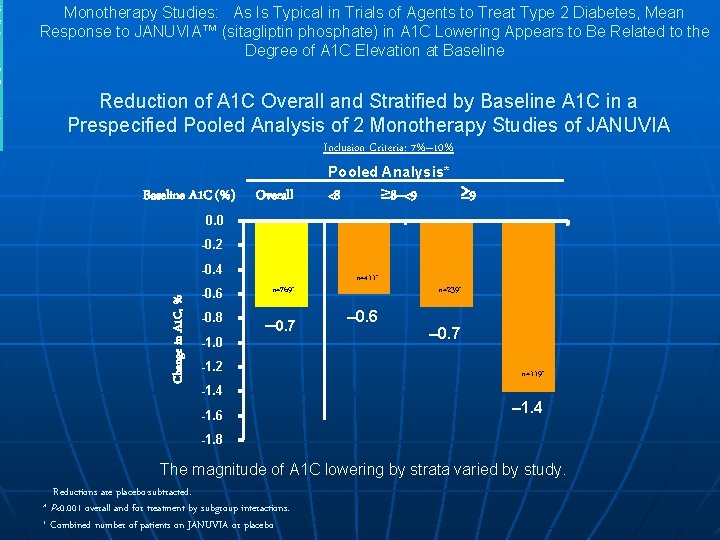
1 4. 1 Monotherapy Studies: As Is Typical in Trials of Agents to Treat Type 2 Diabetes, Mean Response to JANUVIA™ (sitagliptin phosphate) in A 1 C Lowering Appears to Be Related to the Degree of A 1 C Elevation at Baseline Reduction of A 1 C Overall and Stratified by Baseline A 1 C in a Prespecified Pooled Analysis of 2 Monotherapy Studies of JANUVIA Inclusion Criteria: 7%– 10% Pooled Analysis* Baseline A 1 C (%) Overall ≥ 8–<9 <8 ≥ 9 0. 0 -0. 2 -0. 4 Change in A 1 C, % S e c t i o n -0. 6 n=769+ -0. 8 – 0. 7 -1. 0 -1. 2 -1. 4 -1. 6 n=411+ – 0. 6 n=239+ – 0. 7 n=119+ – 1. 4 -1. 8 The magnitude of A 1 C lowering by strata varied by study. Reductions are placebo-subtracted. * P<0. 001 overall and for treatment by subgroup interactions. + Combined number of patients on JANUVIA or placebo

JANUVIA™ (sitagliptin phosphate) Effect on Body Weight n Monotherapy studies • No increase in body weight from baseline with JANUVIA compared with a small decrease in the placebo group n Add-on to metformin • A similar decrease in body weight for both treatment groups n Add-on to pioglitazone • No significant difference in body weight between treatment groups
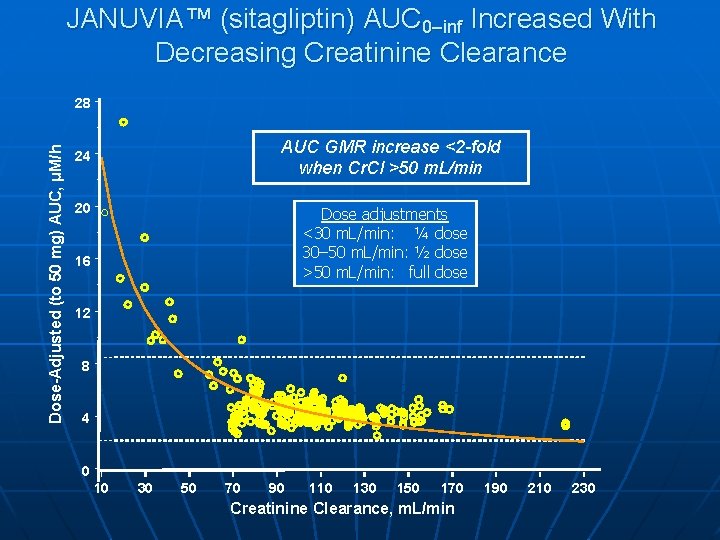
JANUVIA™ (sitagliptin) AUC 0–inf Increased With Decreasing Creatinine Clearance Dose-Adjusted (to 50 mg) AUC, μM/h 28 AUC GMR increase <2 -fold when Cr. Cl >50 m. L/min 24 20 Dose adjustments <30 m. L/min: ¼ dose 30– 50 m. L/min: ½ dose >50 m. L/min: full dose 16 12 8 4 0 10 30 50 70 90 110 130 150 170 Creatinine Clearance, m. L/min 190 210 230

Dosage and Administration Usual Dosing for JANUVIA™ (sitagliptin phosphate)* The recommended dose of JANUVIA is 100 mg once daily as monotherapy or as combination therapy with metformin or a PPAR agonist. Patients With Renal Insufficiency*, † 50 mg once daily 25 mg once daily Moderate Severe and ESRD‡ Cr. Cl 30 to <50 m. L/min (~Serum Cr levels [mg/d. L] Men: >1. 7–≤ 3. 0; Women: >1. 5–≤ 2. 5) Cr. Cl <30 m. L/min (~Serum Cr levels [mg/d. L] Men: >3. 0; Women: >2. 5) Assessment of renal function is recommended prior to initiation of JANUVIA and periodically thereafter. *JANUVIA can be taken with or without food. †Patients with mild renal insufficiency— 100 mg once daily. ‡ESRD = end-stage renal disease requiring hemodialysis or peritoneal dialysis.

PRAMLINTIDE ACETATESYMLIN

Amylin the Hormone Reported in 1987 n 37 -amino acid peptide n Co-located and co-secreted with insulin from pancreatic β-cells n Neuroendocrine hormone n Deficient in diabetes n T A T N C T A Q R L A N F L V H C Amide Y S K T N S G V N S S T L I Adapted from Unger RH, Foster DW. Williams Textbook of Endo (8 th edition) 1992; 1273 -1275 A G F S N N
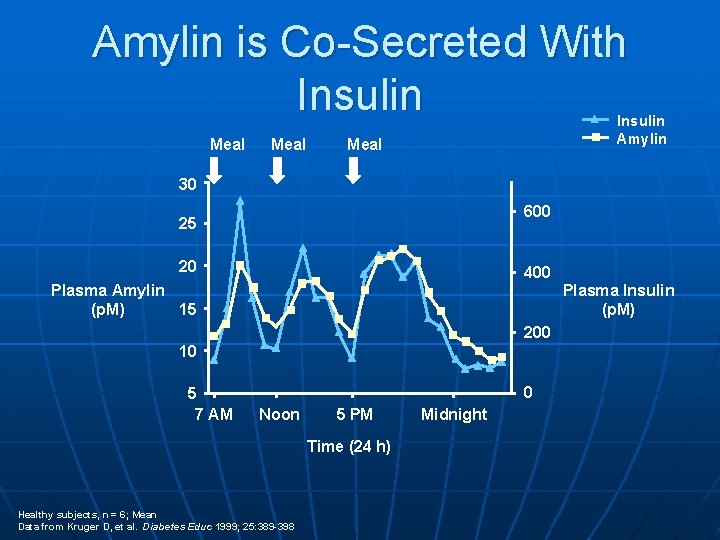
Amylin is Co-Secreted With Insulin Meal Insulin Amylin Meal 30 600 25 20 400 Plasma Amylin 15 (p. M) Plasma Insulin (p. M) 200 10 5 7 AM 0 Noon 5 PM Time (24 h) Healthy subjects, n = 6; Mean Data from Kruger D, et al. Diabetes Educ 1999; 25: 389 -398 Midnight
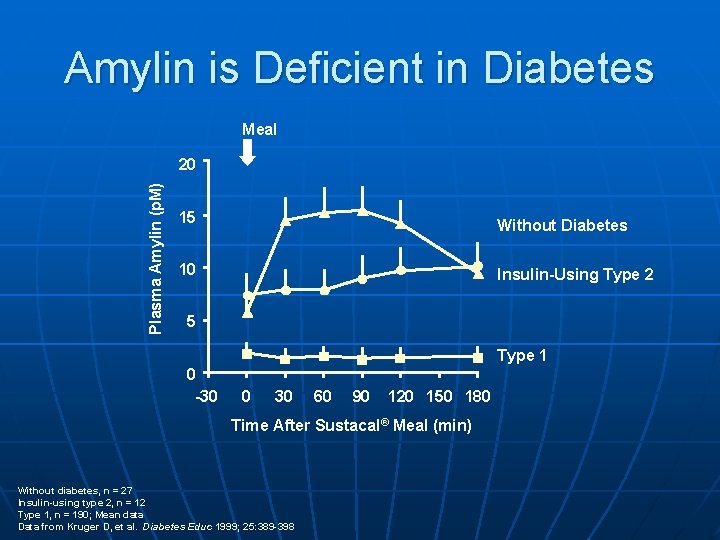
Amylin is Deficient in Diabetes Meal Plasma Amylin (p. M) 20 15 Without Diabetes 10 Insulin-Using Type 2 5 Type 1 0 -30 0 30 60 90 120 150 180 Time After Sustacal® Meal (min) Without diabetes, n = 27 Insulin-using type 2, n = 12 Type 1, n = 190; Mean data Data from Kruger D, et al. Diabetes Educ 1999; 25: 389 -398

Amylin Regulates Postprandial Glucose Appearance Mechanisms of Action Inhibits inappropriately high postprandial glucagon secretion Amylin* Slows gastric emptying Promotes satiety and reduces caloric intake *All amylin studies were performed in animals SYMLIN Prescribing Information, 2005

Pramlintide- Symlin n Synthetic analog of amylin that overcomes the tendency of human amylin to: • Aggregate, form insoluble particles • Adhere to surfaces Pharmacokinetic and pharmacodynamic properties similar to human amylin n Active ingredient of SYMLIN n Human amylin Pramlintide (synthetic analog of amylin) T A T N C T A Q R L A N F L V C Amide Y S T N S G V N S S T L I A G F S N N T N H K A T C A T Q R L A N F L V H C Amide Y S K T N S G V N T P P L I 3/1/2021 3: 51: 54 AM Adapted from Young A, et al. Drug Dev Res 1996; 37: 231 -248 Adapted from Westermark P, et al. Proc Natl Acad Sci 1990; 87: 5036 -5040 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information P G F N N S 43

SYMLIN Indications and Boxed Warning SYMLIN is given at mealtimes and is indicated for: n Type 2 diabetes, as an adjunct treatment in patients who use mealtime insulin therapy and have failed to achieve desired glucose control despite optimal insulin therapy, with or without a concurrent sulfonylurea agent and/or metformin. n Type 1 diabetes, as an adjunct treatment in patients who use mealtime insulin therapy and who have failed to achieve desired glucose control despite optimal insulin therapy. WARNING SYMLIN is used with insulin and has been associated with an increased risk of insulin-induced severe hypoglycemia, particularly in patients with type 1 diabetes. When severe hypoglycemia associated with SYMLIN use occurs, it is seen within 3 hours following a SYMLIN injection. If severe hypoglycemia occurs while operating a motor vehicle, heavy machinery, or while engaging in other high-risk activities, serious injuries may occur. Appropriate patient selection, careful patient instruction, and insulin dose adjustments are critical elements for reducing this risk. SYMLIN Prescribing Information, 2005 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information
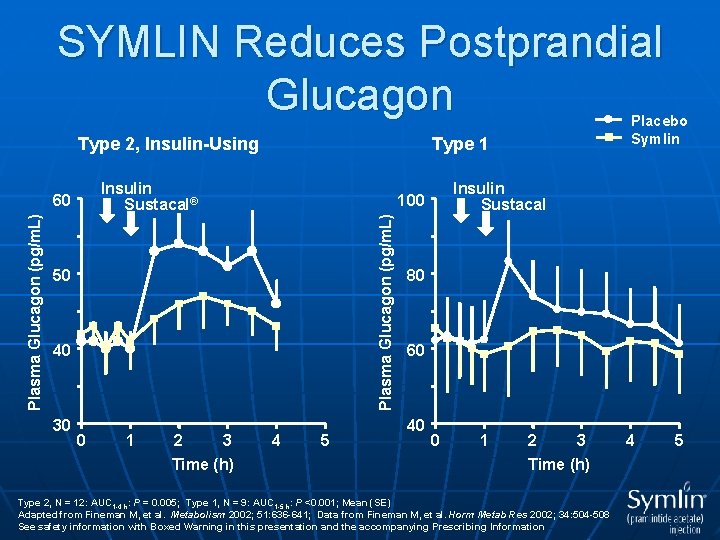
SYMLIN Reduces Postprandial Glucagon Type 2, Insulin-Using 50 40 30 0 1 2 Insulin Sustacal 100 Plasma Glucagon (pg/m. L) Type 1 Insulin Sustacal® 60 3 Time (h) Placebo Symlin 4 5 80 60 40 0 1 2 3 Time (h) Type 2, N = 12: AUC 1 -4 h: P = 0. 005; Type 1, N = 9: AUC 1 -5 h: P <0. 001; Mean (SE) Adapted from Fineman M, et al. Metabolism 2002; 51: 636 -641; Data from Fineman M, et al. Horm Metab Res 2002; 34: 504 -508 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information 4 5
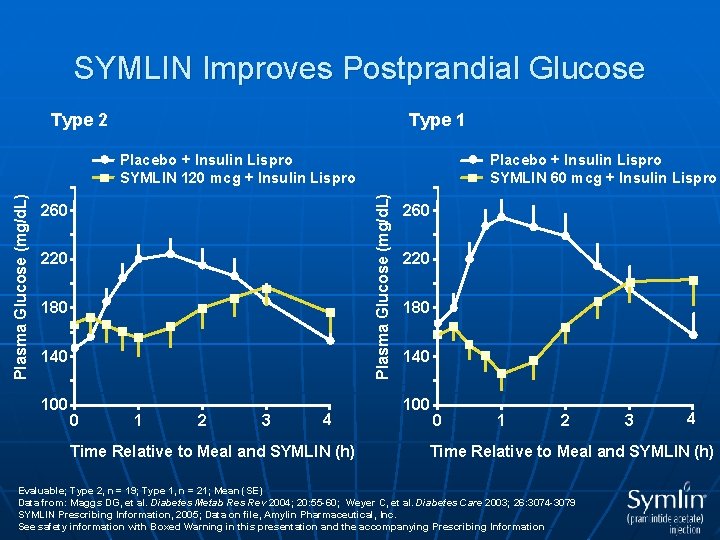
SYMLIN Improves Postprandial Glucose Type 2 Type 1 260 220 180 140 100 Placebo + Insulin Lispro SYMLIN 60 mcg + Insulin Lispro Plasma Glucose (mg/d. L) Placebo + Insulin Lispro SYMLIN 120 mcg + Insulin Lispro 0 1 2 3 4 Time Relative to Meal and SYMLIN (h) 260 220 180 140 100 0 1 2 3 4 Time Relative to Meal and SYMLIN (h) Evaluable; Type 2, n = 19; Type 1, n = 21; Mean (SE) Data from: Maggs DG, et al. Diabetes Metab Res Rev 2004; 20: 55 -60; Weyer C, et al. Diabetes Care 2003; 26: 3074 -3079 SYMLIN Prescribing Information, 2005; Data on file, Amylin Pharmaceutical, Inc. See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information
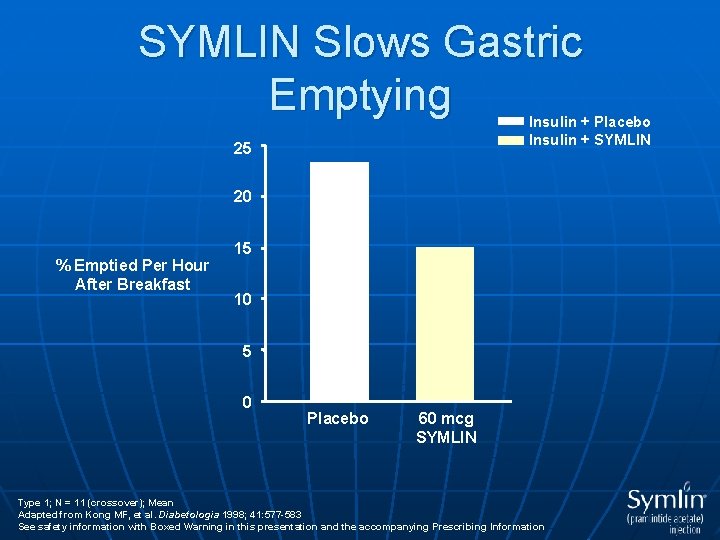
SYMLIN Slows Gastric Emptying Insulin + Placebo Insulin + SYMLIN 25 20 % Emptied Per Hour After Breakfast 15 10 5 0 Placebo 60 mcg SYMLIN Type 1; N = 11 (crossover); Mean Adapted from Kong MF, et al. Diabetologia 1998; 41: 577 -583 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information

SYMLIN Reduces Caloric Intake -202 kcal (-23%) P <0. 01 1000 750 Ad-Libitum Caloric Intake (kcal) CHO 500 CHO Fat 250 Protein Placebo 120 mcg SYMLIN 0 Type 2, n = 11 (crossover); Mean Adapted from Chapman I, et al. Diabetologia 2005; 48: 838 -848 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information
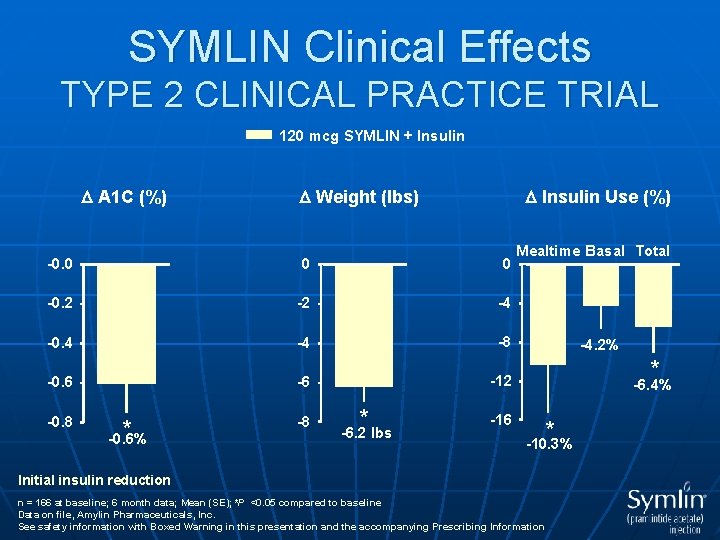
SYMLIN Clinical Effects TYPE 2 CLINICAL PRACTICE TRIAL 120 mcg SYMLIN + Insulin A 1 C (%) Weight (lbs) Insulin Use (%) -0. 0 0 0 -0. 2 -2 -4 -0. 4 -4 -8 -0. 6 -6 -12 -0. 8 * -0. 6% -8 * -6. 2 lbs Mealtime Basal Total -4. 2% * -6. 4% -16 * -10. 3% Initial insulin reduction n = 166 at baseline; 6 month data; Mean (SE); *P <0. 05 compared to baseline Data on file, Amylin Pharmaceuticals, Inc. See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information
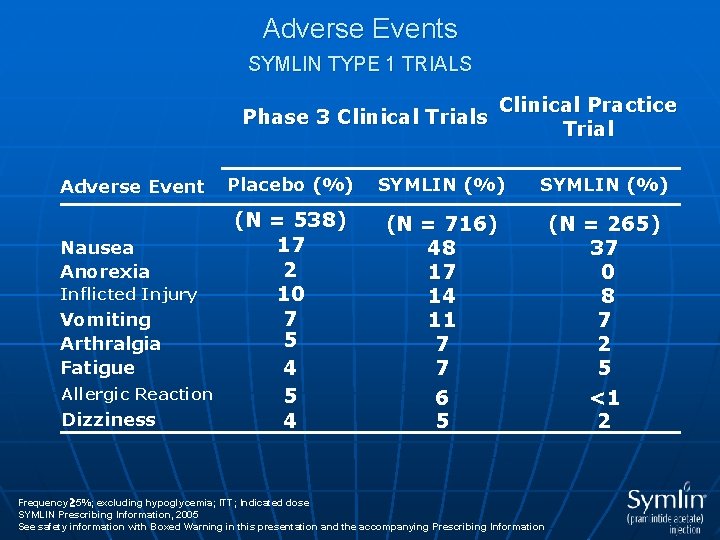
Adverse Events SYMLIN TYPE 1 TRIALS Phase 3 Clinical Trials Adverse Event Nausea Anorexia Inflicted Injury Vomiting Arthralgia Fatigue Allergic Reaction Dizziness Clinical Practice Trial Placebo (%) SYMLIN (%) (N = 538) 17 2 10 7 5 4 (N = 716) 48 17 14 11 7 7 6 5 (N = 265) 37 0 8 7 2 5 <1 2 Frequency 5%; excluding hypoglycemia; ITT; Indicated dose SYMLIN Prescribing Information, 2005 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information

SYMLIN Indications SYMLIN is given at mealtimes and is indicated for: n Type 2 diabetes, as an adjunct treatment in patients who use mealtime insulin therapy and have failed to achieve desired glucose control despite optimal insulin therapy, with or without a concurrent sulfonylurea agent and/or metformin. n Type 1 diabetes, as an adjunct treatment in patients who use mealtime insulin therapy and who have failed to achieve desired glucose control despite optimal insulin therapy. SYMLIN Prescribing Information, 2005 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information

SYMLIN Initiation n n Immediately before major meal/snack Initially reduce mealtime insulin 50% Advance SYMLIN to next dose if no significant nausea for 3 -7 days If nausea occurs and persists, reduce to previous dose Type 2: Type 1: Alternate Maintenance Dose 120 mcg 60 mcg 45 mcg 30 mcg 60 mcg 15 mcg Initiation n Maintenance Dose Adjust insulin based on self-monitoring of blood glucose SYMLIN Prescribing Information, 2005 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information
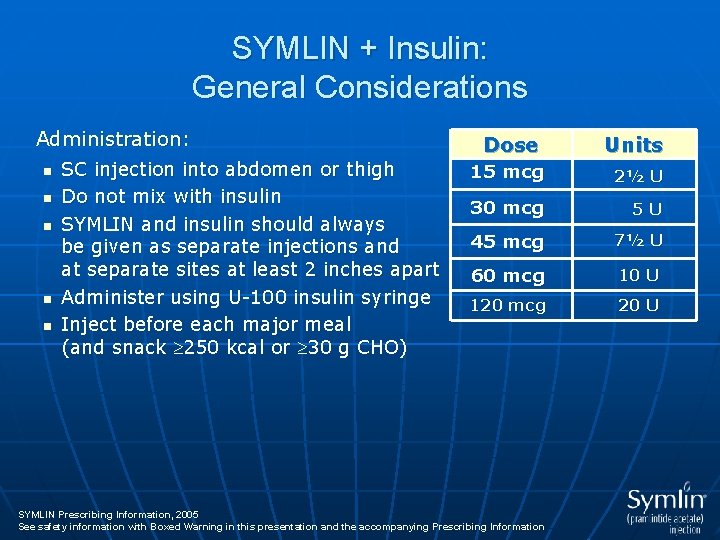
SYMLIN + Insulin: General Considerations Administration: n n n SC injection into abdomen or thigh Do not mix with insulin SYMLIN and insulin should always be given as separate injections and at separate sites at least 2 inches apart Administer using U-100 insulin syringe Inject before each major meal (and snack 250 kcal or 30 g CHO) Dose Units 15 mcg 2½ U 30 mcg 5 U 45 mcg 7½ U 60 mcg 10 U 120 mcg 20 U SYMLIN Prescribing Information, 2005 See safety information with Boxed Warning in this presentation and the accompanying Prescribing Information

EXUBERA

Respiratory Tract Surface Area
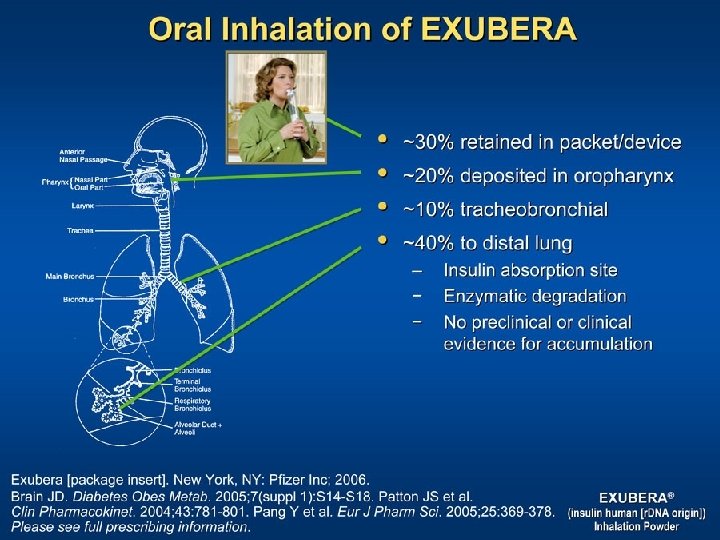






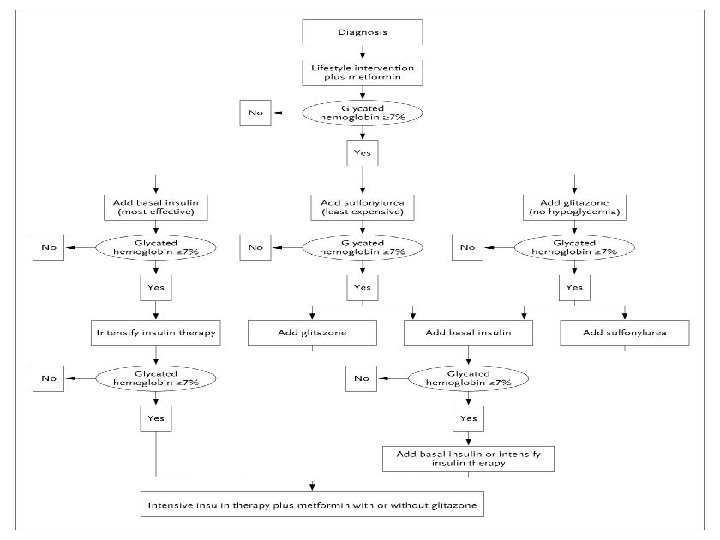
Metabolic Management of Type 2 Diabetes Nathan D. N Engl J Med 2006; 355: 2477 -2480
- Slides: 54