New Agents in Management of Indolent BCell NHLs

New Agents in Management of Indolent B-Cell NHLs F B Hagemeister MD Department of Lymphoma/Myeloma M D Anderson Cancer Center Las Vegas, Nevada February 27, 2014

New Agents in Management of Indolent B-Cell NHLs • • • Monoclonal Antibodies • Rituximab • Ofatumumab • Obinutuzumab Protein Inhibitors • BTK Inhibitors • PI 3 K Inhibitors • Others Immunomodulatory Agents

Phase III MR, 2 Schedules, for Untreated FL Following SA Rituximab x 4: SAKK 35/03 • Med PFS: (MR = 4 doses, vs 5 years, both q 2 mo): – For all 165 enrolled: A-3. 4 yr vs B-5. 3 (p=0. 14) – Thought due to higher relapse rate before MR begun for those in Gp A for “unexplained reasons” – For only those in remission at 8 mo: A-7. 1 yr vs B 2. 9 (p=0. 004) • Gr 3 -4 infections (Pt): A-1 vs B-5 • No differences in OS or ORR • Conclusion: If in remission after SA Rituximab, MR prevents relapse if given longer vs shorter interval Taverna et al, ASH 2013, # 508 But best duration of MR is still unclear.
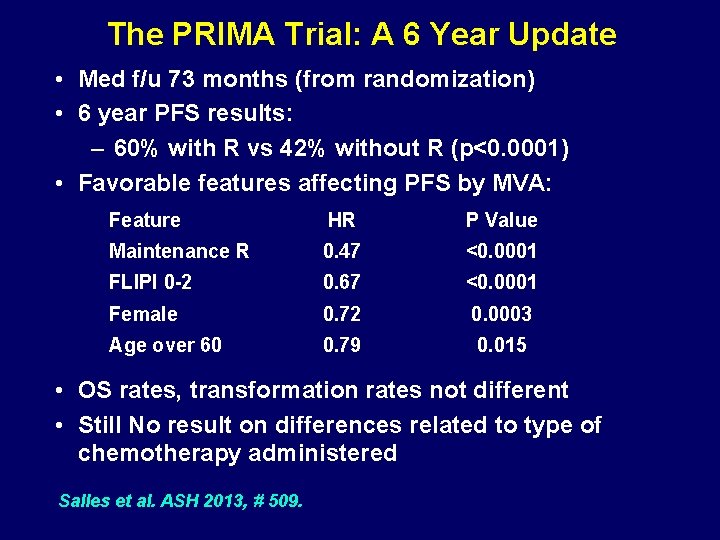
The PRIMA Trial: A 6 Year Update • Med f/u 73 months (from randomization) • 6 year PFS results: – 60% with R vs 42% without R (p<0. 0001) • Favorable features affecting PFS by MVA: Feature HR P Value Maintenance R 0. 47 <0. 0001 FLIPI 0 -2 0. 67 <0. 0001 Female 0. 72 0. 0003 Age over 60 0. 79 0. 015 • OS rates, transformation rates not different • Still No result on differences related to type of chemotherapy administered Salles et al. ASH 2013, # 509.
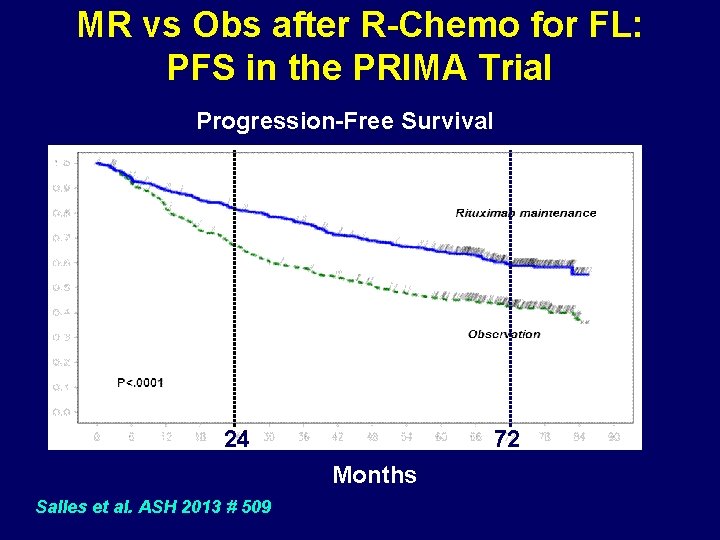
MR vs Obs after R-Chemo for FL: PFS in the PRIMA Trial Progression-Free Survival 24 72 Months Salles et al. ASH 2013 # 509
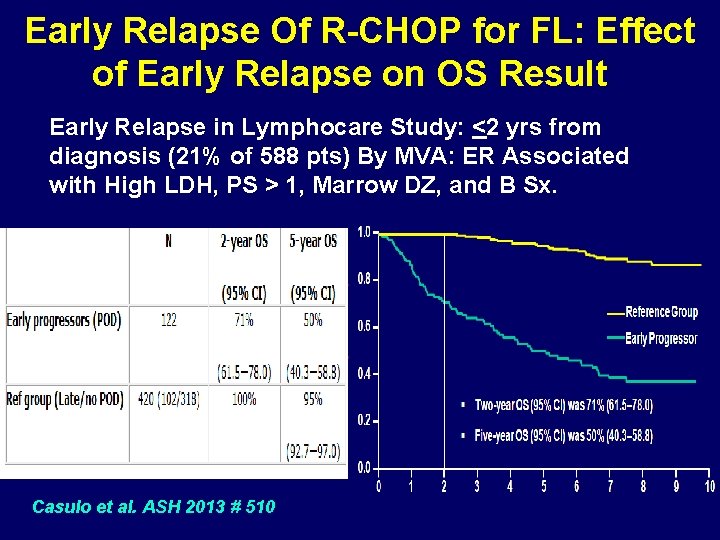
Early Relapse Of R-CHOP for FL: Effect of Early Relapse on OS Result Early Relapse in Lymphocare Study: <2 yrs from diagnosis (21% of 588 pts) By MVA: ER Associated with High LDH, PS > 1, Marrow DZ, and B Sx. Casulo et al. ASH 2013 # 510

90 Y-Ibritumomab for Advanced Stage FL in First Remission: The FIT trial Med PFS Results (Mo) Control 90 Y I P HR Patients in CR (%) 53. 3 87. 4 NR NR All Pts 13. 5 37 <0. 0001 0. 46 Initial PR 6. 3 29. 7 <0. 0001 0. 30 Initial CR 29. 9 54. 6 . 01 0. 61 FLIPI, 0 -1 24. 1 NR 0. 05 0. 6 2 11. 3 53. 9 <0. 1 0. 23 3 -5 6. 5 23. 8 0. 08 0. 59 * All patients Note: Only 10 -15% had received induction rituximab. A second randomized trial was planned with induction R-CHOP. High-risk pts receive RIT vs SCT, low risk receive RIT vs Obs. All would receive maintenance rituximab. But trial abandoned Hagenbeek et. al. ASH 2007, Abstract # 693
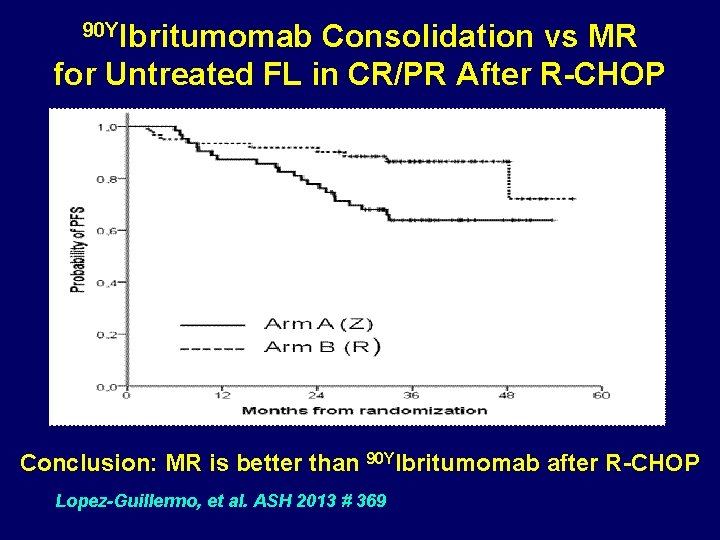
90 YIbritumomab Consolidation vs MR for Untreated FL in CR/PR After R-CHOP Conclusion: MR is better than 90 YIbritumomab after R-CHOP Lopez-Guillermo, et al. ASH 2013 # 369

New Agents in Management of Indolent B-Cell NHLs • • • Monoclonal Antibodies • Rituximab • Ofatumumab • Obinutuzumab Protein Inhibitors • BTK Inhibitors • PI 3 K Inhibitors • Others Immunomodulatory Agents

Novel Anti-CD 20 Mo. Abs for Relapsed/Refractory Indolent Lymphomas Mo. Ab Phase Efficacy I/II Dose (ORR): 300 mg (63%); 500 mg (33%); 700 mg (20%); 1000 mg (50%) II ORR: 11%, 6 -mo PFS in 116 pts with rituximab-refractory FL Ofatumumab IV administration: ORR: 44%, CR: 27% DOR in pts with FL: 19. 7 mos Veltuzumab I/II Ocrelizumab I/II ORR: 38%; PFS: 11. 4 mos in pts with FL I/II/III ORR: 69%, CR: 38% in 13 pts with FL GA 101 Subcutaneous administration: ORR: 53% CR: 20% in pts with indolent NHL Morschhauser. Ann Oncol. 2010; Morschhauser. JCO. 2009; 27: 3346; Negrea. ASH. 2009 (abstr 3757); Hagenbeek. ASH. 2009 (abstr 935); Hagenbeek. Blood. 2008; 111: 5486; Salles. ASH. 2009 (abstr 1704).

New Agents in Management of Indolent B-Cell NHLs • • • Monoclonal Antibodies • Rituximab • Ofatumumab • Obinutuzumab Protein Inhibitors • BTK Inhibitors • PI 3 K Inhibitors • Others Immunomodulatory Agents

Obinutuzumab vs Rituximab for Rel i. NHL: The GAUSS Study • 175 pt with rel CD 20 pos i. NHL, 149 had FL • Eligibility: CR or PR to rituximab-based therapy with response lasting > 6 months • Features: Median 2 prior Txs, others balanced • Therapy: G 1000 mg q wk X 4 or R X 4. 4 -6 wks later, pt with CR, PR, SD could receive drug q 2 mo X 2 yrs • Infusion-related RXNs more common with GA-101 (72% vs 49%, any Gr) • By IR panel, OR for all: G-42% vs R-24%; for FL: G-43% vs R-28% Sehn et al. ASH 2011, abst 269.

Obinutuzumab plus FC or CHOP for Rel/Ref FL: Phase I GAUDI Study • Obinutuzumab (GA-101): glycosylated, Type II moab against CD 20 • 56 pt, stratified by prior therapy • Two Ob regimens chosen based on phase I trial: 1600 mg d 1+8, cycle 1, then 800 mg d 1+8 vs 400/400 for max 8 (CHOP) or 6 (FC) cycles • Toxicity: Not increased with higher doses of Ob • Results: OR=96% (G-CHOP), 93% (G-FC) CR=39%(G-CHOP), 50% (G-FC) • Basis for new G-CHOP study vs R-CHOP for untreated FL Radford et al: ASH 2011, abst 270.
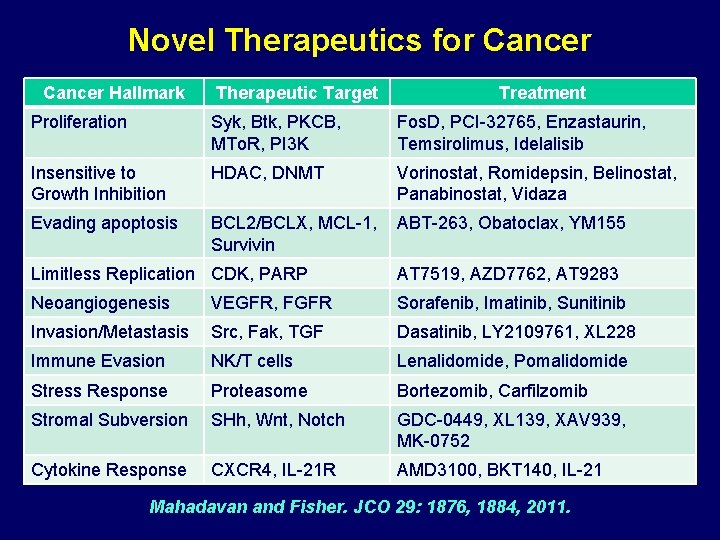
Novel Therapeutics for Cancer Hallmark Therapeutic Target Treatment Proliferation Syk, Btk, PKCB, MTo. R, PI 3 K Fos. D, PCI-32765, Enzastaurin, Temsirolimus, Idelalisib Insensitive to Growth Inhibition HDAC, DNMT Vorinostat, Romidepsin, Belinostat, Panabinostat, Vidaza Evading apoptosis BCL 2/BCLX, MCL-1, Survivin ABT-263, Obatoclax, YM 155 Limitless Replication CDK, PARP AT 7519, AZD 7762, AT 9283 Neoangiogenesis VEGFR, FGFR Sorafenib, Imatinib, Sunitinib Invasion/Metastasis Src, Fak, TGF Dasatinib, LY 2109761, XL 228 Immune Evasion NK/T cells Lenalidomide, Pomalidomide Stress Response Proteasome Bortezomib, Carfilzomib Stromal Subversion SHh, Wnt, Notch GDC-0449, XL 139, XAV 939, MK-0752 Cytokine Response CXCR 4, IL-21 R AMD 3100, BKT 140, IL-21 Mahadavan and Fisher. JCO 29: 1876, 1884, 2011.
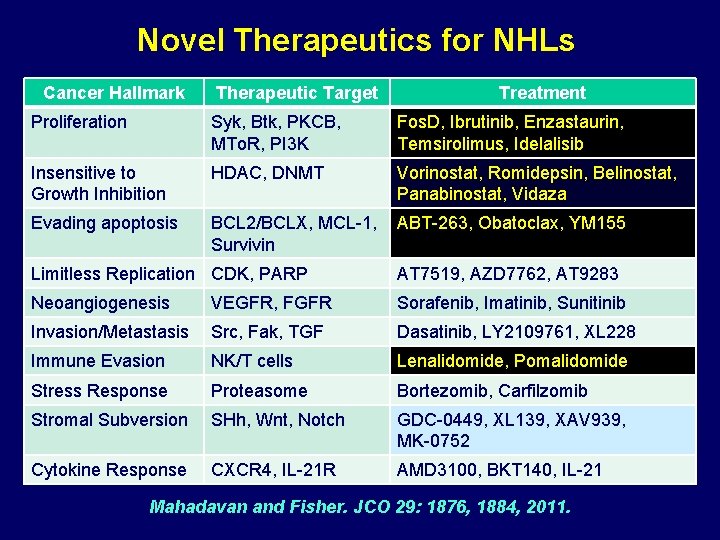
Novel Therapeutics for NHLs Cancer Hallmark Therapeutic Target Treatment Proliferation Syk, Btk, PKCB, MTo. R, PI 3 K Fos. D, Ibrutinib, Enzastaurin, Temsirolimus, Idelalisib Insensitive to Growth Inhibition HDAC, DNMT Vorinostat, Romidepsin, Belinostat, Panabinostat, Vidaza Evading apoptosis BCL 2/BCLX, MCL-1, Survivin ABT-263, Obatoclax, YM 155 Limitless Replication CDK, PARP AT 7519, AZD 7762, AT 9283 Neoangiogenesis VEGFR, FGFR Sorafenib, Imatinib, Sunitinib Invasion/Metastasis Src, Fak, TGF Dasatinib, LY 2109761, XL 228 Immune Evasion NK/T cells Lenalidomide, Pomalidomide Stress Response Proteasome Bortezomib, Carfilzomib Stromal Subversion SHh, Wnt, Notch GDC-0449, XL 139, XAV 939, MK-0752 Cytokine Response CXCR 4, IL-21 R AMD 3100, BKT 140, IL-21 Mahadavan and Fisher. JCO 29: 1876, 1884, 2011.

New Agents in Management of Indolent B-Cell NHLs • • • Monoclonal Antibodies • Rituximab • Ofatumumab • Obinutuzumab Protein Inhibitors • BTK Inhibitors • PI 3 K Inhibitors • Others Immunomodulatory Agents
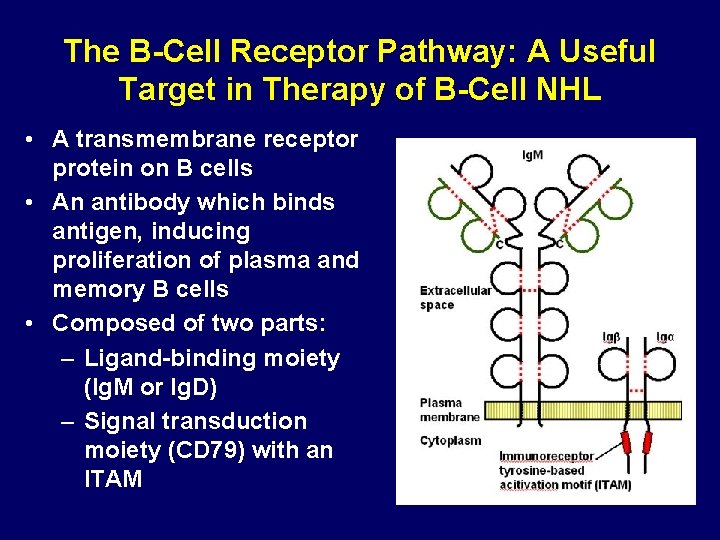
The B-Cell Receptor Pathway: A Useful Target in Therapy of B-Cell NHL • A transmembrane receptor protein on B cells • An antibody which binds antigen, inducing proliferation of plasma and memory B cells • Composed of two parts: – Ligand-binding moiety (Ig. M or Ig. D) – Signal transduction moiety (CD 79) with an ITAM
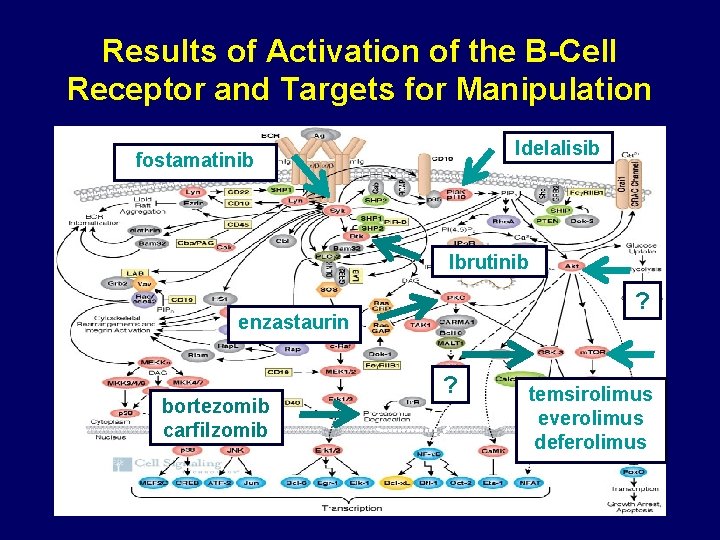
Results of Activation of the B-Cell Receptor and Targets for Manipulation Idelalisib fostamatinib Ibrutinib ? enzastaurin bortezomib carfilzomib ? temsirolimus everolimus deferolimus

Phase I Ibrutinib for Relapsed NHL/CLL: Response Rates Histology N CR PR SD ORR% (ITT, n=56) ORR% (Eval, n=50) CLL/SLL 16 1 10 3* 69 79 MCL 9 3 4 1 78 78 WM 4 3** 1 75 75 FL 16 3 3 38 46 MZL/MALT 4 1 1 25 33 DLBCL 7 2 1 29 29 TOTAL 56 24 9 55 62 3 7 • * 1 CLL pt had nodal response, but increased lymphocytosis • ** On the basis of decreased Ig. M Advani R, Fowler N et al. ICML 2011.
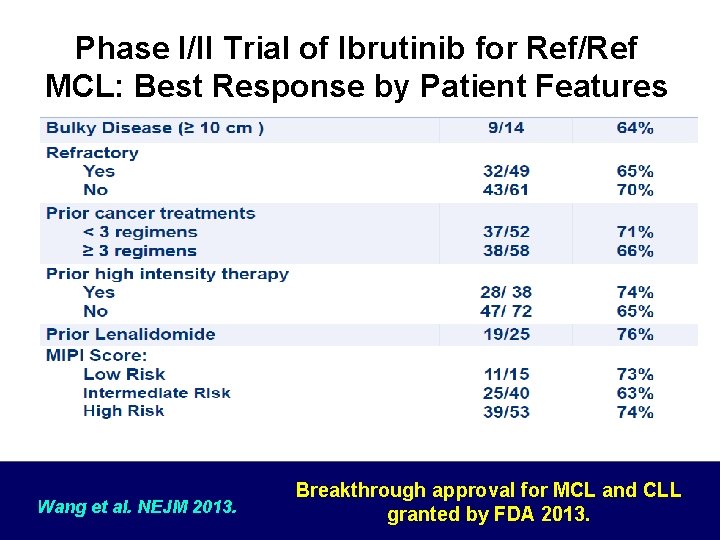
Phase I/II Trial of Ibrutinib for Ref/Ref MCL: Best Response by Patient Features Wang et al. NEJM 2013. Breakthrough approval for MCL and CLL granted by FDA 2013.
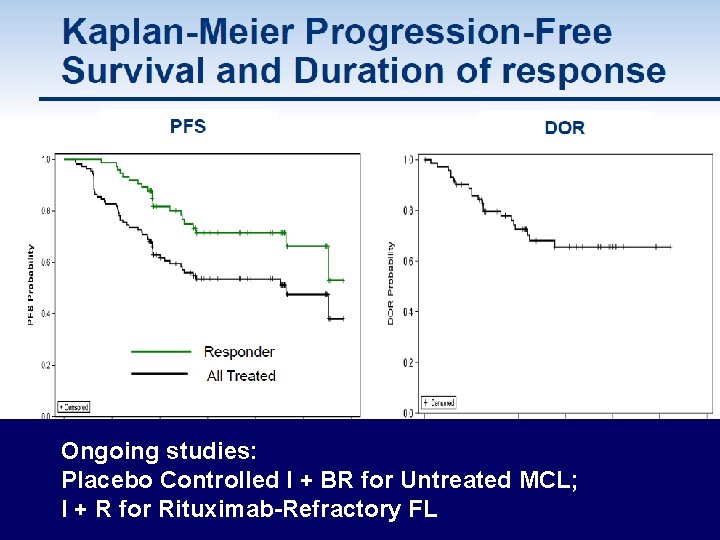
Ongoing studies: Placebo Controlled I + BR for Untreated MCL; I + R for Rituximab-Refractory FL

New Agents in Management of Indolent B-Cell NHLs • • • Monoclonal Antibodies • Rituximab • Ofatumumab • Obinutuzumab Protein Inhibitors • BTK Inhibitors • PI 3 K Inhibitors • Others Immunomodulatory Agents
PI 3 Kδ Inhibition Impacts Multiple Critical Pathways in i. NHL
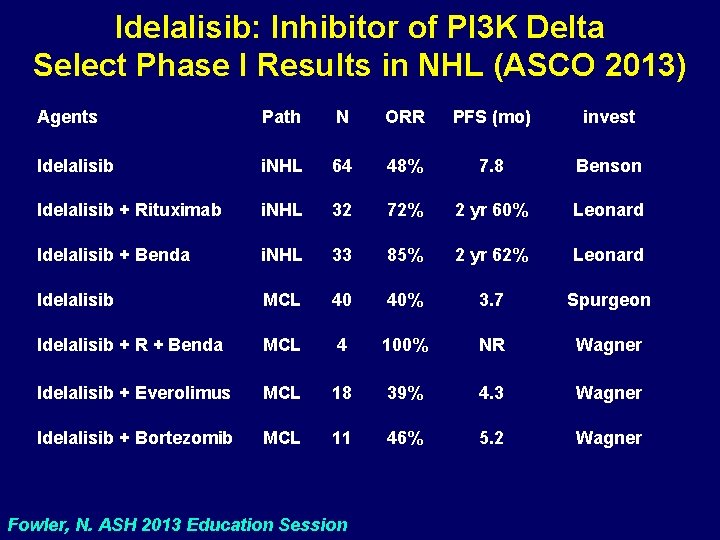
Idelalisib: Inhibitor of PI 3 K Delta Select Phase I Results in NHL (ASCO 2013) Agents Path N ORR PFS (mo) invest Idelalisib i. NHL 64 48% 7. 8 Benson Idelalisib + Rituximab i. NHL 32 72% 2 yr 60% Leonard Idelalisib + Benda i. NHL 33 85% 2 yr 62% Leonard Idelalisib MCL 40 40% 3. 7 Spurgeon Idelalisib + R + Benda MCL 4 100% NR Wagner Idelalisib + Everolimus MCL 18 39% 4. 3 Wagner Idelalisib + Bortezomib MCL 11 46% 5. 2 Wagner Fowler, N. ASH 2013 Education Session
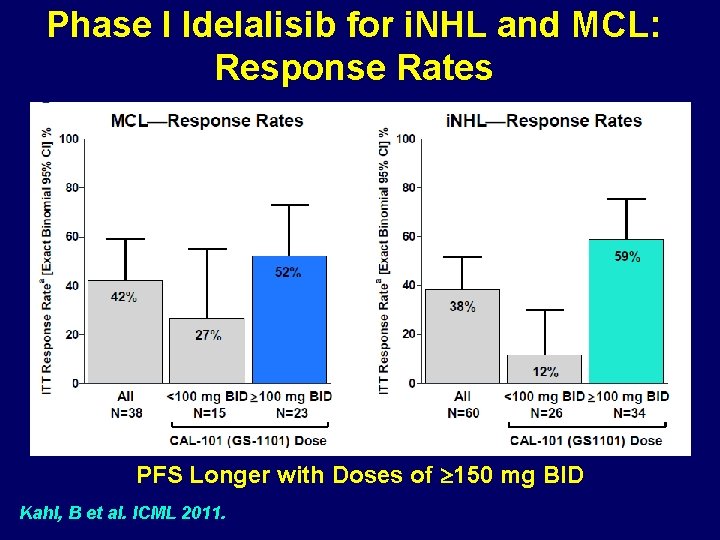
Phase I Idelalisib for i. NHL and MCL: Response Rates PFS Longer with Doses of 150 mg BID Kahl, B et al. ICML 2011.
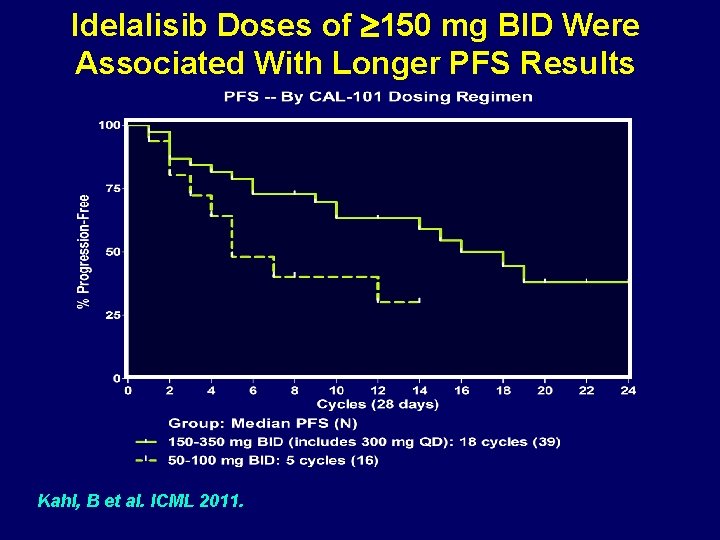
Idelalisib Doses of 150 mg BID Were Associated With Longer PFS Results Kahl, B et al. ICML 2011.
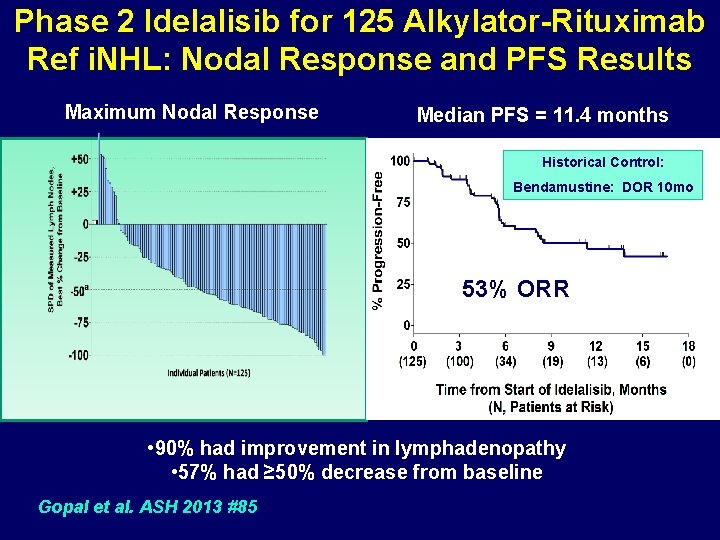
Phase 2 Idelalisib for 125 Alkylator-Rituximab Ref i. NHL: Nodal Response and PFS Results Maximum Nodal Response Median PFS = 11. 4 months Historical Control: Bendamustine: DOR 10 mo 53% ORR • 90% had improvement in lymphadenopathy • 57% had ≥ 50% decrease from baseline Gopal et al. ASH 2013 #85

Phase 1 Idelalisib for 40 Rel/Ref MCL: Dose 50 -350 mg po BID Continuously Features Median age (range), years (N = 40) 69 (52 -83) Age >60 35 (88) PS ≥ 2 4 (10) LDH > ULN 19 (48) Bulky Disease (>5 cm) 24 (60) MIPI-High (>6. 2) 14 (35) Med No Prior TX (Range) 4 (1 -14) Refractory to Last TX (< 6 mo) 17 (43) ORR: 16 (40%), CR 3 (7. 5%) ORR at < 150 = 29% (8/28), ORR with > 150 = 67% (8/12) Spurgeon et al. Lugano 2013.
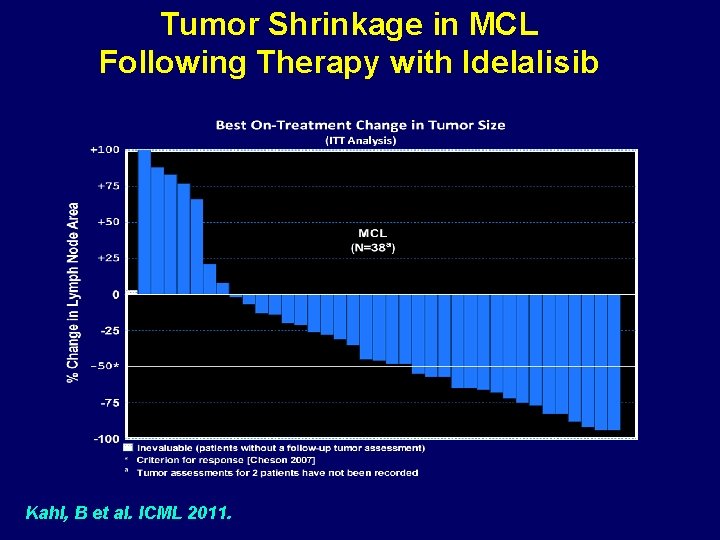
Tumor Shrinkage in MCL Following Therapy with Idelalisib Kahl, B et al. ICML 2011.
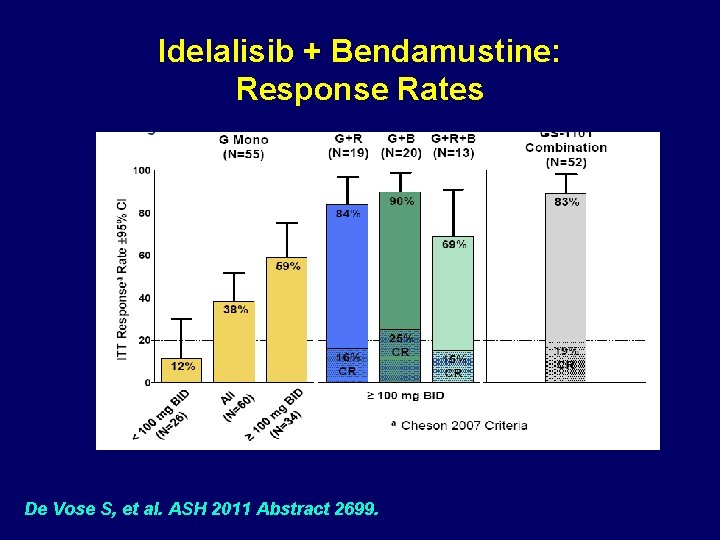
Idelalisib + Bendamustine: Response Rates De Vose S, et al. ASH 2011 Abstract 2699.
Other PI 3 k Inhibitors for Rel/Ref FL: The ARD 12130 Study and BAY 80 -6046 • Phase II SAR 245409: Inhibits Isos α, β, γ and δ, m. TORC 1, TORC 2. – Study enrolls FL, CLL/SLL, MCL, and DLBCL. – Phase 2, Stage 1 results for FL (Gr 1, 2, 3 A) reported. – Doses: 50 mg PO BID; Resp: ORR= 12/24 (50%), CR=2/24 (8%) – AEs: Diarrhea, Pneumonias, cataracts • BAY 80– 6946, Inhibits Isoforms δ and α. May overcome resistance to PI 3 K-δ. • Phase II Study: 27 i. NHL and 34 agg. NHLs • Med Prior TXs: 3. Prior ASCT: 20% Brown et al. ASH 2013 # 86, Dreyling et al. ASH 2013 # 87.

New Agents in Management of Indolent B-Cell NHLs • • • Monoclonal Antibodies • Rituximab • Ofatumumab • Obinutuzumab Protein Inhibitors • BTK Inhibitors • PI 3 K Inhibitors • Others Immunomodulatory Agents
Aurora Kinase A and B: Effects on the Cell Cycle Meraldi et al. Curr Op Genet Dev 2004.
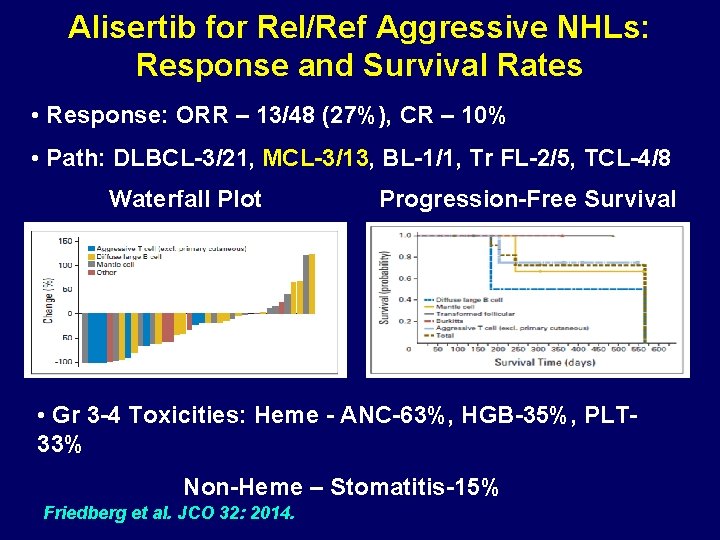
Alisertib for Rel/Ref Aggressive NHLs: Response and Survival Rates • Response: ORR – 13/48 (27%), CR – 10% • Path: DLBCL-3/21, MCL-3/13, BL-1/1, Tr FL-2/5, TCL-4/8 Waterfall Plot Progression-Free Survival • Gr 3 -4 Toxicities: Heme - ANC-63%, HGB-35%, PLT 33% Non-Heme – Stomatitis-15% Friedberg et al. JCO 32: 2014.

Alisertib in Aggressive B-Cell and T-Cell NHL: Response Rates • ORR: 32% (95% CI: 18 -48) in overall population and responses observed in all histologic disease subtypes Response, % ORR Pts (N = 41) 32 ORR by Histology, * % B-cell § CR 12 § DLBCL 20 § PR 20 § MCL 23 SD 39 § Transformed FL 40 PD 29 § Burkitt’s lymphoma 100 T-cell Friedberg J, et al. ASH 2011. Abstract 95. 57

Potential Effects of Anti-PD-1 Antibody in Therapy of Cancer Anti-PD-1 Antibodies: Pidilizumab, Nivolumab, Lambrolizumab Mc. Dermott and Atkins. Cancer Medicine 2: 662 -673, 2013.
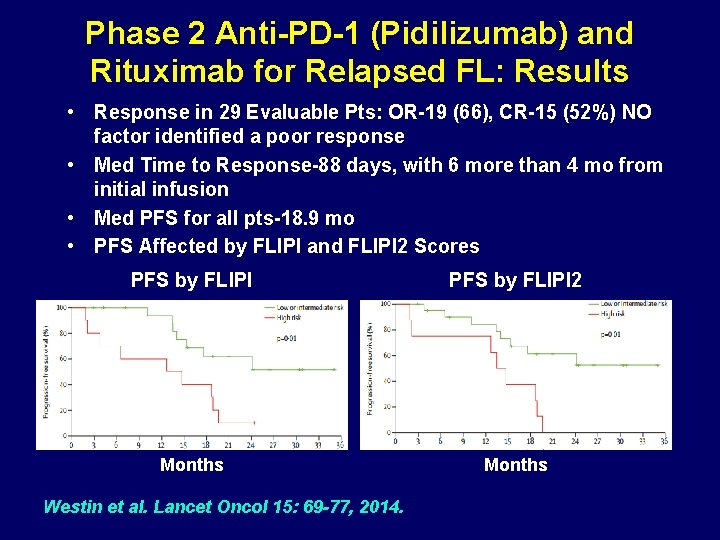
Phase 2 Anti-PD-1 (Pidilizumab) and Rituximab for Relapsed FL: Results • Response in 29 Evaluable Pts: OR-19 (66), CR-15 (52%) NO factor identified a poor response • Med Time to Response-88 days, with 6 more than 4 mo from initial infusion • Med PFS for all pts-18. 9 mo • PFS Affected by FLIPI and FLIPI 2 Scores PFS by FLIPI 2 Months Westin et al. Lancet Oncol 15: 69 -77, 2014.
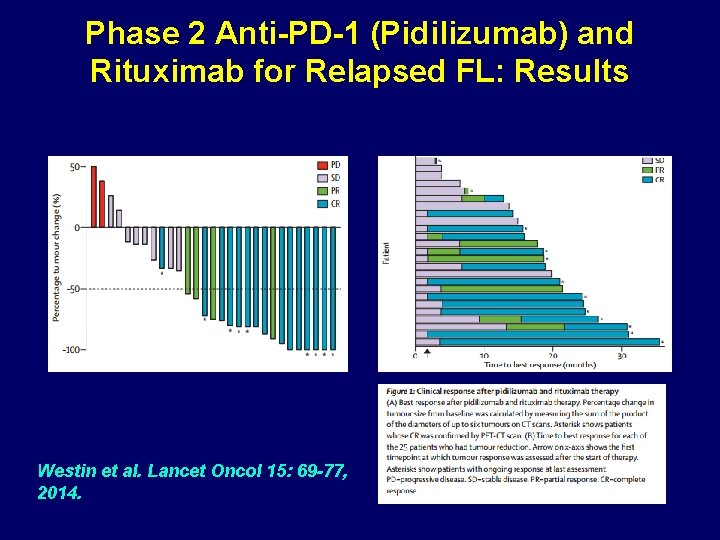
Phase 2 Anti-PD-1 (Pidilizumab) and Rituximab for Relapsed FL: Results Westin et al. Lancet Oncol 15: 69 -77, 2014.

New Agents in Management of Indolent B-Cell NHLs • • • Monoclonal Antibodies • Rituximab • Ofatumumab • Obinutuzumab Protein Inhibitors • BTK Inhibitors • PI 3 K Inhibitors • Others Immunomodulatory Agents

Cereblon A Target for Lenalidomide? • Cereblon: Component of the E 3 ubiquitin ligase complex • Target protein for thalidomide, lenalidomide and pomalidomide – These Inhibit the ubiquitin ligase activity • May explain many of the known effects of immunomodulatory agents: – Teratogenic activity – Anti-myeloma activity – T-cell activation 40
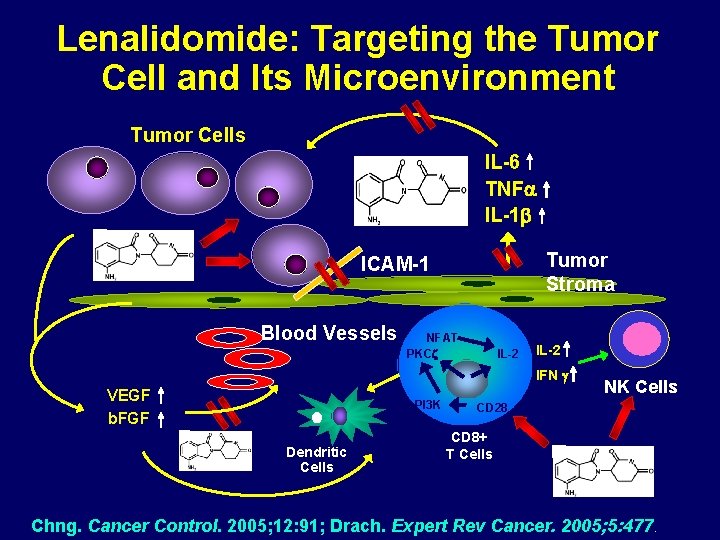
Lenalidomide: Targeting the Tumor Cell and Its Microenvironment Tumor Cells IL-6 TNF IL-1 Tumor Stroma ICAM-1 Blood Vessels NFAT PKC IL-2 IFN VEGF b. FGF PI 3 K Dendritic Cells NK Cells CD 28 CD 8+ T Cells Chng. Cancer Control. 2005; 12: 91; Drach. Expert Rev Cancer. 2005; 5: 477.
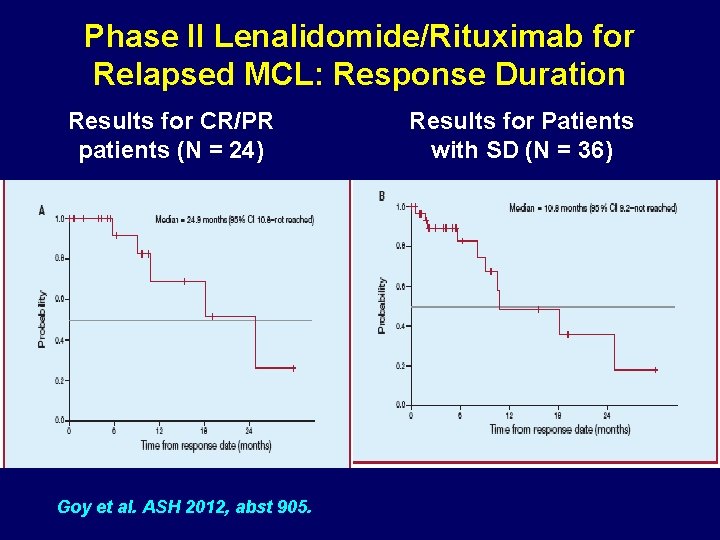
Phase II Lenalidomide/Rituximab for Relapsed MCL: Response Duration Results for CR/PR patients (N = 24) Goy et al. ASH 2012, abst 905. Results for Patients with SD (N = 36)

Rituximab and Lenalidomide for Untreated i. NHL: Study Design 1 2 3 4 5 6 7 8 9 10 11 12 Lenalidomide 20 mg Days 1 -21 Cycles 1 -6* Lenalidomide 20 mg Days 1 -21 Cycles 7 -12* Rituximab 375 mg/M 2 Day 1 of Cycles 1 -6 Rituximab 375 mg/M 2 Day 1 of Cycles 7 -12 R = RESTAGE R If clinical benefit, can proceed to 12 cycles *For SLL patients: Dose escalation of lenalidomide starting with cycle 1: (10 mg, 15 mg, 20 mg) Fowler N, et al ASH 2012. R R R • Phase II, single institution • Planned Enrollment • 50 Follicular Lymphoma (grade I/II) • 30 Small Lymphocytic Lymphoma • 30 Marginal Zone Lymphoma • Groups analyzed independently for response and toxicity
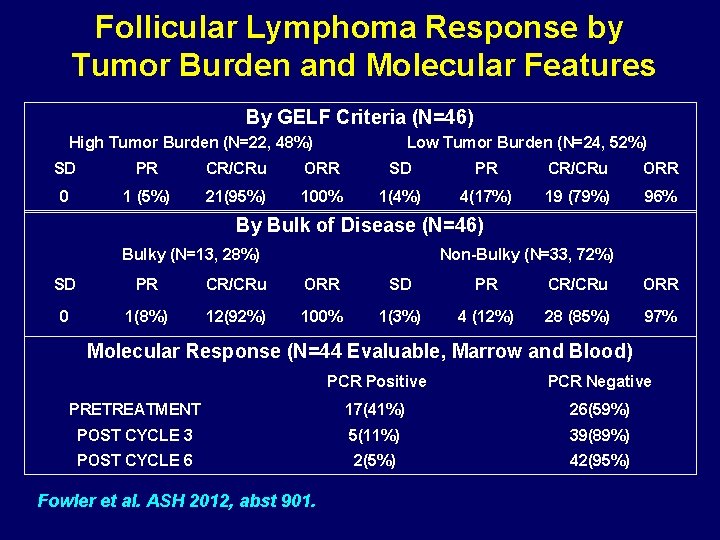
Follicular Lymphoma Response by Tumor Burden and Molecular Features By GELF Criteria (N=46) High Tumor Burden (N=22, 48%) Low Tumor Burden (N=24, 52%) SD PR CR/CRu ORR 0 1 (5%) 21(95%) 100% 1(4%) 4(17%) 19 (79%) 96% By Bulk of Disease (N=46) Bulky (N=13, 28%) Non-Bulky (N=33, 72%) SD PR CR/CRu ORR 0 1(8%) 12(92%) 100% 1(3%) 4 (12%) 28 (85%) 97% Molecular Response (N=44 Evaluable, Marrow and Blood) PCR Positive PCR Negative PRETREATMENT 17(41%) 26(59%) POST CYCLE 3 5(11%) 39(89%) POST CYCLE 6 2(5%) 42(95%) Fowler et al. ASH 2012, abst 901.

Lenalidomide Plus Rituximab as Initial Therapy for i. NHL: Response Rates • • All Patients Resp, % SLL, N=30 MZL, N=27 FL, N=46 ITT, Eval, N=103 N=110 ORR 80 89 98 90 85 § CR/CRu 27 67 87 64 60 § PR 53 22 11 26 25 SD 13 11 2 8 7 PD 7 0 0 2 2 Responses for FL independent of GELF criteria or disease bulk Molecular responses for FL increased with treatment duration Molecular Response, % Pretreatment After cycle 3 After cycle 6 PCR POS PCR NEG 41 11 5 59 89 95 *Major or minor breakpoints from bone marrow, peripheral blood samples. Fowler et al. ASH 2012. Abst 901.

Lenalidomide/Rituximab for i. NHLs: PFS by Histology Follicular Lymphoma Marginal Zone N=46 N=27 36 mo PFS: 81% 36 mo PFS: 89% SLL N=30 Fowler et al. ASH 2012, abst 901. 36 mo PFS: 66%

Lenalidomide-R for FcγRIIIa-F i. NHLs or MCLs Refractory To Rituximab • FCGR 3 A polymorphisms cause significant impact on ORR, CR rate, and TTP after SA rituximab (ORR 26% and 2 -Yr PFS of only 14% if F allele present). • Study: R-Refractory (SA or chemo combo) and F • Schema: 2 mo Len/Dex (10 mg QD/8 mg QWk, Part 1), then Rituximab q Wk X 4 with Len/Dex (Part 2), then continue Len/Dex alone. • Pts: 17/18 tested had F/F alleles, one V/V. Resp All F MCL , SLL , MZL Pts 17 12 5 24/53% 25/50% 24/51% 3/5 pt 2/4 pt 1/1 pt ORR (Part 1/2) Cartron 2002, Weng 2003, Chong et al. ASH 2013, #250.
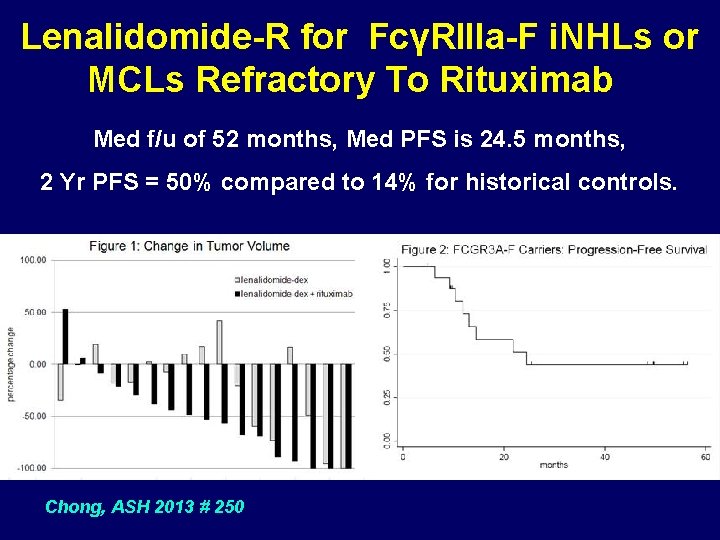
Lenalidomide-R for FcγRIIIa-F i. NHLs or MCLs Refractory To Rituximab Med f/u of 52 months, Med PFS is 24. 5 months, 2 Yr PFS = 50% compared to 14% for historical controls. Chong, ASH 2013 # 250

Phase II Lenalidomide-R-CHOP for Untreated High- Risk (GELF) FL • Patients: 80 with FL Gr 1, 2, 3 a; Med age 57, High LDH -40%, FLIPI 3 -5 in 63%, Mass > 10 cm-25% • Therapy: Induction of Standard R-CHOP, plus Len 25 mg QD days 1 -14, X 6 cycles + 2 R doses – Maintenance: MR q 8 wks X 2 yrs – Supportive: Pegfilgrastim day 4, QASA 10 mg QD • Med F/U 12 mo: ORR – 94%, CR/CRu – 74% – Gr 4 Toxicity: ANC-64%, PLT-12. 5% – Gr 3 neuropathy: 36%, Gr 1 -3 rash (2 Gr 3) – Thrombosis in 5 (3 catheter related) Tilly et al. ASH 2013 # 248
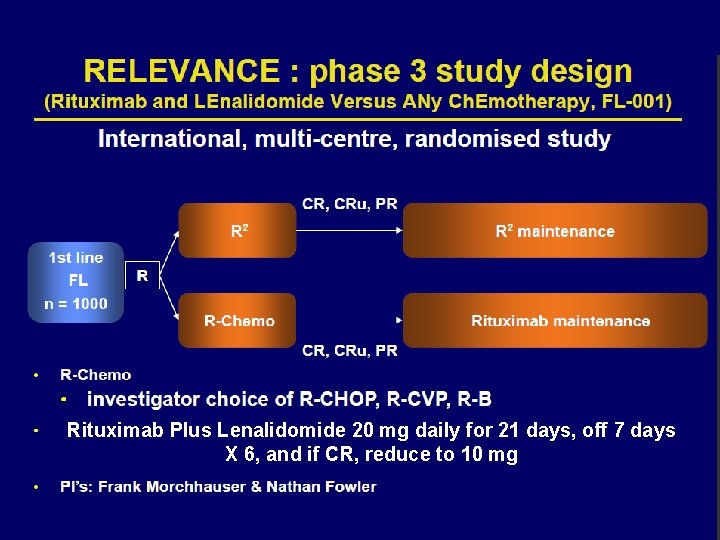
Rituximab Plus Lenalidomide 20 mg daily for 21 days, off 7 days X 6, and if CR, reduce to 10 mg

New Agents in Management of Indolent B-Cell NHLs F B Hagemeister MD Department of Lymphoma/Myeloma M D Anderson Cancer Center Las Vegas, Nevada February 27, 2014
- Slides: 51